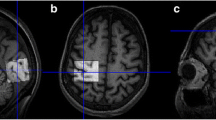
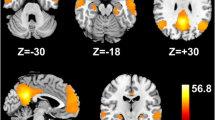
Abstract
Hepatic encephalopathy (HE) is a common neurological manifestation of liver cirrhosis and is characterized by an increase of ammonia in the brain accompanied by a disrupted neurotransmitter balance, including the GABAergic and glutamatergic systems. The aim of this study is to investigate metabolic abnormalities in the cerebello-thalamo-cortical system of HE patients using GABA-edited MRS and links between metabolite levels, disease severity, critical flicker frequency (CFF), motor performance scores, and blood ammonia levels. GABA-edited MRS was performed in 35 participants (16 controls, 19 HE patients) on a clinical 3 T MRI system. MRS voxels were placed in the right cerebellum, left thalamus, and left motor cortex. Levels of GABA+ and of other metabolites of interest (glutamine, glutamate, myo-inositol, glutathione, total choline, total NAA, and total creatine) were assessed. Group differences in metabolite levels and associations with clinical metrics were tested. GABA+ levels were significantly increased in the cerebellum of patients with HE. GABA+ levels in the motor cortex were significantly decreased in HE patients, and correlated with the CFF (r = 0.73; p < .05) and motor performance scores (r = -0.65; p < .05). Well-established HE-typical metabolite patterns (increased glutamine, decreased myo-inositol and total choline) were confirmed in all three regions and were closely linked to clinical metrics. In summary, our findings provide further evidence for alterations in the GABAergic system in the cerebellum and motor cortex in HE. These changes were accompanied by characteristic patterns of osmolytes and oxidative stress markers in the cerebello-thalamo-cortical system. These metabolic disturbances are a likely contributor to HE motor symptoms in HE.
Graphical abstract

In patients with hepatic encephalopathy, GABA+ levels in the cerebello-thalamo-cortical loop are significantly increased in the cerebellum and significantly decreased in the motor cortex. GABA+ levels in the motor cortex strongly correlate with critical flicker frequency (CFF) and motor performance score (pegboard test tPEG), but not blood ammonia levels (NH3).





Similar content being viewed by others

Data availability
The data will be made available by the authors upon reasonable request.
Abbreviations
- HE:
-
Hepatic encephalopathy
- MRS:
-
Magnetic resonance spectroscopy
- ROS:
-
Reactive oxidative species
- CFF:
-
Critical flicker frequency
- tPEG:
-
Timed pegboard test
- NH3 :
-
Blood ammonia levels
- GSH:
-
Glutathione
- Gln:
-
Glutamine
- Glu:
-
Glutamate
- mI:
-
Myo-inositol
- NAAG:
-
N-acetylaspartylglutamate
- NAA:
-
N-acetylaspartate
- sI:
-
Scyllo-inositol
- MM:
-
Macromolecules
- DKNTMN:
-
LCModel baseline knot spacing control parameter
- GAP:
-
LCModel gap control parameter
- GM:
-
Gray matter
- WM:
-
White matter
- CSF:
-
Cerebrospinal fluid
- tCr:
-
Total creatine
- FWHM:
-
Full-width half maximum
- CRLB:
-
Cramér rao lower bound
- tNAA:
-
Total NAA
References
Allen M, Poggiali D, Whitaker K, Marshall TR, Kievit RA (2019) Raincloud plots: a multi-platform tool for robust data visualization. Wellcome Open Res 4:63. https://doi.org/10.12688/wellcomeopenres.15191.1
Barantin L, Pape AL, Akoka S (1997) A new method for absolute quantitation MRS metabolites. Magn Reson Med 38:179–182. https://doi.org/10.1002/mrm.1910380203
Barkhuijsen H, de Beer R, van Ormondt D (1987) Improved algorithm for noniterative time-domain model fitting to exponentially damped magnetic resonance signals. J Magn Reson 1969(73):553–557. https://doi.org/10.1016/0022-2364(87)90023-0
Baumgarten TJ, Oeltzschner G, Hoogenboom N, Wittsack H-J, Schnitzler A, Lange J (2016) Beta peak frequencies at rest correlate with endogenous GABA+/Cr concentrations in sensorimotor cortex areas. PloS One 11:e0156829. https://doi.org/10.1371/journal.pone.0156829
Behar KL, Rothman DL, Petersen KF, Hooten M, Delaney R, Petroff OAC, Shulman GI, Navarro V, Petrakis IL, Charney DS, Krystal JH (1999) Preliminary evidence of low cortical gaba levels in localized 1 H-MR spectra of alcohol-dependent and hepatic encephalopathy patients. Am J Psychiatry 156:952–954. https://doi.org/10.1176/ajp.156.6.952
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B Methodol 57:289–300
Binesh N, Huda A, Bugbee M, Gupta R, Rasgon N, Kumar A, Green M, Han S, Thomas MA (2005) Adding another spectral dimension to 1H magnetic resonance spectroscopy of hepatic encephalopathy. J Magn Reson Imaging 21:398–405. https://doi.org/10.1002/jmri.20291
Butz M, Timmermann L, Braun M, Groiss SJ, Wojtecki L, Ostrowski S, Krause H, Pollok B, Gross J, Südmeyer M, Kircheis G, Häussinger D, Schnitzler A (2010) Motor impairment in liver cirrhosis without and with minimal hepatic encephalopathy. Acta Neurol Scand 122:27–35. https://doi.org/10.1111/j.1600-0404.2009.01246.x
Cao Q, Xu S, Zhu W, Zhu W, Wan Y, Wang J, Chen X, Pi L, Lobo MK, Ren B, Ying Z, Morris M (2018) Decreased taurine and creatine in the thalamus may relate tominimal hepatic encephalopathy in ethanol-fed mice using proton magnetic resonance spectroscopy. J Nucl Med 59:345–345
Cauli O, Llansola M, Erceg S, Felipo V (2006) Hypolocomotion in rats with chronic liver failure is due to increased glutamate and activation of metabotropic glutamate receptors in substantia nigra. J Hepatol 45:654–661. https://doi.org/10.1016/j.jhep.2006.06.019
Cauli O, Mansouri MT, Agusti A, Felipo V (2009a) Hyperammonemia Increases GABAergic Tone in the Cerebellum but Decreases It in the Rat Cortex. Gastroenterology 136:1359-1367.e2. https://doi.org/10.1053/j.gastro.2008.12.057
Cauli O, Rodrigo R, Llansola M, Montoliu C, Monfort P, Piedrafita B, El Mlili N, Boix J, Agustí A, Felipo V (2009b) Glutamatergic and gabaergic neurotransmission and neuronal circuits in hepatic encephalopathy. Metab Brain Dis 24:69–80. https://doi.org/10.1007/s11011-008-9115-4
Choi I-Y, Andronesi OC, Barker P, Bogner W, Edden RAE, Kaiser LG, Lee P, Marjańska, M, Terpstra M, de Graaf RA (2021) Spectral editing in 1H magnetic resonance spectroscopy: experts’ consensus recommendations. NMR Biomed n/a: e4411. https://doi.org/10.1002/nbm.4411
Cichoz-Lach H, Michalak A (2013) Current pathogenetic aspects of hepatic encephalopathy and noncirrhotic hyperammonemic encephalopathy. World J Gastroenterol. https://doi.org/10.3748/wjg.v19.i1.26 (Baishideng Publishing Group Inc)
Cudalbu C, Taylor-Robinson SD (2019) Brain edema in chronic hepatic encephalopathy. J Clin Exp Hepatol 9:362–382. https://doi.org/10.1016/j.jceh.2019.02.003
de Graaf RA (2018) In Vivo NMR Spectroscopy: Principles and Techniques, 3rd ed. Wiley. https://doi.org/10.1002/9781119382461
Edden RAE, Puts NAJ, Harris AD, Barker PB, Evans CJ (2014) Gannet: a batch-processing tool for the quantitative analysis of gamma-aminobutyric acid–edited MR spectroscopy spectra. J Magn Reson Imaging JMRI 40:1445–1452
Felipo V (2013) Hepatic encephalopathy: effects of liver failure on brain function. Nat Rev Neurosci 14:851–858. https://doi.org/10.1038/nrn3587
Ferenci P, Blei AT, Lockwood AH, Mullen K, Tarter R, Weissenborn K (2002) Hepatic encephalopathy - definition, nomenclature, diagnosis, and quantification: final report of the working party at the 11th world congresses of gastroenterology, Vienna, 1998. Hepatology 35:716–721. https://doi.org/10.1053/jhep.2002.31250
Fowler CF, Madularu D, Dehghani M, Devenyi GA, Near J (2021) Longitudinal quantification of metabolites and macromolecules reveals age- and sex-related changes in the healthy Fischer 344 rat brain. Neurobiol Aging 101:109–122. https://doi.org/10.1016/j.neurobiolaging.2020.12.012
Friston K (2007) Statistical Parametric Mapping: The Analysis of Functional Brain Images. Academic Press, Book
Gasparovic C, Song T, Devier D, Bockholt HJ, Caprihan A, Mullins PG, Posse S, Jung RE, Morrison LA (2006) Use of tissue water as a concentration reference for proton spectroscopic imaging. Magn Reson Med 55:1219–1226. https://doi.org/10.1002/mrm.20901
Hassan SS, Baumgarten TJ, Ali AM, Füllenbach N-D, Jördens MS, Häussinger D, Butz M, Schnitzler A, Groiss SJ (2019) Cerebellar inhibition in hepatic encephalopathy. Clin Neurophysiol 130:886–892. https://doi.org/10.1016/J.CLINPH.2019.02.020
Häussinger D, Butz M, Schnitzler A, Görg B (2021) Pathomechanisms in hepatic encephalopathy. Biol Chem 402:1087–1102. https://doi.org/10.1515/hsz-2021-0168
Häussinger D, Dhiman RK, Felipo V, Görg B, Jalan R, Kircheis G, Merli M, Montagnese S, Romero-Gomez M, Schnitzler A, Taylor-Robinson SD, Vilstrup H (2022) Hepatic encephalopathy. Nat Rev Dis Primer 8:1–22. https://doi.org/10.1038/s41572-022-00366-6
Heaton RK (2004) Revised comprehensive norms for an expanded Halstead-Reitan Battery: Demographically adjusted neuropsychological norms for African American and Caucasian adults. Psychological Assessment Resources, Lutz, FL
Henry PG, Dautry C, Hantraye P, Bloch G (2001) Brain gaba editing without macromolecule contamination. Magn Reson Med 45:517–520. https://doi.org/10.1002/1522-2594(200103)45:3%3c517::AID-MRM1068%3e3.0.CO;2-6
Hermann B, Rudler M, Galanaud D, Thabut D, Weiss N (2019) Magnetic resonance spectroscopy: a surrogate marker of hepatic encephalopathy? J Hepatol 71:1055–1057. https://doi.org/10.1016/j.jhep.2019.07.008
Hilgier W, Węgrzynowicz M, Ruszkiewicz, Oja JSS, Saransaari P, Albrecht J (2010) Direct exposure to ammonia and hyperammonemia increase the extracellular accumulation and degradation of astroglia-derived glutathione in the rat prefrontal cortex. Toxicol Sci 117:163–168. https://doi.org/10.1093/toxsci/kfq171
Hui SCN, Saleh MG, Zöllner HJ, Oeltzschner G, Fan H, Li Y, Song Y, Jiang H, Near J, Lu H, Mori S, Edden RAE (2022) MRSCloud: a cloud-based MRS tool for basis set simulation. Magn Reson Med 88:1994–2004. https://doi.org/10.1002/mrm.29370
JAMA (2013) World Medical Association declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA - J Am Med Assoc 310:2191–2194. https://doi.org/10.1001/jama.2013.281053
Kajla N (2020) Comparison of arterial vs venous ammonia levels in hepatic encephalopathy. Ameis Curr Trends Diagn Treat 3:59–63. https://doi.org/10.5005/jp-journals-10055-0076
Kircheis G, Häussinger D, Wettstein M, Schnitzler A, Timmermann L (2002) Critical flicker frequency for quantification of low-grade hepatic encephalopathy. Hepatology 35:357–366. https://doi.org/10.1053/jhep.2002.30957
Kircheis G, Hilger N, Häussinger D (2014) Value of Critical Flicker Frequency and psychometric hepatic encephalopathy score in diagnosis of low-grade hepatic encephalopathy. Gastroenterology 146:961-969.e11. https://doi.org/10.1053/j.gastro.2013.12.026
Klose U (1990) In vivo proton spectroscopy in presence of eddy currents. Magn Reson Med 14:26–30. https://doi.org/10.1002/mrm.1910140104
Klove H (1963) Clinical neuropsychology (1963) the medical clinics of North America. FM Forster Ed 47:1647–1658
Kreis R, Farrow N, Ross BD (1991) Localized 1H NMR spectroscopy in patients with chronic hepatic encephalopathy. Analysis of changes in cerebral glutamine, choline and inositols. NMR Biomed 4:109–116. https://doi.org/10.1002/nbm.1940040214
Larsen RJ, Gagoski B, Morton SU, Ou Y, Vyas R, Litt J, Grant PE, Sutton BP (2021) Quantification of magnetic resonance spectroscopy data using a combined reference: application in typically developing infants. NMR Biomed. 34:e4520. https://doi.org/10.1002/nbm.4520
Lien Y-HH, Michaelis T, Moats RA, Ross BD (1994) Scyllo-inositol depletion in hepatic encephalopathy. Life Sci 54:1507–1512. https://doi.org/10.1016/0024-3205(94)90018-3
Lin A, Andronesi O, Bogner W, Choi I-Y, Coello E, Cudalbu C, Juchem C, Kemp GJ, Kreis R, Krššák M, Lee P, Maudsley AA, Meyerspeer M, Mlynarik V, Near J, Öz G, Peek AL, Puts NA, Ratai E-M, Tkáč I, Mullins PG, Spectroscopy EWG, on R.S. for M., (2021) Minimum Reporting Standards for in vivo Magnetic Resonance Spectroscopy (MRSinMRS): experts’ consensus recommendations. NMR Biomed. 34:e4484. https://doi.org/10.1002/nbm.4484
Lockwood AH (2004) Blood ammonia levels and hepatic encephalopathy. Metab Brain Dis 19:345–349. https://doi.org/10.1023/B:MEBR.0000043980.74574.eb
Lockwood AH, Yap EW, Wong WH (1991) Cerebral ammonia metabolism in patients with severe liver disease and minimal hepatic encephalopathy. J Cereb Blood Flow Metab Off J Int Soc Cereb Blood Flow Metab 11:337–341. https://doi.org/10.1038/jcbfm.1991.67
Mallet M, Desplats V, Bouzbib C, Sultanik P, Alioua I, Rudler M, Weiss N, Thabut D (2022) Blood ammonia in patients with chronic liver diseases: a better defined role in clinical practice. Anal. Biochem 657:114873. https://doi.org/10.1016/j.ab.2022.114873
Menshchikov P, Ivantsova A, Manzhurtsev A, Ublinskiy M, Yakovlev A, Melnikov I, Kupriyanov D, Akhadov T, Semenova N (2020) Separate N-acetyl aspartyl glutamate, N-acetyl aspartate, aspartate, and glutamate quantification after pediatric mild traumatic brain injury in the acute phase. Magn Reson Med 84:2918–2931. https://doi.org/10.1002/mrm.28332
Mescher M, Merkle H, Kirsch J, Garwood M, Gruetter R (1998) Simultaneous in vivo spectral editing and water suppression. NMR Biomed 11:266–272. https://doi.org/10.1002/(SICI)1099-1492(199810)11:6%3c266::AID-NBM530%3e3.0.CO;2-J
Michels L, Schulte-Vels T, Schick M, O’Gorman RL, Zeffiro T, Hasler G, Mueller-Pfeiffer C (2014) Prefrontal GABA and glutathione imbalance in posttraumatic stress disorder: preliminary findings. Psychiatry Res Neuroimaging 224:288–295. https://doi.org/10.1016/j.pscychresns.2014.09.007
Miller JJ, Cochlin L, Clarke K, Tyler DJ (2017) Weighted averaging in spectroscopic studies improves statistical power. Magn Reson Med 78:2082–2094. https://doi.org/10.1002/mrm.26615
Murthy CRK, Bender AS, Dombro RS, Bai G, Norenberg MD (2000) Elevation of glutathione levels by ammonium ions in primary cultures of rat astrocytes. Neurochem Int 37:255–268. https://doi.org/10.1016/S0197-0186(00)00028-0
Near J, Edden RAE, Evans CJ, Paquin R, Harris AD, Jezzard P (2015) Frequency and phase drift correction of magnetic resonance spectroscopy data by spectral registration in the time domain. Magn Reson Med 73:44–50. https://doi.org/10.1002/mrm.25094
Near J, Harris AD, Juchem C, Kreis R, Marjańska M, Öz G, Slotboom J, Wilson M, Gasparovic C (2020) Preprocessing, analysis and quantification in single-voxel magnetic resonance spectroscopy: experts’ consensus recommendations. NMR Biomed. 34:e4257. https://doi.org/10.1002/nbm.4257
Oeltzschner G, Butz M, Baumgarten TJ, Hoogenboom N, Wittsack H-J, Schnitzler A (2015) Low visual cortex GABA levels in hepatic encephalopathy: links to blood ammonia, critical flicker frequency, and brain osmolytes. Metab Brain Dis 30:1429–1438. https://doi.org/10.1007/s11011-015-9729-2
Oeltzschner G, Butz M, Wickrath F, Wittsack H-J, Schnitzler A (2016) Covert hepatic encephalopathy: elevated total glutathione and absence of brain water content changes. Metab Brain Dis 31:517–527. https://doi.org/10.1007/s11011-015-9760-3
Oeltzschner G, Saleh MG, Rimbault D, Mikkelsen M, Chan KL, Puts NAJ, Edden RAE (2019) Advanced Hadamard-encoded editing of seven low-concentration brain metabolites: principles of HERCULES. Neuroimage 185:181–190. https://doi.org/10.1016/j.neuroimage.2018.10.002
Oeltzschner G, Zöllner HJ, Hui SCN, Mikkelsen M, Saleh MG, Tapper S, Edden RAE (2020) Osprey: open-source processing, reconstruction & estimation of magnetic resonance spectroscopy data. J Neurosci Methods 343:108827. https://doi.org/10.1016/j.jneumeth.2020.108827
Palmieri L, Pardo B, Lasorsa FM, del Acro A, Kobayashi K, Iijima M, Runswick MJ, Walker JE, Saheki T, Satrústegui J, Palmieri F (2001) Citrin and aralar1 are Ca2+-stimulated aspartate/glutamate transporters in mitochondria. EMBO J 20:5060–5069. https://doi.org/10.1093/emboj/20.18.5060
Pasek J (2021) Weights: weighting and weighted statistics (manual) [WWW Document]. https://cran.r-project.org/web/packages/weights/index.html. Accessed 28 Apr 2021
Petroff OAC, Hyder F, Rothman DL, Mattson RH (2001) Topiramate rapidly raises brain GABA in epilepsy patients. Epilepsia 42:543–548. https://doi.org/10.1046/j.1528-1157.2001.18800.x
Prakash R, Mullen KD (2010) Mechanisms, diagnosis and management of hepatic encephalopathy. Nat Rev Gastroenterol Hepatol 7:515–525. https://doi.org/10.1038/nrgastro.2010.116
Provencher S (2020) LCModel & LCMgui User’s Manual [WWW Document]. LCModel LCMgui Users Man. URL http://s-provencher.com/pub/LCModel/manual/manual.pdf. Accessed 28 April 2020
Provencher SW (2001) Automatic quantitation of localized in vivo1H spectra with LCModel. NMR Biomed 14:260–264. https://doi.org/10.1002/nbm.698
Rae CD (2014) A guide to the metabolic pathways and function of metabolites observed in human brain 1 H magnetic resonance spectra. Neurochem Res 39:1–36
Rao KVR, Norenberg MD (2001) Cerebral energy metabolism in hepatic encephalopathy and hyperammonemia. Metab Brain Dis 16:67–78. https://doi.org/10.1023/A:1011666612822
Rothman DL, Petroff OA, Behar KL, Mattson RH (1993) Localized 1H NMR measurements of gamma-aminobutyric acid in human brain in vivo. Proc Natl Acad Sci 90:5662–5666. https://doi.org/10.1073/pnas.90.12.5662
Sathyasaikumar KV, Swapna I, Reddy PVB, Murthy ChRK, Dutta Gupta A, Senthilkumaran B, Reddanna P (2007) Fulminant hepatic failure in rats induces oxidative stress differentially in cerebral cortex, cerebellum and pons medulla. Neurochem Res 32:517–524. https://doi.org/10.1007/s11064-006-9265-x
Schafer DF, Anthony Jones E (1982) Hepatic encephalopathy and the γ-aminobutyric-acid neurotransmitter system. The Lancet 319:18–20. https://doi.org/10.1016/S0140-6736(82)92559-4
Shah NJ, Neeb H, Kircheis G, Engels P, Häussinger D, Zilles K (2008) Quantitative cerebral water content mapping in hepatic encephalopathy. Neuroimage 41:706–717. https://doi.org/10.1016/j.neuroimage.2008.02.057
Shah NJ, Neeb H, Zaitsev M, Steinhoff S, Kircheis G, Amunts K, Häussinger D, Zilles K (2003) Quantitative T1Mapping of hepatic encephalopathy using magnetic resonance imaging. Hepatology 38:1219–1226. https://doi.org/10.1053/jhep.2003.50477
Simicic D, Cudalbu C, Pierzchala K (2022) Overview of oxidative stress findings in hepatic encephalopathy: from cellular and ammonium-based animal models to human data. Anal Biochem 654:114795. https://doi.org/10.1016/j.ab.2022.114795
Simpson R, Devenyi GA, Jezzard P, Hennessy TJ, Near J (2015) Advanced processing and simulation of MRS data using the FID appliance (FID-A)—An open source, MATLAB-based toolkit. Magn Reson Med 77:23–33. https://doi.org/10.1002/mrm.26091
Singhal A, Nagarajan R, Hinkin CH, Kumar R, Sayre J, Elderkin-Thompson V, Huda A, Gupta RK, Han S-H, Thomas MA (2010) Two-dimensional MR spectroscopy of minimal hepatic encephalopathy and neuropsychological correlates in vivo. J Magn Reson Imaging 32:35–43. https://doi.org/10.1002/jmri.22216
Stagg CJ, Bachtiar V, Johansen-Berg H (2011) What are we measuring with GABA magnetic resonance spectroscopy? Commun Integr Biol 4:573–575. https://doi.org/10.4161/cib.4.5.16213
Terpstra M, Henry P-G, Gruetter R (2003) Measurement of reduced glutathione (GSH) in human brain using LCModel analysis of difference-edited spectra. Magn Reson Med 50:19–23. https://doi.org/10.1002/mrm.10499
Thoma R, Mullins P, Ruhl D, Monnig M, Yeo RA, Caprihan A, Bogenschutz M, Lysne P, Tonigan S, Kalyanam R, Gasparovic C (2011) Perturbation of the glutamate-glutamine system in alcohol dependence and remission. Neuropsychopharmacology 36:1359–1365. https://doi.org/10.1038/npp.2011.20
Timmermann L, Gross J, Butz M, Kircheis G, Häussinger D, Schnitzler A (2003) Mini-asterixis in hepatic encephalopathy induced by pathologic thalamo-motor-cortical coupling. Neurology 61:689–692. https://doi.org/10.1212/01.WNL.0000078816.05164.B1
Viola A, Nicoli F, Denis B, Confort-Gouny S, Fur YL, Ranjeva J-P, Viout P, Cozzone PJ (2004) High cerebral scyllo-inositol: a new marker of brain metabolism disturbances induced by chronic alcoholism. Magn Reson Mater Phys Biol Med 17:47–61. https://doi.org/10.1007/s10334-004-0044-x
Węgrzynowicz M, Hilgier W, Dybel A, Oja SS, Saransaari P, Albrecht J (2007) Upregulation of cerebral cortical glutathione synthesis by ammonia in vivo and in cultured glial cells: the role of cystine uptake. Neurochem Int Mechanisms Neurodegeneration 50:883–889. https://doi.org/10.1016/j.neuint.2006.12.003
Wijtenburg SA, Knight-Scott J (2011) Very short echo time improves the precision of glutamate detection at 3T in 1H magnetic resonance spectroscopy. J Magn Reson Imaging 34:645–652. https://doi.org/10.1002/jmri.22638
Wilson M, Andronesi O, Barker PB, Bartha R, Bizzi A, Bolan PJ, Brindle KM, Choi I-Y, Cudalbu C, Dydak U, Emir UE, Gonzalez RG, Gruber S, Gruetter R, Gupta RK, Heerschap A, Henning A, Hetherington HP, Huppi PS, Hurd RE, Kantarci K, Kauppinen RA, Klomp DWJ, Kreis R, Kruiskamp MJ, Leach MO, Lin AP, Luijten PR, Marjańska M, Maudsley AA, Meyerhoff DJ, Mountford CE, Mullins PG, Murdoch JB, Nelson SJ, Noeske R, Öz G, Pan JW, Peet AC, Poptani H, Posse S, Ratai E-M, Salibi N, Scheenen TWJ, Smith ICP, Soher BJ, Tkáč I, Vigneron DB, Howe FA (2019) Methodological consensus on clinical proton MRS of the brain: review and recommendations. Magn Reson Med 82:527–550. https://doi.org/10.1002/mrm.27742
Zeng G, Penninkilampi R, Chaganti J, Montagnese S, Brew BJ, Danta M (2020) Meta-analysis of magnetic resonance spectroscopy in the diagnosis of hepatic encephalopathy. Neurology 94:e1147–e1156. https://doi.org/10.1212/WNL.0000000000008899
Zöllner HJ, Butz M, Jördens M, Füllenbach N-D, Häussinger D, Schmitt B, Wittsack H-J, Schnitzler A (2019) Chemical exchange saturation transfer imaging in hepatic encephalopathy. NeuroImage Clin 22:101743. https://doi.org/10.1016/j.nicl.2019.101743
Zöllner HJ, Butz M, Kircheis G, Klinker S, Häussinger D, Schmitt B, Schnitzler A, Wittsack H-J (2018) Ammonia-weighted imaging by chemical exchange saturation transfer MRI at 3 T. NMR Biomed. 31:e3947. https://doi.org/10.1002/nbm.3947
Zöllner HJ, Považan M, Hui SCN, Tapper S, Edden RAE, Oeltzschner G (2021a) Comparison of different linear-combination modeling algorithms for short-TE proton spectra. NMR Biomed 34:e4482. https://doi.org/10.1002/nbm.4482
Zöllner HJ, Tapper S, Hui SCN, Barker PB, Edden RAE, Oeltzschner G (2021b) Comparison of linear combination modeling strategies for edited magnetic resonance spectroscopy at 3 T. NMR Biomed: e4618. https://doi.org/10.1002/nbm.4618
Acknowledgements
The authors would like to thank Erika Rädisch (Department of Diagnostic and Interventional Radiology, University Hospital Düsseldorf) and Dr. Gerald Antoch (Department of Diagnostic and Interventional Radiology, University Hospital Düsseldorf) for support with the MR measurements.
Funding
This study was supported by the Sonderforschungsbereich (SFB) 974 (TP B07) of the German Research foundation and by NIH grant R21 EB033516. The funding sources were not involved in the study design, collection, analysis, and interpretation of the presented data.
Author information
Authors and Affiliations
Contributions
Helge J. Zöllner: Data acquisition, Formal Analysis, Investigation, Writing – Original Draft, Writing – Review & Editing, Visualization. Thomas Thiel: Data acquisition, Writing – Review & Editing, Visualization. Markus S. Jördens: Patient Recruitment, Clinical Evaluation, Formal Analysis, Writing – Review & Editing. Sinyeob Ahn: Data acquisition – Sequence Development, Writing – Review & Editing. Lena M. Wilms: Clinical Evaluation, Formal Analysis. Alexandra Ljimani: Clinical Evaluation, Formal Analysis. Dieter Häussinger: Patient Recruitment, Formal Analysis, Writing – Review & Editing, Funding acquisition. Markus Butz: Data acquisition, Formal Analysis, Writing – Review & Editing, Project administration, Supervision, Funding acquisition. Hans-Jörg Wittsack: Conceptualization, Formal Analysis, Supervision, Writing – Review & Editing. Alfons Schnitzler: Formal Analysis, Project administration, Writing – Review & Editing, Supervision, Funding acquisition. Georg Oeltzschner: Conceptualization, Methodology, Writing – Review & Editing, Supervision, Funding acquisition.
Corresponding author
Ethics declarations
Ethics approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the local institutional review board of the Heinrich-Heine University Düsseldorf (Date 01/2016; study number 5179R).
Consent to participants
All participants gave written informed consent before the examination.
Consent to publish
The authors affirm that human research participants provided informed consent for the publication of the MR images and behavioral data in the figures.
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zöllner, H.J., Thiel, T.A., Füllenbach, ND. et al. J-difference GABA-edited MRS reveals altered cerebello-thalamo-cortical metabolism in patients with hepatic encephalopathy. Metab Brain Dis 38, 1221–1238 (2023). https://doi.org/10.1007/s11011-023-01174-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-023-01174-x