Abstract
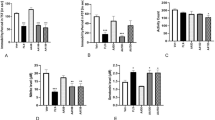
In the present study, antidepressant like effect of amantadine was studied in mice using tail suspension test (TST) and forced swim test (FST). Further the effect of amantadine treatment on the brain nitrite, glutamate and serotonin levels was also determined. Amantadine (AMT) (50, 100 and 150 mg/kg, i.p.) was administered to the mice and after 30 min of administration the mice were subjected to TST and FST. It was observed that the administration of AMT (100 and 150 mg/kg, i.p.) decreased the immobility period of mice in TST and FST significantly as compared to control. The findings from the whole brain neurochemical assay suggested that the AMT (100 and 150 mg/kg, i.p.) treatment decreased the brain nitrite and glutamate level but increased the brain serotonin significantly as compared to control. Further the influence of NO-cGMP signaling in the antidepressant like effect of amantadine was also determined. It was observed that the NO donor (i.e. L-Arginine (50 mg/kg, i.p.)) potentiated the effect elicited by AMT (50 mg/kg, i.p.) in FST and decreased the brain serotonin level of AMT (50 mg/kg, i.p.) treated mice. Further the pretreatment of cGMP modulator (i.e. Sildenafil (1 mg/kg, i.p.)) potentiated the behavioral effect elicited by AMT (50 mg/kg, i.p.) in TST and FST and decreased the brain nitrite and glutamate level of AMT (50 mg/kg, i.p.) treated mice. In conclusion, amantadine exerted antidepressant like effect in mice and NO-cGMP signaling influences the antidepressant like effect of amantadine in mice.



Similar content being viewed by others
Availability of data and material
Data was collected following the behavioral and neurochemical assay and put for the statistical analysis.
Code Availability
None.
References
Almeida RC, Felisbino CS, Lopez MG, Rodrigues AL, Gabilan NH (2006) Evidence for the involvement of L-arginine-nitric oxide-cyclic guanosine monophosphate pathway in the antidepressant-like effect of memantine in mice. Behav Brain Res 168:318–322. doi: https://doi.org/10.1016/j.bbr.2005.11.023
Bernt E, Bergmeyer HU (1965) L-glutamate UV assay with glutamate dehydrogenase and NAD. Methods of enzymatic analysis, 2nd edn. Academic Press, New York, pp 1704–1708. https://doi.org/10.1007/BF00390373
Bishop A, Anderson JE (2005) NO signaling in the CNS: from the physiological to the pathological. Toxicology 208(2):193–205. doi: https://doi.org/10.1016/j.tox.2004.11.034
Blanpied TA, Clarke RJ, Johnson JW (2005) Amantadine inhibits NMDA receptors by accelerating channel closure during channel block. J Neurosci 25(13):3312–3322. doi: https://doi.org/10.1523/JNEUROSCI.4262-04.2005
Cárdenas A, Moro MA, Hurtado O, Leza JC, Lizasoain I (2005) Dual role of nitric oxide in adult neurogenesiss. Brain Res Brain Res Rev 50(1):1–6. doi: https://doi.org/10.1016/j.brainresrev.2005.03.006
Chen HS, Pellegrini JW, Aggarwal SK, Lei SZ, Warach S, Jensen FE, Lipton SA (1992) Open-channel block of N-methyl-D-aspartate (NMDA) responses by memantine: therapeutic advantage against NMDA receptor-mediated neurotoxicity. J Neurosci 12:4427–4436. doi: https://doi.org/10.1523/JNeurosci
de Bittencourt-Navarrete RE, do Nascimento IC, Santiago MF, Mendez-Otero R (2009) NMDA receptor blockade alters the intracellular distribution of neuronal nitric oxide synthase in the superficial layers of the rat superior colliculus. Braz J Med Biol Res 42(2):189–196. doi: https://doi.org/10.1590/s0100-879x2009000200007
Dhir A, Kulkarni SK (2007) Involvement of nitric oxide (NO) signaling pathway in the antidepressant action of bupropion, a dopamine reuptake inhibitor. Eur J Pharmacol 568:177–185. doi: https://doi.org/10.1016/j.ejphar.2007.04.028
Dhir A, Kulkarni SK (2011) Nitric oxide, and major depression. Nitric Oxide 24(3):125–131. doi: https://doi.org/10.1016/j.niox.2011.02.002
Downey D, Dutta A, McKie S, Dawson GR, Dourish CT, Craig K, Smith MA, McCarthy DJ, Harmer CJ, Goodwin GM, Williams S (2016) Comparing the actions of lanicemine and ketamine in depression: key role of the anterior cingulate. Eur Neuropsychopharmacol 26(6):994–1003. doi: https://doi.org/10.1016/j.euroneuro.2016.03.006
Erceg S, Monfort P, Hernandez-Viadel M, Llansola M, Montoliu C, Felipo V (2005) Restoration of learning ability in hyperammonemic rats by increasing extracellular cGMP in brain. Brain Res 1036(1–2):115–121. doi: https://doi.org/10.1016/j.brainres.2004.12.045
Fasipe OJ (2018) Neuropharmacological classification of antidepressant agents based on their mechanisms of action. Arch Med Health Sci 6:81. doi: https://doi.org/10.4103/amhs.amhs_7_18
Ghasemi M, Sadeghipour H, Mosleh A, Sadeghipour HR, Mani AR, Dehpour AR (2008) Nitric oxide involvement in the antidepressant-like effects of acute lithium administration in the mouse forced swimming test. Eur Neuropsychopharmacol 18:323–332. doi: https://doi.org/10.1016/j.euroneuro.2007.07.011
Green LC, Wagner DA, Glogowski J, Skipper PL, Wishnok JS, Tannenbaum SR (1982) Analysis of nitrate, nitrite, and [15 N] nitrate in biological fluids. Anal Biochem 126:131–138. doi: https://doi.org/10.1016/0003-2697(82)90118-x
Guix FX, Uribesalgo I, Coma M, Munoz FJ (2005) The physiology and pathophysiology of nitric oxide in the brain. Prog Neurobiol 76:126–152. doi: https://doi.org/10.1016/j.pneurobio.2005.06.001
Gupta G, Jia Jia T, Yee Woon L, Kumar Chellappan D, Candasamy M, Dua K (2015) Pharmacological Evaluation of Antidepressant-Like Effect of Genistein and Its Combination with Amitriptyline: An Acute and Chronic Study. Adv Pharmacol Sci 2015:164943. doi: https://doi.org/10.1155/2015/164943
Harkin AJ, Bruce KH, Craft B, Paul IA (1999) Nitric oxide synthase inhibitors have antidepressant-like properties in mice. 1. Acute treatments are active in the forced swim test. Eur J Pharmacol 372(3):207–213. doi: https://doi.org/10.1016/s0014-2999(99)00191-0
Harmer CJ, Cowen PJ (2013) ‘It’s the way that you look at it’—a cognitive neuropsychological account of SSRI action in depression. Phil Trans R Soc B: Biol Sci 368:20120407. doi: https://doi.org/10.1098/rstb.2012.0407
Hazzaa SM, Ewida SF, Elbatsh M (2014) Influence of the Phosphodiesterase type 5 inhibitor, sildenafil, on some behavioral and central biochemical changes on chronic restraint stress in rats. Med J Cairo Univ 82(1):483–493
Hoyer D, Clarke DE, Fozard JR, Hartig PR, Martin GR, Mylecharane EJ, Saxena PR, Humphrey PP (1994) International Union of Pharmacology classification of receptors for 5-hydroxytryptamine (Serotonin). Pharmacol Rev 46(2):157–203
Kaehler ST, Singewald N, Sinner C, Philippu A (1999) Nitric oxide modulates the release of serotonin in the rat hypothalamus. Brain Res 835(2):346–349. doi: https://doi.org/10.1016/s0006-8993(99)01599-1
Khakpai F, Ebrahimi-Ghiri M, Alijanpour S, Zarrindast MR (2019) Ketamine-induced antidepressant like effects in mice: A possible involvement of cannabinoid system. Biomed Pharmacother 112:108717. doi: https://doi.org/10.1016/j.biopha.2019.108717
Kiss JP, Szasz BK, Fodor L, Mike A, Lenkey N, Kurkó D, Nagy J, Vizi ES (2012) GluN2B-containing NMDA receptors as possible targets for the neuroprotective and antidepressant effects of fluoxetine. Neurochem Int 60(2):170–176. doi: https://doi.org/10.1016/j.neuint.2011.12.005
Kucukibrahimoglu E, Saygin MZ, Caliskan M, Kaplan OK, Unsal C, Goren MZ (2009) The change in plasma GABA, glutamine, and glutamate levels in fluoxetine-or S-citalopram-treated female patients with major depression. Eur J Clin Pharmacol 65:571–577. doi: https://doi.org/10.1007/s00228-009-0650-7
Kyratsas C, Dalla C, Anderzhanova E, Polissidis A, Kokras N, Konstantinides K, Papadopoulou-Daifoti Z (2013) Experimental evidence for sildenafil’s action in the central nervous system: dopamine and serotonin changes in the medial preoptic area and nucleus accumbens during sexual arousal. J Sex Med 10:719–729. https://doi.org/10.1111/j.1743-6109.2012.03000.x
Lustig HS, Ahern KVB, Greenberg DA (1992) Antiparkinsonian drugs and in vitro excitotoxicity. Brain Res 597:148–150
Mathews DC, Henter ID, Zarate CA (2012) Targeting the glutamatergic system to treat major depressive disorder: rationale and progress to date. Drugs 72(10):1313–1333. doi: https://doi.org/10.2165/11633130-000000000-00000
Mayer A, Szasz BK, Kiss JP (2009) Inhibitory effect of antidepressants on the NMDA-evoked [(3)H] noradrenaline release from rat hippocampal slices. Neurochem Int 55(6):383–388. doi: https://doi.org/10.1016/j.neuint.2009.04.005
Mitani H, Shirayama Y, Yamada T, Maeda K, Ashby CR Jr, Kawahara R (2006) Correlation between plasma levels of glutamate, alanine, and serine with severity of depression. Prog Neuropsychopharmacol Biol Psychiatry 30:1155–1158. doi: https://doi.org/10.1016/j.pnpbp.2006.03.036
Montoliu C, Llansola M, Monfort P, Corbalan R, Fernandez-Marticorena I, Hernandez-Viadel ML, Felipo V (2001) Role of nitric oxide and cyclic GMP in glutamate-induced neuronal death. Neurotoxic Res 3(2):179–188. doi: https://doi.org/10.1007/BF03033190
Nakano T, Hasegawa T, Suzuki D, Motomura E, Okada M (2019) Amantadine Combines Astroglial System Xc- Activation with Glutamate/NMDA Receptor Inhibition. Biomolecules 9(5):191. doi: https://doi.org/10.3390/biom9050191
Okere CO, Waterhouse BD (2006) Activity-dependent heterogeneous populations of nitric oxide synthase neurons in the rat dorsal raphe nucleus (2006). Brain Res 1086(1):117–132. doi: https://doi.org/10.1016/j.brainres.2006.02.107
Padovan CM, Guimarães FS (2004) Antidepressant-like effects of NMDA-receptor antagonist injected into the dorsal hippocampus of rats. Pharmacol Biochem Behav 77(1):15–19. doi: https://doi.org/10.1016/j.pbb.2003.09.015
Paquette MA, Martinez AA, Macheda T, Meshul CK, Johnson SW, Berger SP, Giuffrida A (2012) Anti-dyskinetic mechanisms of amantadine and dextromethorphan in the 6-OHDA rat model of Parkinson’s disease: role of NMDA vs. 5-HT1A receptors. Eur J Neurosci 36(9):3224–3234. doi: https://doi.org/10.1111/j.1460-9568.2012.08243.x
Porsolt RD, Le Pichon M, Jalfre ML (1977) Depression: a new animal model sensitive to antidepressant treatments. Nature 266:730. doi: https://doi.org/10.1038/266730a0
Rameau GA, Tukey DS, Garcin-Hosfield ED, Titcombe RF, Misra C, Khatri L, Getzoff ED, Ziff EB (2007) Biphasic coupling of neuronal nitric oxide synthase phosphorylation to the NMDA receptor regulates AMPA receptor trafficking and neuronal cell death. J Neurosci 27(13):3445–3455. doi: https://doi.org/10.1523/JNEUROSCI.4799-06.2007
Roni MA, Rahman S (2015) Effects of lobeline and reboxetine, fluoxetine, or bupropion combination on depression-like behaviors in mice. Pharmacol Biochem Behav 139(Pt A) 1–6. doi: https://doi.org/10.1016/j.pbb.2015.10.006
Rosen RC, McKenna KE (2002) PDE-5 inhibition and sexual response: pharmacological mechanisms and clinical outcomes. Annu Rev Sex Res 13:36–88
Salvadore G, Zarate CA (2010) Jr Magnetic resonance spectroscopy studies of the glutamatergic system in mood disorders: a pathway to diagnosis, novel therapeutics, and personalized medicine? Biol Psychiatry 68(9):780–782. doi: https://doi.org/10.1016/j.biopsych.2010.09.011
Sanacora G, Smith MA, Pathak S, Su HL, Boeijinga PH, McCarthy DJ, Quirk MC (2014) Lanicemine: a low-trapping NMDA channel blocker produces sustained antidepressant efficacy with minimal psychotomimetic adverse effects. Mol Psychiatry 19(9):978–985. doi: https://doi.org/10.1038/mp.2013.130
Sanacora G, Treccani G, Popoli M (2012) Towards a glutamate hypothesis of depression: an emerging frontier of neuropsychopharmacology for mood disorders. Neuropharmacol 62(1):63–77. doi: https://doi.org/10.1016/j.neuropharm.2011.07.036
Schlumpf M, Lichtensteiger W, Langemann H, Waser PG, Hefti F (1974) A fluorometric micro method for the simultaneous determination of serotonin, noradrenaline, and dopamine in milligram amounts of brain tissue. Biochem Pharmacol 23:2437–2446. doi: https://doi.org/10.1016/0006-2952(74)90235-4
Serafini G, Pompili M, Innamorati M, Dwivedi Y, Brahmachari G, Girardi P (2013) Pharmacological properties of glutamatergic drugs targeting NMDA receptors and their application in major depression. Curr Pharm Des 19:1898–1922. doi: https://doi.org/10.2174/13816128113199990293
Shinde V, Yegnanarayan R, Shah P, Gupta A, Pophale P (2015) Antidepressant-like activity of flunarizine in modified tail suspension test in rats. N Am J Med Sci 7(3):100–103. doi: https://doi.org/10.4103/1947-2714.153921
Socała K, Nieoczym D, Wyska E, Poleszak E, Wlaź P (2012a) Influence of sildenafil on the antidepressant activity of bupropion and venlafaxine in the forced swim test in mice. Pharmacol Biochem Behav 103:273–278. doi: https://doi.org/10.1016/j.pbb.2012.08.012
Socała K, Nieoczym D, Wyska E, Poleszak E, Wlaź P (2012b) Sildenafil, a phosphodiesterase type 5 inhibitor, enhances the antidepressant activity of amitriptyline but not desipramine, in the forced swim test in mice. J Neural Transm 119:645–652. doi: https://doi.org/10.1007/s00702-011-0756-9
Socała K, Nieoczym D, Wyska E, Poleszak E, Wlaź P (2012c) Sildenafil, a phosphodiesterase type 5 inhibitor, reduces antidepressant-like activity of paroxetine in the forced swim test in mice. Pharmacol Rep 64:1259–1266. doi: https://doi.org/10.1016/s1734-1140(12)70922-2
Steru L, Chermat R, Thierry B, Simon P (1985) The tail suspension test: a new method for screening antidepressants in mice. Psychopharmacol 85:367–370. doi: https://doi.org/10.1007/BF00428203
Szasz BK (2007) Direct inhibitory effect of fluoxetine on N-methyl-D-aspartate receptors in the central nervous system. Biol Psychiatry 62:1303–1309. doi: https://doi.org/10.1016/j.biopsych.2007.04.014
Tagliaferro P (2001) Increased nitric oxide synthase activity in a model of serotonin depletion. Brain Res Bull 54:199–205. doi: https://doi.org/10.1016/s0361-9230(00)00450-0
Trivedi MH, Wisniewski SRRA, Nierenberg AA, Warden D, Ritz L, Norquist G, Howland RH, Lebowitz B, McGrath PJ, Shores-Wilson K, Biggs MM, Balasubramanian GK, Fava M (2006) STAR* D Study Team: Evaluation of outcomes with citalopram for depression using measurement-based care in STAR-D: implications for clinical practice. Am J Psychiatr 163:28–40. doi: https://doi.org/10.1176/appi.ajp.163.1.28
Volke V, Wegener G, Bourin M, Vasar E (2003) Antidepressant- and anxiolytic-like effects of selective neuronal NOS inhibitor 1-(2-trifluoromethylphenyl)-imidazole in mice. Behav Brain Res 140:141–147. doi: https://doi.org/10.1016/s0166-4328(02)00312-1
Walia V, Garg C, Garg M (2019) Lithium potentiated, pyridoxine abolished, and fluoxetine attenuated the anxiolytic effect of diazepam in mice. Brain Res Bull 150:343–353. doi: https://doi.org/10.1016/j.brainresbull.2019.06.008
Walia V, Garg C, Garg M (2020) Amantadine exerts anxiolytic like effect in mice: Evidence for the involvement of nitrergic and GABAergic signaling pathways. Behav Brain Res 380:112432. doi: https://doi.org/10.1016/j.bbr.2019.112432
Wesemann W, Dette-Wildenhahn G, Fellellner H (1979) In vitro studies on the possible effects of L-aminoadamantanes of the serotonergic system in M. Parkinson J Neural Transrn 44:263–285. doi: https://doi.org/10.1007/BF01250322
Whitton PS, Biggs CS, Pearce BR, Fowler LJ (1992a) MK-801 increases extracellular 5-hydroxytryptamine in rat hippocampus and striatum in vivo J. Neurochem 58:1573–1575. doi: https://doi.org/10.1111/j.1471-4159. 1992.tb11381.x
Whitton PS, Biggs CS, Pearce BR, Fowler LJ (1992b) Regional effects of MK-801 on dopamine and its metabolites studied by in vivo microdialysis. Neurosci Lett 142:5–8. doi: https://doi.org/10.1016/0304-3940(92)90607-9
Whitton PS, Richards DA, Biggs CS, Fowler LJ (1994) N-methyl-D-aspartate receptors modulate extracellular 5-hydroxytryptamine concentration in rat hippocampus and striatum in vivo. Neurosci Lett 169:215–218. doi: https://doi.org/10.1016/0304-3940(94)90395-6
Williams NR, Schatzberg AF (2016) NMDA antagonist treatment of depression. Curr Opin Neurobiol 36:112–117. doi: https://doi.org/10.1016/j.conb.2015.11.001
Zhang J, Huang XY, Ye ML, Luo CX, Wu HY, Hu Y, Zhou QG, Wu DL, Zhu LJ, Zhu DY (2010) Neuronal nitric oxide synthase alteration accounts for the role of 5-HT1A receptor in modulating anxiety-related behaviors. J Neurosci 30(7):2433–2441. doi: https://doi.org/10.1523/JNeurosci.5880-09.2010
Zhu CB, Hewlett WA, Feoktistov I, Biaggioni I, Blakely RD (2004) Adenosine receptor, protein kinase G, and p38 mitogen-activated protein kinase-dependent up-regulation of serotonin transporters involves both transporter trafficking and activation. Mol Pharmacol 65(6):1462–1474. doi: https://doi.org/10.1016/j.niox.2009.03.001
Zomkowski AD, Engel D, Cunha MP, Gabilan NH, Rodrigues AL (2012) The role of the NMDA receptors and L-arginine-nitric oxide-cyclic guanosine monophosphate pathway in the antidepressant-like effect of duloxetine in the forced swimming test. Pharmacol Biochem Behav 103(2):408–417. doi: https://doi.org/10.1016/j.pbb.2012.09.011
Zomkowski AD, Engel D, Gabilan NH, Rodrigues AL (2010) Involvement of NMDA receptors and L-arginine-nitric oxide-cyclic guanosine monophosphate pathway in the antidepressant-like effects of escitalopram in the forced swimming test. Eur Neuropsychopharmacol 20:793–801. doi: https://doi.org/10.1016/j.euroneuro.2010.07.011
Zorumski CF, Nagele P, Mennerick S, Conway CR (2015) Treatment-Resistant Major Depression: Rationale for NMDA Receptors as Targets and Nitrous Oxide as Therapy. Front Psychiatry 6:172. doi: https://doi.org/10.3389/fpsyt.2015.00172
Funding
No funding was obtained for this research work.
Author information
Authors and Affiliations
Contributions
Ms. Sushma Maratha: Animal experimentation and manuscript writing. Dr. Vijay Sharma: Proof Reading. Dr. Vaibhav Walia: Study Design, Statistical analysis and proof reading.
Corresponding author
Ethics declarations
Conflict of interest/Competing interests
None.
Ethics approval
The study was approved by the IAEC, SGT University, Gurugram.
Consent to participate
Not applicable for this study.
Consent for publication
All the authors agreed for the publication.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Maratha, S., Sharma, V. & Walia, V. Possible involvement of NO-cGMP signaling in the antidepressant like Effect of Amantadine in mice. Metab Brain Dis 37, 2067–2075 (2022). https://doi.org/10.1007/s11011-022-01006-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-022-01006-4