Abstract
Background
IL-10 knockout (KO) mice can be protected against experimental autoimmune encephalomyelitis (EAE) with low-dose estrogen (E2) treatment similar to wild type (WT) mice, indicating that IL-10 is not required for E2-induced EAE protection. Our previous study demonstrated that E2 treatment induced an increase in programmed death ligands 1 (PD-L1) and 2 (PD-L2) on monocytes and macrophages in the periphery and within the CNS. In this study, we selectively inhibited the function of PD-L1 and PD-L2 to evaluate their critical role in maintaining E2-induced protection against EAE in IL-10-KO mice.
Methods
This study used female IL-10 KO mice pre-treated with either E2 or sham pellets seven days prior to induction of EAE and subsequently treated with Vehicle or antibodies to PD-L1, PD-L2 or respective isotype controls. Mice were scored daily for EAE severity over 21 days post-EAE induction. Cells from the spleen and brain were evaluated by flow cytometry.
Results
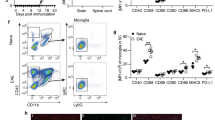
Differences in EAE severity were assessed in E2 and sham pre-treated IL-10-KO mice treated with α-PD-L1 or α-PD-L2 antibodies over the course of disease compared to treatment with Vehicle or isotype control antibodies. The results revealed real-time development of severe EAE in E2-pre-treated IL-10-KO mice treated with α-PD-L1 but not α-PD-L2 antibodies, mediated in part by increased percentages of activated CD74+CD11b+ myeloid cells in spleen and brain as well as splenic B-cells, T-cells and CD73+ cells.
Conclusion
These results demonstrate unequivocally that PD-L1 but not PD-L2 was required to retain the inhibitory effects of E2 on clinical EAE scores in female IL-10-KO mice and further implicate the emergence of the MIF/CD74 axis as a contributing pathogenic mechanism.






Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Abramsky O (1994) Pregnancy and multiple sclerosis. Ann Neurol 36. https://doi.org/10.1002/ana.410360712. Suppl:S38-41
Bebo BFJ, Fyfe-Johnson A, Adlard K et al (2001) Low-dose estrogen therapy ameliorates experimental autoimmune encephalomyelitis in two different inbred mouse strains. J Immunol 166:2080–2089. https://doi.org/10.4049/jimmunol.166.3.2080
Benedek G, Meza-Romero R, Andrew S et al (2013) Partial MHC class II constructs inhibit MIF/CD74 binding and downstream effects. Eur J Immunol 43:1309–1321. https://doi.org/10.1002/eji.201243162
Benedek G, Zhang J, Bodhankar S et al (2016) Estrogen induces multiple regulatory B cell subtypes and promotes M2 microglia and neuroprotection during experimental autoimmune encephalomyelitis. J Neuroimmunol 293:45–53. https://doi.org/10.1016/j.jneuroim.2016.02.009
Benedek G, Zhang J, Nguyen H et al (2017) Novel feedback loop between M2 macrophages/microglia and regulatory B cells in estrogen-protected EAE mice. J Neuroimmunol 305:59–67. https://doi.org/10.1016/j.jneuroim.2016.12.018
Ben-Nun A, Kaushansky N, Kawakami N et al (2014) From classic to spontaneous and humanized models of multiple sclerosis: impact on understanding pathogenesis and drug development. J Autoimmun 54:33–50. https://doi.org/10.1016/j.jaut.2014.06.004
Bettelli E, Das MP, Howard ED et al (1998) IL-10 is critical in the regulation of autoimmune encephalomyelitis as demonstrated by studies of IL-10- and IL-4-deficient and transgenic mice. J Immunol 161:3299–3306
Bodhankar S, Galipeau D, Vandenbark AA, Offner H (2013) PD-1 Interaction with PD-L1 but not PD-L2 on B-cells mediates Protective Effects of Estrogen against EAE. J Clin Cell Immunol 4:143. https://doi.org/10.4172/2155-9899.1000143
Brooks DG, Ha S-J, Elsaesser H et al (2008) IL-10 and PD-L1 operate through distinct pathways to suppress T-cell activity during persistent viral infection. Proc Natl Acad Sci U S A 105:20428–20433. https://doi.org/10.1073/pnas.0811139106
Campanella M, Sciorati C, Tarozzo G, Beltramo M (2002) Flow cytometric analysis of inflammatory cells in ischemic rat brain. Stroke 33:586–592. https://doi.org/10.1161/hs0202.103399
Candando KM, Lykken JM, Tedder TF (2014) B10 cell regulation of health and disease. Immunol Rev 259:259–272. https://doi.org/10.1111/imr.12176
Confavreux C, Hutchinson M, Hours MM et al (1998) Rate of pregnancy-related relapse in multiple sclerosis. Pregnancy in multiple sclerosis Group. N Engl J Med 339:285–291. https://doi.org/10.1056/NEJM199807303390501
Cross AH, Trotter JL, Lyons J (2001) B cells and antibodies in CNS demyelinating disease. J Neuroimmunol 112:1–14. https://doi.org/10.1016/s0165-5728(00)00409-4
Cua DJ, Groux H, Hinton DR et al (1999) Transgenic interleukin 10 prevents induction of experimental autoimmune encephalomyelitis. J Exp Med 189:1005–1010. https://doi.org/10.1084/jem.189.6.1005
Du C, Sriram S (2002) Increased severity of experimental allergic encephalomyelitis in lyn-/- mice in the absence of elevated proinflammatory cytokine response in the central nervous system. J Immunol 168:3105–3112. https://doi.org/10.4049/jimmunol.168.6.3105
Fillatreau S, Sweenie CH, McGeachy MJ et al (2002) B cells regulate autoimmunity by provision of IL-10. Nat Immunol 3:944–950. https://doi.org/10.1038/ni833
Haghmorad D, Salehipour Z, Nosratabadi R et al (2016) Medium-dose estrogen ameliorates experimental autoimmune encephalomyelitis in ovariectomized mice. J Immunotoxicol 13:885–896. https://doi.org/10.1080/1547691X.2016.1223768
Ito A, Bebo BF, Matejuk A et al (2001) Estrogen treatment down-regulates TNF-alpha production and reduces the severity of experimental autoimmune encephalomyelitis in cytokine knockout mice. J Immunol 167:542–552. https://doi.org/10.4049/jimmunol.167.1.542
Kaku H, Cheng KF, Al-Abed Y, Rothstein TL (2014) A novel mechanism of B cell-mediated immune suppression through CD73 expression and adenosine production. J Immunol 193:5904–5913. https://doi.org/10.4049/jimmunol.1400336
Khan D, Ansar Ahmed S (2015) The Immune System is a natural target for Estrogen Action: Opposing Effects of Estrogen in two prototypical Autoimmune Diseases. Front Immunol 6:635. https://doi.org/10.3389/fimmu.2015.00635
Korniotis S, Gras C, Letscher H et al (2016) Treatment of ongoing autoimmune encephalomyelitis with activated B-cell progenitors maturing into regulatory B cells. Nat Commun 7:12134. https://doi.org/10.1038/ncomms12134
Liu HY, Buenafe AC, Matejuk A et al (2002) Estrogen inhibition of EAE involves effects on dendritic cell function. J Neurosci Res 70:238–248. https://doi.org/10.1002/jnr.10409
Matsushita T, Horikawa M, Iwata Y, Tedder TF (2010) Regulatory B cells (B10 cells) and regulatory T cells have independent roles in controlling experimental autoimmune encephalomyelitis initiation and late-phase immunopathogenesis. J Immunol 185:2240–2252. https://doi.org/10.4049/jimmunol.1001307
Matsushita T, Yanaba K, Bouaziz J-D et al (2008) Regulatory B cells inhibit EAE initiation in mice while other B cells promote disease progression. J Clin Invest 118:3420–3430. https://doi.org/10.1172/JCI36030
Morales LBJ, Loo KK, Liu H-B et al (2006) Treatment with an estrogen receptor alpha ligand is neuroprotective in experimental autoimmune encephalomyelitis. J Neurosci 26:6823–6833. https://doi.org/10.1523/JNEUROSCI.0453-06.2006
Pennati A, Ng S, Wu Y et al (2016) Regulatory B cells induce formation of IL-10-Expressing T cells in mice with autoimmune neuroinflammation. J Neurosci 36:12598–12610. https://doi.org/10.1523/JNEUROSCI.1994-16.2016
Polanczyk M, Zamora A, Subramanian S et al (2003) The protective effect of 17beta-estradiol on experimental autoimmune encephalomyelitis is mediated through estrogen receptor-alpha. Am J Pathol 163:1599–1605. https://doi.org/10.1016/s0002-9440(10)63516-x
Polanczyk MJ, Hopke C, Vandenbark AA, Offner H (2006) Estrogen-mediated immunomodulation involves reduced activation of effector T cells, potentiation of Treg cells, and enhanced expression of the PD-1 costimulatory pathway. J Neurosci Res 84:370–378. https://doi.org/10.1002/jnr.20881
Polanczyk MJ, Hopke C, Vandenbark AA, Offner H (2007) Treg suppressive activity involves estrogen-dependent expression of programmed death-1 (PD-1). Int Immunol 19:337–343. https://doi.org/10.1093/intimm/dxl151
Polanczyk MJ, Jones RE, Subramanian S et al (2004) T lymphocytes do not directly mediate the protective effect of estrogen on experimental autoimmune encephalomyelitis. Am J Pathol 165:2069–2077. https://doi.org/10.1016/S0002-9440(10)63257-9
Saxena A, Khosraviani S, Noel S et al (2015) Interleukin-10 paradox: a potent immunoregulatory cytokine that has been difficult to harness for immunotherapy. Cytokine 74:27–34. https://doi.org/10.1016/j.cyto.2014.10.031
Seifert HA, Gerstner G, Kent G et al (2019) Estrogen-induced compensatory mechanisms protect IL-10-deficient mice from developing EAE. J Neuroinflammation 16:195. https://doi.org/10.1186/s12974-019-1588-z
Trenova AG, Slavov GS, Manova MG et al (2013) Female sex hormones and cytokine secretion in women with multiple sclerosis. Neurol Res 35:95–99. https://doi.org/10.1179/1743132812Y.0000000120
Whitacre CC (2001) Sex differences in autoimmune disease. Nat Immunol 2:777–780. https://doi.org/10.1038/ni0901-777
Wiedrick J, Meza-Romero R, Gerstner G et al (2021) Sex differences in EAE reveal common and distinct cellular and molecular components. Cell Immunol 359:104242. https://doi.org/10.1016/j.cellimm.2020.104242
Wolf SD, Dittel BN, Hardardottir F, Janeway CA (1996) Experimental autoimmune encephalomyelitis induction in genetically b cell-deficient mice. J Exp Med 184:2271–2278. https://doi.org/10.1084/jem.184.6.2271
Zhang J, Benedek G, Bodhankar S et al (2015a) IL-10 producing B cells partially restore E2-mediated protection against EAE in PD-L1 deficient mice. J Neuroimmunol 285:129–136. https://doi.org/10.1016/j.jneuroim.2015.06.002
Zhang J, Lapato A, Bodhankar S et al (2015b) Treatment with IL-10 producing B cells in combination with E2 ameliorates EAE severity and decreases CNS inflammation in B cell-deficient mice. Metab Brain Dis 30:1117–1127. https://doi.org/10.1007/s11011-015-9661-5
Acknowledgements
The authors would like to thank the VAPORHCS Veterinary staff for housing and maintaining mouse environments.
Funding
This work was supported by the National Institute of Allergy and Infectious Diseases R21AI148490 (HO) and R42AI122574 (AV), the Department of Veterans Affairs, Veterans Health Administration, Office of Research and Development, Biomedical Laboratory Research and Development Merit Review Award 2I01 BX000226 (AV), BLR&D Merit Review for Pre-IND studies of Drugs and Biologics Award 5I01 BX005112 (AV), Senior Research Career Scientist Award 1IK6BX004209 (AV). The contents do not represent the views of the Department of Veterans Affairs or the United States Government.
Author information
Authors and Affiliations
Contributions
HO contributed to experimental design and wrote and reviewed the manuscript. DL performed experiments, collected and analyzed data, and helped with manuscript preparation. RMR and AAV reviewed and helped with manuscript preparation. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Ethics approval and consent to participate
All applicable international, national and/or institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted. This article does not contain any studies with human participants performed by any of the authors.
Consent to publish
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Offner, H., Lockwood, D., Meza-Romero, R. et al. PD-L1 is required for estrogen-induced protection against severe EAE in IL-10 deficient mice1. Metab Brain Dis 38, 589–599 (2023). https://doi.org/10.1007/s11011-022-01129-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-022-01129-8