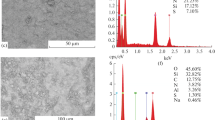
The possibility is demonstrated of adsorption extraction of vanadium compounds from acid media with modified aluminum silicate. Very fine layered aluminum silicate modified with surfactant cations (didecyldimethyl ammonium chloride) were used for extracting vanadium compounds. Sorbent modification proceeds as a result of fastening didecyldimethyl ammonium chloride molecules in the space between sorbent layers. As a result of modification, the sorbent acquires the property of selective absorption of vanadium compounds from solution. In order that the sorbent is not compacted and passes solution without hindrance, it is applied to quartz sand with a particle size of 1–3 mm treated with polyacrylamide. Vanadium compound adsorption proceeds most completely at pH 2.8–3.4, which is connected with the existence in the solution of various polyoxo compounds. The possibility is established of repeated sorbent use in adsorption and desorption cycles. The constant volume content of sorbent is 0.64 mmole/g, the dynamic exchange capacity is 0.38 mmole/g, and the static exchange capacity is 0.26 mmole/g. The solution after the desorption cycle is used for preparing pure vanadium pentoxide by hydrolytic precipitation under conditions of achieving the required concentration. Results of extracting vanadium compounds from model solutions correspond to vanadium adsorption on colloidal modified aluminum silicate from Chusovskoi Metallurgical Plant (ChMZ) washing solutions.




Similar content being viewed by others
References
A. A. Fotiev, B. V. Slobodin, and M. Ya. Khodov, Vanadates. Composition, Synthesis, Structure, Properties, Nauka, Moscow (1988).
E. M. Rabinovich and V. G. Mizin, Comprehensive Treatment of Vanadium Raw Material: Metallurgy, UrO RAN, Ekaterinburg (2005).
V. N. Muzgin, L. B. Khamzina, V. L. Zolotavin, and I. Ya. Bezrukov, Vanadium Analytical Chemistry, Nauka, Moscow (1981).
L. Zhang, X. Liu, W. Xia, and W. Zhang, “Preparation and characterization of chitosan-zirconium (IV) composite for adsorption of vanadium (V),” Int. J. Biol. Macromol., No. 64, 155–161 (2014).
T. Wang et al., “The influence of vanadate in calcined Mg/Al hydrotalcite synthesis on adsorption of vanadium (V) from aqueous solution,” Chem. Eng. J., 181, 182–188 (2012).
S. U. Nve, A. V. Shilaev, and I. D. Troshkina, “Absorption extraction of vanadium from mineralized solutions with fiber ionite,” Usp. Khim. Tekhnol., 26, No. 6 (135), 126–129 (2012).
A. V. Sviridov, E. V. Ganebnykh, G. I. Mal’tsev, and K. L. Timofeeev, “Cleaning industrial effluent with aluminosilicate sorbents,” Tsvet. Met., No. 12, 42–47 (2015).
V. I. Vigdorovich, L. E. Tsygankova, V. V. Nikolenko, and A. I. Akulov, “Extraction of copper (II) ions and phenol in flowing solution of glauconite of the Bandar area of the Tambov region,” Sorb. Prom. Protsessy, 10, No. 6, 930–937 (2010).
D. P. Ordinartsev, A. V. Svirodov, S. S. Naboichenko, and V. V. Sviridov, “Absorption extraction of vanadium from acid solutions,” Butler. Soobsh., 46, No. 2, 22–28 (2016).
A. V. Sviridov, E. V. Ganebnykh, and V. A. Elizarov, “Aluminosilicate sorbents in water cleaning technology,” Ekol. Prom. Rossii, No. 11, 28–30 (2009).
V. L. Volkov, Interstitial Phases Based on Vanadium Oxides, UNTs AN SSSR, Sverdlovsk (1987).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Metallurg, No. 10, pp. 79–82, October, 2017.
Rights and permissions
About this article
Cite this article
Ordinartsev, D.P., Sviridov, A.V., Naboichenko, S.S. et al. Sorption Extraction of Vanadium Compounds from Acid Solutions with Finely Divided Modified Aluminosilicate. Metallurgist 61, 912–916 (2018). https://doi.org/10.1007/s11015-018-0586-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11015-018-0586-1