Abstract
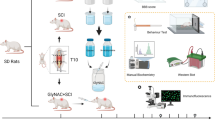
Multiple sclerosis (MS) is an autoimmune disorder characterized by the degeneration of myelin and inflammation in the central nervous system. Trans sodium crocetinate (TSC), a novel synthetic carotenoid compound, possesses antioxidant, anti-inflammatory and neuroprotective effects. This study aimed to evaluate the protective effects of TSC against the development of experimental autoimmune encephalomyelitis (EAE), a well-established model for MS. Female BALB/C57 mice were divided into different groups, including control, EAE, vehicle, TSC-treated (25, 50, and 100 mg/kg, administered via gavage) + EAE, methyl prednisone acetate + EAE, and TSC-treated (100 mg/kg, administered via gavage for 28 days) groups. EAE was induced using MOG35-55, complete Freund’s adjuvant, and pertussis toxin. In the mice spinal cord tissues, the oxidative markers (GSH and MDA) were measured using spectrophotometry and histological evaluation was performed. Mitophagic pathway proteins (PINK1and PARKIN) and inflammatory factors (IL-1β and TNF-α) were evaluated by western blot. Following 21 days post-induction, EAE mice exhibited weight loss, and the paralysis scores increased on day 13 but recovered after TSC (100 mg/kg) administration on day 16. Furthermore, TSC (50 and 100 mg/kg) reversed the altered levels of MDA and GSH in the spinal cord tissue of EAE mice. TSC (100 mg/kg) also decreased microgliosis, demyelination, and the levels of inflammatory markers IL-1β and TNF-α. Notably, TSC (100 mg/kg) modulated the mitophagy pathway by reducing PINK1 and Parkin protein levels. These findings demonstrate that TSC protects spinal cord tissue against EAE-induced MS through anti-inflammatory, antioxidant, and anti-mitophagy mechanisms.













Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Abbreviations
- ATG5:
-
Autophagy related 5
- CFA:
-
Freund’s Adjuvant, Complete
- CNS:
-
central nervous system
- COX-2:
-
Cyclooxygenase 2
- CQ:
-
clioquinol
- CSF:
-
cerebrospinal fluid
- Drp1:
-
Dynamin-related protein 1
- EAE:
-
Experimental Autoimmune Encephalomyelitis
- GSH:
-
Glutathione
- H&E:
-
Hematoxylin-eosin
- IL-1β:
-
Interleukin‐1β
- IL-6:
-
Interleukin 6
- iNOS:
-
Inducible nitric oxide synthase
- LC3:
-
Microtubule-associated protein 1 A/1B-light chain 3
- LFB:
-
Luxol fast blue
- MP:
-
Methyl prednisone acetate
- MDA:
-
Malondialdehyde
- MOG:
-
Myelin oligodendrocyte glycoprotein
- mRNA:
-
Messenger RNA
- MS:
-
Multiple Sclerosis
- MSIF:
-
MS International Federation
- NF-Κb:
-
nuclear transcription factor-kappa B
- PINK1:
-
Parkin and PTEN-induced kinase 1
- PTX:
-
Pertussis toxin
- PVDF:
-
Polyvinylidene fluoride
- ROS:
-
Reactive oxygen species
- SOD:
-
Superoxide dismutase
- TBST:
-
Tris-Buffered Saline and Tween 20
- TNF-α:
-
Tumor necrosis factor alpha
- TSC:
-
Trans sodium crocetinate
- TMEV:
-
Theiler׳s murine encephalomyelitis virus
References
Acharjee S, Nayani N, Tsutsui M, Hill MN, Ousman SS, Pittman QJ (2013) Altered cognitive-emotional behavior in early experimental autoimmune encephalitis–cytokine and hormonal correlates. Brain Behav Immun 33:164–172. https://doi.org/10.1016/j.bbi.2013.07.003
Adelmann M, Wood J, Benzel I, Fiori P, Lassmann H, Matthieu JM, Gardinier M, Dornmair K, Linington C (1995) The N-terminal domain of the myelin oligodendrocyte glycoprotein (MOG) induces acute demyelinating experimental autoimmune encephalomyelitis in the Lewis rat. J Neuroimmunol 63:17–27. https://doi.org/10.1016/0165-5728(95)00124-7
Ahmad AS, Ansari MA, Ahmad M, Saleem S, Yousuf S, Hoda MN, Islam F (2005) Neuroprotection by crocetin in a hemi-parkinsonian rat model. Pharmacol Biochem Behav 81:805–813. https://doi.org/10.1016/j.pbb.2005.06.007
Akbari-Fakhrabadi M, Najafi M, Mortazavian S, Rasouli M, Memari AH, Shidfar F (2019) Effect of saffron (Crocus sativus L.) and endurance training on mitochondrial biogenesis, endurance capacity, inflammation, antioxidant, and metabolic biomarkers in Wistar rats. J Food Biochem 43:e12946. https://doi.org/10.1111/jfbc.12946
Alavizadeh SH, Hosseinzadeh H (2014) Bioactivity assessment and toxicity of crocin: a comprehensive review. Food Chem Toxicol 64:65–80. https://doi.org/10.1016/j.fct.2013.11.016
Arano T, Imai Y (2015) Mitophagy regulated by the PINK1-Parkin pathway. Cell Death-Autophagy Apoptosis Necrosis 113–131. https://doi.org/10.5772/61284
Ashrafi G, Schwarz T (2013) The pathways of mitophagy for quality control and clearance of mitochondria. Cell Death Differ 20:31–42. https://doi.org/10.1038/cdd.2012.81
Bathaie SZ, Mousavi SZ (2010) New applications and mechanisms of action of saffron and its important ingredients. Crit Rev Food Sci Nutr 50:761–786. https://doi.org/10.1080/10408390902773003
Batoulis H, Recks MS, Addicks K, Kuerten S (2011) Experimental autoimmune encephalomyelitis–achievements and prospective advances. Apmis 119:819–830. https://doi.org/10.1111/j.1600-0463.2011.02794.x
Berkovich R (2016) Treatment of acute relapses in multiple sclerosis. Translational Neuroimmunol Multiple Scler 307–326. https://doi.org/10.1016/B978-0-12-801914-6.00024-6
Broadwater L, Pandit A, Clements R, Azzam S, Vadnal J, Sulak M, Yong VW, Freeman EJ, Gregory RB, McDonough J (2011) Analysis of the mitochondrial proteome in multiple sclerosis cortex. Acta Mol Basis Dis 1812:630–641. https://doi.org/10.1016/j.bbadis.2011.01.012
Cai J, Yi FF, Bian ZY, Shen DF, Yang L, Yan L, Tang QZ, Yang XC, Li H (2009) Crocetin protects against cardiac hypertrophy by blocking MEK-ERK1/2 signalling pathway. J Cell Mol Med 13:909–925. https://doi.org/10.1111/j.1582-4934.2008.00620.x
Chang G, Chen Y, Zhang H, Zhou W (2019) Trans sodium crocetinate alleviates ischemia/reperfusion-induced myocardial oxidative stress and apoptosis via the SIRT3/FOXO3a/SOD2 signaling pathway. Int Immunopharmacol 71:361–371. https://doi.org/10.1016/j.intimp.2019.03.056
Choi BY, Jang BG, Kim JH, Seo J-N, Wu G, Sohn M, Chung TN, Suh SW (2013) Copper/zinc chelation by clioquinol reduces spinal cord white matter damage and behavioral deficits in a murine MOG-induced multiple sclerosis model. Neurobiol Dis 54:382–391. https://doi.org/10.1016/j.nbd.2013.01.012
Colapietro A, Mancini A, D’Alessandro AM, Festuccia C (2019) Crocetin and crocin from saffron in cancer chemotherapy and chemoprevention. Curr Med Chem Anticancer Agents 19:38–47. https://doi.org/10.2174/1871520619666181231112453
Cossu D, Yokoyama K, Sato S, Noda S, Sechi LA, Hattori N (2021a) PARKIN modifies peripheral immune response and increases neuroinflammation in active experimental autoimmune encephalomyelitis (EAE). J Neuroimmunol 359:577694. https://doi.org/10.1016/j.jneuroim.2021.577694
Cossu D, Yokoyama K, Sechi LA, Hattori N (2021b) Potential of PINK1 and PARKIN proteins as biomarkers for active multiple sclerosis: a Japanese Cohort Study. Front Immunol 12. https://doi.org/10.3389/fimmu.2021.681386
Dasgupta A, Zheng J, Perrone-Bizzozero NI, Bizzozero OA (2013) Increased carbonylation, protein aggregation and apoptosis in the spinal cord of mice with experimental autoimmune encephalomyelitis. ASN Neuro 5:e00111. https://doi.org/10.1042/AN20120088
Day MJ (2005) Histopathology of EAE. Experimental models of multiple sclerosis Springer, pp 25–43
Deng J, Xiong L, Zuo Z (2015) Trans-sodium crocetinate provides neuroprotection against cerebral ischemia and reperfusion in obese mice. J Neurosci Res 93:615–622. https://doi.org/10.1002/jnr.23522
Deslauriers AM, Afkhami-Goli A, Paul AM, Bhat RK, Acharjee S, Ellestad KK, Noorbakhsh F, Michalak M, Power C (2011) Neuroinflammation and endoplasmic reticulum stress are coregulated by crocin to prevent demyelination and neurodegeneration. J Immunol 187:4788–4799. https://doi.org/10.4049/jimmunol.1004111
Dong N, Dong Z, Chen Y, Gu X (2020) Crocetin alleviates inflammation in MPTP-Induced Parkinson’s Disease models through improving mitochondrial functions. Parkinson’s Disease 2020(9864370). https://doi.org/10.1155/2020/9864370
Ebrahimzadeh A, Yousefi Moghadam S, Rahimi H, Motaghinejad M, Motevalian M, Safari S, Abbasi Mesrabadi M (2019) Crocin acts as a neuroprotective mediator against methylphenidate–induced neurobehavioral and neurochemical sequelae: possible role of the CREB-BDNF signaling pathway. Acta Neurobiol Exp (Wars) 79(4):352–366. https://doi.org/10.21307/ane-2019-033
Erhardt W, Hebestedt A, Aschenbrenner G, Pichotka B, Blümel G (1984) A comparative study with various anesthetics in mice (pentobarbitone, ketamine-xylazine, carfentanyl-etomidate). Res Exp Med 184:159–169. https://doi.org/10.1007/BF01852390
Escribano J, Alonso GL, Coca-Prados M, Fernández JA (1996) Crocin, safranal and picrocrocin from saffron (Crocus sativus L.) inhibit the growth of human cancer cells in vitro. Cancer Lett 100:23–30. https://doi.org/10.1016/0304-3835(95)04067-6
Escribano BM, Medina-Fernández FJ, Aguilar-Luque M, Agüera E, Feijoo M, Garcia-Maceira FI, Lillo R, Vieyra-Reyes P, Giraldo AI, Luque E (2017) Lipopolysaccharide binding protein and oxidative stress in a multiple sclerosis model. Neurotherapeutics 14:199–211. https://doi.org/10.1007/s13311-016-0480-0
Eteghadi MR, Nasehi M, Vaseghi S, Hesami–Tackallou S (2021) The effect of Crocin on TFAM and PGC–1α expression and Catalaseand Superoxide dismutase activities following cholestasis–inducedneuroinflammation in the striatum of male Wistar rats. Metab Brain Dis 36(7):1791–1801. https://doi.org/10.1007/s11011-021-00748-x
Feng P, Li Q, Liu L, Wang S, Wu Z, Tao Y, Huang P, Wang P (2022) Crocetin prolongs recovery period of DSS-induced colitis via altering intestinal microbiome and increasing intestinal permeability. Int J Mol Sci 23:3832. https://doi.org/10.3390/ijms23073832
Fletcher JM, Lalor S, Sweeney C, Tubridy N, Mills K (2010) T cells in multiple sclerosis and experimental autoimmune encephalomyelitis. Clin Exp Immunol 162:1–11. https://doi.org/10.1111/j.1365-2249.2010.04143.x
Giatti S, Boraso M, Abbiati F, Ballarini E, Calabrese D, Santos-Galindo M, Rigolio R, Pesaresi M, Caruso D, Viviani B (2013) Multimodal analysis in acute and chronic experimental autoimmune encephalomyelitis. J Neuroimmune Pharmacol 8:238–250. https://doi.org/10.1007/s11481-012-9385-9
Gibson-Corley KN, Olivier AK, Meyerholz DK (2013) Principles for valid histopathologic scoring in research. Vet Pathol 50:1007–1015. https://doi.org/10.1177/0300985813485099
Gibson-Corley KN, Boyden AW, Leidinger MR, Lambertz AM, Ofori-Amanfo G, Naumann PW, Goeken JA, Karandikar NJ (2016) A method for histopathological study of the multifocal nature of spinal cord lesions in murine experimental autoimmune encephalomyelitis. Peer J 4:e1600. https://doi.org/10.7717/peerj.1600
Gonçalves FB, Morais VA (2021) PINK1: a bridge between mitochondria and Parkinson’s disease. Life 11:371. https://doi.org/10.3390/life11050371
Grigoriadis N, Van Pesch V (2015) A basic overview of multiple sclerosis immunopathology. Eur J Neurol 22:3–13. https://doi.org/10.1111/ene.12798
Hashemi M, Hosseinzadeh H (2019) A comprehensive review on biological activities and toxicology of crocetin. Food Chem Toxicol 130:44–60. https://doi.org/10.1016/j.fct.2019.05.017
Hassanpour M, Cheraghi O, Laghusi D, Nouri M, Panahi Y (2020) The relationship between ANT1 and NFL with autophagy and mitophagy markers in patients with multiple sclerosis. J Clin Neurosci 78:307–312. https://doi.org/10.1016/j.jocn.2020.04.122
Hosseini A, Razavi BM, Hosseinzadeh H (2018) Pharmacokinetic properties of saffron and its active components. Eur J Drug Metab Pharmacokinet 43:383–390. https://doi.org/10.1007/s13318-017-0449-3
Hosseinzadeh H, Younesi HM (2002) Antinociceptive and anti-inflammatory effects of Crocus sativus L. stigma and petal extracts in mice. BMC Pharmacol Toxicol 2:1–8. https://doi.org/10.1186/1471-2210-2-7
Huang WJ, Chen WW, Zhang X (2017) Multiple sclerosis: pathology, diagnosis and treatments. Exp Ther Med 13:3163–3166. https://doi.org/10.3892/etm.2017.4410
Khoshandam A, Razavi BM, Hosseinzadeh H (2022) Interaction of saffron and its constituents with Nrf2 signaling pathway: a review. Iran J Basic Med Sci 25. https://doi.org/10.22038/IJBMS.2022.61986.13719
Kuhlmann T, Ludwin S, Prat A, Antel J, Brück W, Lassmann H (2017) An updated histological classification system for multiple sclerosis lesions. Acta Neuropathol 133:13–24. https://doi.org/10.1007/s00401-016-1653-y
Kurschus F (2015) T cell mediated pathogenesis in EAE: molecular mechanisms. Biomed J 38. https://doi.org/10.4103/2319-4170.155590
Kuznetsov AV, Margreiter R (2009) Heterogeneity of mitochondria and mitochondrial function within cells as another level of mitochondrial complexity. Int J Mol Sci 10:1911–1929. https://doi.org/10.3390/ijms10041911
Kyong Nyon N, Young-Min P, Hoon-Ji J, Jung Yeon L, Byung Duk M, Seong-Uk P, Woo-Sang J, Ki-Ho C, Ji-Ho P, Insug K, Joung-Woo H, Eunjoo HL (2010) Anti-inflammatory effects of crocin and crocetin in rat brain microglial cells. Eur J Pharmacol 1;648(1–3):110-6. https://doi.org/10.1016/j.ejphar.2010.09.003
Lassmann H (2018) Multiple sclerosis pathology. Cold Spring Harb Perspect Med 8:a028936. https://doi.org/10.1101/cshperspect.a028936
Lassmann H, van Horssen J (2011) The molecular basis of neurodegeneration in multiple sclerosis. FEBS Lett 585:3715–3723. https://doi.org/10.1016/j.febslet.2011.08.004
Lee DH, Gold R, Linker RA (2012) Mechanisms of oxidative damage in multiple sclerosis and neurodegenerative diseases: therapeutic modulation via fumaric acid esters. Int J Mol Sci 13:11783–11803. https://doi.org/10.3390/ijms130911783
Li W, Feng J, Gao C, Wu M, Du Q, Tsoi B, Wang Q, Yang D, Shen J (2019a) Nitration of Drp1 provokes mitophagy activation mediating neuronal injury in experimental autoimmune encephalomyelitis. Free Radic Biol Med 143:70–83. https://doi.org/10.1016/j.freeradbiomed.2019.07.037
Li YH, Xu F, Thome R, Guo MF, Sun ML, Song GB, Li R, Chai Z, Ciric B, Rostami A (2019b) Mdivi-1, a mitochondrial fission inhibitor, modulates T helper cells and suppresses the development of experimental autoimmune encephalomyelitis. J Neuroinflammation 16:1–11. https://doi.org/10.1186/s12974-019-1542-0
Li Z, Liu F, He X, Yang X, Shan F, Feng J (2019c) Exosomes derived from mesenchymal stem cells attenuate inflammation and demyelination of the central nervous system in EAE rats by regulating the polarization of microglia. Int Immunopharmacol 67:268–280. https://doi.org/10.1016/j.intimp.2018.12.001
Li Q, Feng P, Lin S, Xu Z, Zhao J, Chen Z, Luo Z, Tao Y, Chen S, Wang P (2023) Crocetin confers neuroprotection and is anti-inflammatory in rats with induced glaucoma. Mol Biol Rep 50:1321–1331. https://doi.org/10.1007/s11033-022-08102-9
Liu C, Huang Y, Pang M, Yang Y, Li S, Liu L, Shu T, Zhou W, Wang X, Rong L (2015) Tissue-engineered regeneration of completely transected spinal cord using induced neural stem cells and gelatin-electrospun poly (lactide-co-glycolide)/polyethylene glycol scaffolds. PLoS ONE 10:e0117709. https://doi.org/10.1371/journal.pone.0117709
Liu Gy, Wu Y, Kong Fy, Ma S, Fu Ly, Geng J (2021) BMSCs differentiated into neurons, astrocytes and oligodendrocytes alleviated the inflammation and demyelination of EAE mice models. PLoS ONE 16:e0243014. https://doi.org/10.1371/journal.pone.0243014
Luthra R, Roy A (2022) Role of medicinal plants against neurodegenerative diseases. Curr Pharm Biotechnol 23:123–139. https://doi.org/10.2174/1389201022666210211123539
Manabe H, Okonkwo DO, Gainer JL, Clarke RH, Lee KS (2010) Protection against focal ischemic injury to the brain by trans-sodium crocetinate. JNS 113:802–809. https://doi.org/10.3171/2009.10.JNS09562
Mehri S, Abnous K, Mousavi SH, Shariaty VM, Hosseinzadeh H (2012) Neuroprotective effect of crocin on acrylamide-induced cytotoxicity in PC12 cells. Cell Mol Neurobiol 32:227–235. https://doi.org/10.1007/s10571-011-9752-8
Mehri S, Abnous K, Khooei A, Mousavi SH, Shariaty VM, Hosseinzadeh H (2015) Crocin reduced acrylamide-induced neurotoxicity in Wistar rat through inhibition of oxidative stress. Iran J Basic Med Sci 18: 902
Melnyk JP, Wang S, Marcone MF (2010) Chemical and biological properties of the world’s most expensive spice: Saffron. Food Res Int 43:1981–1989. https://doi.org/10.1016/j.foodres.2010.07.033
Mohammadzadeh L, Abnous K, Razavi BM, Hosseinzadeh H (2020) Crocin-protected malathion-induced spatial memory deficits by inhibiting TAU protein hyperphosphorylation and antiapoptotic effects. Nutr Neurosci 23(3):221–236. https://doi.org/10.1080/1028415X.2018.1492772
Mohler IIIER, Gainer JL, Whitten K, Eraso LH, Thanaporn PK, Bauer T (2011) Evaluation of trans sodium crocetinate on safety and exercise performance in patients with peripheral artery disease and intermittent claudication. Vasc Med 16:346–353. https://doi.org/10.1177/1358863X11422742
Moloudizargari M, Asghari MH, Ghobadi E, Fallah M, Rasouli S, Abdollahi M (2017) Autophagy, its mechanisms and regulation: implications in neurodegenerative diseases. Ageing Res Rev 40:64–74. https://doi.org/10.1016/j.arr.2017.09.005
Moron MS, Depierre JW, Mannervik B (1979) Levels of glutathione, glutathione reductase and glutathione S-transferase activities in rat lung and liver. Biochim. Biophys Acta Gen Subj 582:67–78. https://doi.org/10.1016/0304-4165(79)90289-7
Morris RL, Hollenbeck PJ (1993) The regulation of bidirectional mitochondrial transport is coordinated with axonal outgrowth. J Cell Sci 104:917–927. https://doi.org/10.1242/jcs.104.3.917
Nam KN, Park YM, Jung HJ, Lee JY, Min BD, Park SU, Jung WS, Cho KH, Park JH, Kang I (2010) Anti-inflammatory effects of crocin and crocetin in rat brain microglial cells. Eur J Pharmacol 648:110–116. https://doi.org/10.1016/j.ejphar.2010.09.003
Nourazarian A, Khaki-Khatibi F, Nikanfar M, Laghousi D, Vatankhah AM, Moharami S (2020) Evaluation of the diagnostic and predictive value of serum levels of ANT1, ATG5, and Parkin in multiple sclerosis. Clin Neurol Neurosurg 197:106197. https://doi.org/10.1016/j.clineuro.2020.106197
Ochiai T, Soeda S, Ohno S, Tanaka H, Shoyama Y, Shimeno H (2004) Crocin prevents the death of PC-12 cells through sphingomyelinase-ceramide signaling by increasing glutathione synthesis. Neurochem Int 44:321–330. https://doi.org/10.1016/S0197-0186(03)00174-8
Ochiai T, Shimeno H, Mishima Ki, Iwasaki K, Fujiwara M, Tanaka H, Shoyama Y, Toda A, Eyanagi R, Soeda S (2007) Protective effects of carotenoids from saffron on neuronal injury in vitro and in vivo. Biochim Biophys Acta 1770:578–584. https://doi.org/10.1016/j.bbagen.2006.11.012
Ohno Y, Nakanishi T, Umigai N, Tsuruma K, Shimazawa M, Hara H (2012) Oral administration of crocetin prevents inner retinal damage induced by N-methyl-D-aspartate in mice. Eur J Pharmacol 690:84–89. https://doi.org/10.1016/j.ejphar.2012.06.035
Okonkwo DO, Wagner J, Melon DE, Alden T, Stone JR, Helm GA, Jane Sr JA (2003) Trans-sodium crocetinate increases oxygen delivery to brain parenchyma in rats on oxygen supplementation. Neurosci Lett 352:97–100. https://doi.org/10.1016/j.neulet.2003.08.044
Patergnani S, Castellazzi M, Bonora M, Marchi S, Casetta I, Pugliatti M, Giorgi C, Granieri E, Pinton P (2018) Autophagy and mitophagy elements are increased in body fluids of multiple sclerosis-affected individuals. J Neurol Neurosurg Psychiatry 89:439–441. https://doi.org/10.1136/jnnp-2017-316234
Peng T, Li S, Liu L, Yang C, Farhan M, Chen L, Su Q, Zheng W (2022) Artemisinin attenuated ischemic stroke induced cell apoptosis through activation of ERK1/2/CREB/BCL-2 signaling pathway in vitro and in vivo. Int J Biol Sci 18(11):4578–4594. https://doi.org/10.7150/ijbs.69892
Peyravi A, Yazdanpanahi N, Nayeri H, Hosseini SA (2020) The effect of endurance training with crocin consumption on the levels of MFN2 and DRP1 gene expression and glucose and insulin indices in the muscle tissue of diabetic rats. J Food Biochem 44(e13125). https://doi.org/10.1111/jfbc.13125
Procaccini C, De Rosa V, Pucino V, Formisano L, Matarese G (2015) Animal models of multiple sclerosis. Eur J Pharmacol 759:182–191. https://doi.org/10.1016/j.ejphar.2015.03.042
Rameshrad M, Razavi BM, Hosseinzadeh H (2018) Saffron and its derivatives, crocin, crocetin and safranal: a patent review. Expert Opin Ther Pat 28:147–165. https://doi.org/10.1080/13543776.2017.1355909
Rao SV, Yenisetti SC, Rajini PS (2016) Evidence of neuroprotective effects of saffron and crocin in a Drosophila model of parkinsonism. Neurotoxicology 52:230–242. https://doi.org/10.1016/j.neuro.2015.12.010
Sajad M, Zargan J, Chawla R, Umar S, Sadaqat M, Khan HA (2009) Hippocampal neurodegeneration in experimental autoimmune encephalomyelitis (EAE): potential role of inflammation activated myeloperoxidase. Mol Cell Biochem 328:183–188. https://doi.org/10.1007/s11010-009-0088-3
Sawcer S, Franklin RJ, Ban M (2014) Multiple sclerosis genetics. Lancet Neurol 13:700–709. https://doi.org/10.1016/S1474-4422(14)70041-9
Sheehan J, Ionescu A, Pouratian N, Hamilton DK, Schlesinger D, Oskouian RJ, Sansur C (2008) Use of trans sodium crocetinate for sensitizing glioblastoma multiforme to radiation. J Neurosurg 108:972–978. https://doi.org/10.3171/JNS/2008/108/5/0972
Sheehan J, Sherman J, Cifarelli C, Jagannathan J, Dassoulas K, Olson C, Rainey J, Han S (2009) Effect of trans sodium crocetinate on brain tumor oxgenation. J Neurosurg 111:226–229. https://doi.org/10.3171/2009.3.JNS081339
Sloka J, Stefanelli M (2005) The mechanism of action of methylprednisolone in the treatment of multiple sclerosis. Mult Scler 11:425–432. https://doi.org/10.1191/1352458505ms1190oa
Stennett AK, Gainer JL (2004) TSC for hemorrhagic shock: effects on cytokines and blood pressure. Shock 22:569–574. https://doi.org/10.1097/01.shk.0000144133.21524.1e
Stennett AK, Murray RJ, Roy JW, Gainer JL (2007) Trans-sodium crocetinate and hemorrhagic shock. Shock 28:339–344. https://doi.org/10.1097/shk.0b013e3180487b2d
Stevanovic I, Ninkovic M, Mancic B, Milivojevic M, Stojanovic I, Ilic T, Vujovic M, Djukic M (2020) Compensatory neuroprotective response of thioredoxin reductase against oxidative-nitrosative stress induced by experimental autoimmune encephalomyelitis in rats: modulation by theta burst stimulation. Molecules 25:3922. https://doi.org/10.3390/molecules25173922
Trapp BD, Nave KA (2008) Multiple sclerosis: an immune or neurodegenerative disorder? Annu Rev Neurosci 31:247–269. https://doi.org/10.1146/annurev.neuro.30.051606.094313
Twig G, Shirihai OS (2011) The interplay between mitochondrial dynamics and mitophagy. Antioxid Redox Signal 14:1939–1951. https://doi.org/10.1089/ars.2010.3779
Wang GH, Liu Y, Wu XB, Lu Y, Liu J, Qin YR, Li T, Duan HF (2016) Neuroprotective effects of human umbilical cord–derived mesenchymal stromal cells combined with nimodipine against radiation-induced brain injury through inhibition of apoptosis. Cytotherapy 18:53–64. https://doi.org/10.1016/j.jcyt.2015.10.006
Wang X, Weidling I, Koppel S, Menta B, Ortiz JP, Kalani A, Wilkins HM, Swerdlow RH (2020a) Detection of mitochondria-pertinent components in exosomes. Mitochondrion 55:100–110. https://doi.org/10.1016/j.mito.2020.09.006
Wang Y, Yu W, Shi C, Hu P (2020b) Crocetin attenuates sepsis-induced cardiac dysfunction via regulation of inflammatory response and mitochondrial function. Front Physiol 11: 514. https://doi.org/10.3389/fphys.2020.00514. eCollection 2020
Witte ME, Bol JG, Gerritsen WH, van der Valk P, Drukarch B, van Horssen J, Wilhelmus MM (2009) Parkinson’s disease-associated parkin colocalizes with Alzheimer’s disease and multiple sclerosis brain lesions. Neurobiol Dis 36:445–452. https://doi.org/10.1016/j.nbd.2009.08.009
Wu H, Li Y, Zhang Q, Wang H, Xiu W, Xu P, Deng Y, Huang W, Wang DO (2023) Crocetin antagonizes parthanatos in ischemic stroke via inhibiting NOX2 and preserving mitochondrial hexokinase-I. Cell Death Dis 14:50. https://doi.org/10.1038/s41419-023-05581-x
Xiong Y, Wang J, Yu H, Zhang X, Miao C (2015) Anti-asthma potential of crocin and its effect on MAPK signaling pathway in a murine model of allergic airway disease. Immunopharmacol Immunotoxicol 37:236–243. https://doi.org/10.3109/08923973.2015.1021356
Yang T, Zheng Q, Wang S, Fang L, Liu L, Zhao H, Wang L, Fan Y (2017) Effect of catalpol on remyelination through experimental autoimmune encephalomyelitis acting to promote Olig1 and Olig2 expressions in mice. BMC Complement Altern Med 17:1–15. https://doi.org/10.1186/s12906-017-1642-2
Yang W, Qiu X, Wu Q, Chang F, Zhou T, Zhou M, Pei J (2023) Active constituents of saffron (Crocus sativus L.) and their prospects in treating neurodegenerative diseases. Exp Ther Med 25:1–14. https://doi.org/10.3892/etm.2023.11934
Yaribeygi H, Noroozadeh A, Mohammadi MT, Johnston TP, Sahebkar A (2019) Crocin improves oxidative stress by potentiating intrinsic anti-oxidant defense systems in pancreatic cells during uncontrolled hyperglycemia. Pharmacopuncture 22:83. https://doi.org/10.3831/KPI.2019.22.010
Zeinali M, Zirak MR, Rezaee SA, Karimi G, Hosseinzadeh H (2019) Immunoregulatory and anti-inflammatory properties of Crocus sativus (Saffron) and its main active constituents: a review. Iran J Basic Med Sci 22:334. https://doi.org/10.22038/ijbms.2019.34365.8158
Zhang C, Ma J, Fan L, Zou Y, Dang X, Wang K, Song J (2015) Neuroprotective effects of safranal in a rat model of traumatic injury to the spinal cord by anti-apoptotic, anti-inflammatory and edema-attenuating. Tissue Cell 47:291–300. https://doi.org/10.1016/j.tice.2015.03.007
Zhang R, Zeng H, Zhang Y, Chen K, Zhang C, Song C, Fang L, Xu Z, Yang K, Jin B (2016) CD226 ligation protects against EAE by promoting IL-10 expression via regulation of CD4 + T cell differentiation. Oncotarget 7:19251. https://doi.org/10.18632/oncotarget.7834
Zhang J, Wang Y, Dong X, Liu J (2018) Crocetin attenuates inflammation and amyloid-β accumulation in APPsw transgenic mice. Immun Ageing 15:1–8. https://doi.org/10.1186/s12979-018-0132-9
Zhang SY, Gui LN, Liu YY, Shi S, Cheng Y (2020) Oxidative stress marker aberrations in multiple sclerosis: a meta-analysis study. Front Neurosci 14:823. https://doi.org/10.3389/fnins.2020.00823
Zheng YQ, Liu JX, Wang JN, Xu L (2007) Effects of crocin on reperfusion-induced oxidative/nitrative injury to cerebral microvessels after global cerebral ischemia. Brain Res 1138:86–94. https://doi.org/10.1016/j.brainres.2006.12.064
Zidan A, Hedya SE, Elfeky DM, Abdin AA (2018) The possible anti-apoptotic and antioxidant effects of acetyl l-carnitine as an add-on therapy on a relapsing-remitting model of experimental autoimmune encephalomyelitis in rats. Biomed Pharmacother 103:1302–1311. https://doi.org/10.1016/j.biopha.2018.04.173
Web reference:
Acknowledgements
The authors are thankful to the Vice-Chancellor of Research, Mashhad University of Medical Sciences for financial support (Grant/Award Number: 971984). The results presented in this paper are part of a Ph.D. thesis.
Funding
This work was supported by the Mashhad University of Medical Sciences. Start-up funding to A/Prof. Hossein Hosseinzadeh.
Author information
Authors and Affiliations
Contributions
Sara Banaeeyeh: Investigation, Data curation, Data analyses, Writing, Original draft preparation, and Editing. Zahra Moosavi: Pathology analysis. Amir Afkhami-Goli: Supervision, EAE induction, Reviewing, and Editing. Bibi Marjan Razavi: Conceptualization, Supervision, Data analyses, Reviewing and Editing. Hossein Hosseinzade: Conceptualization, Supervision, Data analyses, Reviewing and Editing. All the authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Authorship statement
All persons who meet authorship criteria are listed as authors, and all authors certify that they have participated sufficiently in the work to take public responsibility for the content, including participation in the concept, design, analysis, writing, or revision of the manuscript. Furthermore, each author certifies that this material or similar material has not been and will not be submitted to or published in any other publication before its appearance in the Metabolic Brain Disease Journal.
Consent for publication
All the authors have consented to publishing this manuscript.
Ethics statement
The animal study was reviewed and approved by the Mashhad University of Medical Sciences Animal Care and Use Committee (971984), and every protocol used in this study was followed.
Consent to participate
Not applicable to animal studies.
Competing interest
The financial support for this study from Mashhad University of Medical Sciences is disclosed. The authors declare no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Banaeeyeh, S., Afkhami-Goli, A., Moosavi, Z. et al. Anti-inflammatory, antioxidant and anti-mitophagy effects of trans sodium crocetinate on experimental autoimmune encephalomyelitis in BALB/C57 mice. Metab Brain Dis (2024). https://doi.org/10.1007/s11011-024-01349-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11011-024-01349-0