Abstract
Communication between astrocytes and neurons has a profound effect on the pathophysiology of Alzheimer’s disease (AD). Astrocytes regulate homeostasis and increase synaptic plasticity in physiological situations, however, they become activated during the progression of AD. Whether or not these reactions are supportive or detrimental for the central nervous system have not been understood yet. Considering epigenetic regulation of neuroinflammatory genes by chromatin readers, particularly bromodomain and extraterminal domain (BET) family, here we examined the effect of chronic co-inhibition of astrocytes metabolism (with fluorocitrate) and also BRD4 (with JQ1) on cognition deficit at early stages of AD. Forty adult male Wistar rats underwent stereotaxic cannulation for inducing AD by intrahippocampal injection of Aβ1-42 (4 μg/8 μl/rat). Then animals were divided into five groups of Saline+DMSO, Aβ + saline+DMSO, Aβ + JQ1, Aβ + FC (fluorocitrate), and Aβ + JQ1 + FC and received the related treatments. Two weeks later, spatial memory was recorded by Morris Water Maze (MWM), and the levels of phosphorylated cyclic-AMP response element binding protein (CREB), postsynaptic density 95 (PSD95), synaptophysin (SYP), and tumor necrosis factor-alpha (TNF-α) were measured in the hippocampus by western blotting and RT-qPCR. Administration of JQ1 significantly improved both acquisition and retrieval of spatial memory, which were evident by decreased escape latency and increased total time spent (TTS) in target quadrant, and significant rise in p-CREB, PSD95, and synaptophysin compared with Aβ + saline+DMSO group. In contrast, both groups receiving FC demonstrated memory decline, and reduction in p-CREB, PSD95 and synaptophysin in parallel with increase in TNF-α. Our data indicate that chronic inhibition of BRD4 significantly restores memory impaired by amyloid β partly via CREB signaling and upregulating synaptic proteins of PSD95 and synaptophysin. However, inhibition of astrocytes nullifies the memory-boosting effects of JQ1 and reduces CREB/PSD95/synaptophysin levels in hippocampus.
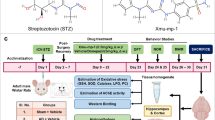
Graphical abstract








Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available within the supplementary materials.
Abbreviations
- Aβ:
-
Amyloid beta
- AD:
-
Alzheimer’s disease
- BRD4:
-
Bromodomain-containing protein 4
- Ca2+ :
-
Calcium ion
- CREB:
-
cAMP-response element binding protein
- FC:
-
Fluorocitrate
- MWM:
-
Morris water maze
- NF-κB:
-
Nuclear factor kappa B
- NMDAR :
-
N-methyl D-aspartate receptor
- p-CREB :
-
Phosphorylated cAMP-response element binding protein
- PSD95 :
-
Postsynaptic density 95
- P-TEFb :
-
Positive transcription elongation factor
- RT-qPCR:
-
Real-time quantitative polymerase chain reaction
- SYP:
-
Synaptophysin
- TNF-α:
-
Tumor necrosis factor alpha
- TTS:
-
Total time spent in target quadrant
References
Abe K, Saito H (1993) Tyrosine kinase inhibitors, herbimycin A and lavendustin A, block formation of long-term potentiation in the dentate gyrus in vivo. Brain Res 621:167–170. https://doi.org/10.1016/0006-8993(93)90315-E
Azam S, Jakaria M, Kim I-S, Kim J, Haque M, Choi D-K (2019) Regulation of toll-like receptor (TLR) signaling pathway by polyphenols in the treatment of age-linked neurodegenerative diseases: focus on TLR4 signaling. Front Immunol 10:1000. https://doi.org/10.3389/fimmu.2019.01000
Babaei P (2021) NMDA and AMPA receptors dysregulation in Alzheimer's disease. Eur J Pharmacol 908:174310. https://doi.org/10.1016/j.ejphar.2021.174310
Babaei P, Shirkouhi SG, Hosseini R, Soltani Tehrani B (2017) Vitamin D is associated with metabotropic but not neurotrophic effects of exercise in ovariectomized rats. Diabetol Metab Syndr 9:91. https://doi.org/10.1186/s13098-017-0288-z
Badrikoohi M, Esmaeili-bandboni A, Babaei P (2022) Simultaneous administration of bromodomain and histone deacetylase I inhibitors alleviates cognition deficit in Alzheimer's model of rats. Brain Res Bull 179:49–56. https://doi.org/10.1016/j.brainresbull.2021.12.004
Benito E, Barco A (2010) CREB's control of intrinsic and synaptic plasticity: implications for CREB-dependent memory models. Trends Neurosci 33:230–240. https://doi.org/10.1016/j.tins.2010.02.001
Benito E, Ramachandran B, Schroeder H, Schmidt G, Urbanke H, Burkhardt S, Capece V, Dean C, Fischer A (2017) The BET/BRD inhibitor JQ1 improves brain plasticity in WT and APP mice. Transl Psychiatry 7:e1239. https://doi.org/10.1038/tp.2017.202
Bouvier DS, Jones EV, Quesseveur G, Davoli MA, Ferreira TA, Quirion R, Mechawar N, Murai KK (2016) High resolution dissection of reactive glial nets in Alzheimer’s disease. Sci Rep 6:1–15. https://doi.org/10.1038/srep24544
Broadhead MJ, Horrocks MH, Zhu F, Muresan L, Benavides-Piccione R, DeFelipe J, Fricker D, Kopanitsa MV, Duncan RR, Klenerman D (2016) PSD95 nanoclusters are postsynaptic building blocks in hippocampus circuits. Sci Rep 6:1–14. https://doi.org/10.1038/srep24626
Byrne JH (2017) Learning and memory: a comprehensive reference. Academic Press, Cambridge
Chan CH, Fang C, Qiao Y, Yarilina A, Prinjha RK, Ivashkiv LB (2015) BET bromodomain inhibition suppresses transcriptional responses to cytokine-Jak-STAT signaling in a gene-specific manner in human monocytes. Eur J Immunol 45:287–297. https://doi.org/10.1002/eji.201444862
Chen A, Akinyemi RO, Hase Y, Firbank MJ, Ndung’u MN, Foster V, Craggs LJ, Washida K, Okamoto Y, Thomas AJ (2016) Frontal white matter hyperintensities, clasmatodendrosis and gliovascular abnormalities in ageing and post-stroke dementia. Brain 139:242–258. https://doi.org/10.1093/brain/awv328
Cragnolini AB, Lampitella G, Virtuoso A, Viscovo I, Panetsos F, Papa M, Cirillo G (2020) Regional brain susceptibility to neurodegeneration: what is the role of glial cells? Neural Regen Res 15:838. https://doi.org/10.4103/1673-5374.268897
Curran BP, O'Connor JJ (2003) The inhibition of long-term potentiation in the rat dentate gyrus by pro-inflammatory cytokines is attenuated in the presence of nicotine. Neurosci Lett 344:103–106. https://doi.org/10.1016/S0304-3940(03)00440-3
Daly C, Ziff EB (2002) Ca2+−dependent formation of a dynamin-synaptophysin complex: potential role in synaptic vesicle endocytosis. J Biol Chem 277:9010–9015. https://doi.org/10.1074/jbc.M110815200
De Strooper B, Karran E (2016) The cellular phase of Alzheimer’s disease. Cell 164:603–615. https://doi.org/10.1016/j.cell.2015.12.056
Dore K, Malinow R (2021) Elevated PSD-95 blocks ion-flux independent LTD: a potential new role for PSD-95 in synaptic plasticity. Neuroscience 456:43–49. https://doi.org/10.1016/j.neuroscience.2020.02.020
Evans G, Cousin M (2005) Tyrosine phosphorylation of synaptophysin in synaptic vesicle recycling. Biochem Soc Trans 33:1350–1353. https://doi.org/10.1042/BST0331350
Filippakopoulos P, Knapp S (2014) Targeting bromodomains: epigenetic readers of lysine acetylation. Nat Rev Drug Discov 13:337–356. https://doi.org/10.1038/nrd4286
Fonnum F, Johnsen A, Hassel B (1997) Use of fluorocitrate and fluoroacetate in the study of brain metabolism. Glia 21:106–113. https://doi.org/10.1002/(SICI)1098-1136(199709)21:1
Frost GR, Li Y-M (2017) The role of astrocytes in amyloid production and Alzheimer's disease. Open Biol 7:170228. https://doi.org/10.1098/rsob.170228
Fujita A, Yamaguchi H, Yamasaki R, Cui Y, Matsuoka Y, Yamada KI, Kira JI (2018) Connexin 30 deficiency attenuates A2 astrocyte responses and induces severe neurodegeneration in a 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine hydrochloride Parkinson's disease animal model. J Neuroinflammation 15:227. https://doi.org/10.1186/s12974-018-1251-0
Gibney E, Nolan C (2010) Epigenetics and gene expression. Heredity 105:4–13. https://doi.org/10.1038/hdy.2010.54
Glyvuk N, Tsytsyura Y, Geumann C, D’hooge R, Hüve J, Kratzke M, Baltes J, Boening D, Klingauf J, Schu P (2010) AP-1/σ1B-adaptin mediates endosomal synaptic vesicle recycling, learning and memory. EMBO J 29:1318–1330. https://doi.org/10.1038/emboj.2010.15
Gupta RC (2015) Handbook of toxicology of chemical warfare agents. Academic Press, Cambridge
Hirsch EC, Hunot S (2009) Neuroinflammation in Parkinson's disease: a target for neuroprotection? Lancet Neurol 8:382–397. https://doi.org/10.1016/S1474-4422(09)70062-6
Huang M, Zeng S, Zou Y, Shi M, Qiu Q, Xiao Y, Chen G, Yang X, Liang L, Xu H (2017) The suppression of bromodomain and extra-terminal domain inhibits vascular inflammation by blocking NF-kappaB and MAPK activation. Br J Pharmacol 174:101–115. https://doi.org/10.1111/bph.13657
Janz R, Südhof TC, Hammer RE, Unni V, Siegelbaum SA, Bolshakov VY (1999) Essential roles in synaptic plasticity for synaptogyrin I and synaptophysin I. Neuron 24:687–700. https://doi.org/10.1016/S0896-6273(00)81122-8
Jeong J, Pandey S, Li Y, Badger JD, Lu W, Roche KW (2019) PSD-95 binding dynamically regulates NLGN1 trafficking and function. Proc Natl Acad Sci 116:12035–12044. https://doi.org/10.1073/pnas.1821775116
Kapogiannis D, Mattson MP (2011) Disrupted energy metabolism and neuronal circuit dysfunction in cognitive impairment and Alzheimer's disease. Lancet Neurol 10:187–198. https://doi.org/10.1016/S1474-4422(10)70277-5
Korb E, Herre M, Zucker-Scharff I, Darnell RB, Allis CD (2015) BET protein Brd4 activates transcription in neurons and BET inhibitor Jq1 blocks memory in mice. Nat Neurosci 18:1464–1473. https://doi.org/10.1038/nn.4095
Kornau H-C, Schenker LT, Kennedy MB, Seeburg PH (1995) Domain interaction between NMDA receptor subunits and the postsynaptic density protein PSD-95. Science 269:1737–1740. https://doi.org/10.1126/science.7569905
Kwon SE, Chapman ER (2011) Synaptophysin regulates the kinetics of synaptic vesicle endocytosis in central neurons. Neuron 70:847–854. https://doi.org/10.1016/j.neuron.2011.04.001
Lambert JT, Hill TC, Park DK, Culp JH, Zito K (2017) Protracted and asynchronous accumulation of PSD95-family MAGUKs during maturation of nascent dendritic spines. Develop Neurobiol 77:1161–1174. https://doi.org/10.1002/dneu.22503
Liddelow SA, Barres BA (2017) Reactive astrocytes: production, function, and therapeutic potential. Immunity 46:957–967. https://doi.org/10.1016/j.immuni.2017.06.006
Liddelow SA, Guttenplan KA, Clarke LE, Bennett FC, Bohlen CJ, Schirmer L, Bennett ML, Munch AE, Chung WS, Peterson TC, Wilton DK, Frouin A, Napier BA, Panicker N, Kumar M, Buckwalter MS, Rowitch DH, Dawson VL, Dawson TM et al (2017) Neurotoxic reactive astrocytes are induced by activated microglia. Nature 541:481–487. https://doi.org/10.1038/nature21029
Lorenzo A, Yankner BA (1994) Beta-amyloid neurotoxicity requires fibril formation and is inhibited by Congo red. Proc Natl Acad Sci 91:12243–12247. https://doi.org/10.1073/pnas.91.25.12243
Magistri M, Velmeshev D, Makhmutova M, Patel P, Sartor CG, Volmar C-H, Wahlestedt C, Ali Faghihi M (2016) The BET-bromodomain inhibitor JQ1 reduces inflammation and tau phosphorylation at Ser396 in the brain of the 3xTg model of Alzheimer’s disease. Curr Alzheimer Res 13:985–995
Masliah E, Mallory M, Alford M, DeTeresa R, Hansen L, McKeel D, Morris J (2001) Altered expression of synaptic proteins occurs early during progression of Alzheimer’s disease. Neurology 56:127–129. https://doi.org/10.1212/WNL.56.1.127
McGeer EG, McGeer PL (2003) Inflammatory processes in Alzheimer's disease. Prog Neuro-Psychopharmacol Biol Psychiatry 27:741–749. https://doi.org/10.1016/j.jneuroim.2006.11.017
Munoz L, Ranaivo HR, Roy SM, Hu W, Craft JM, McNamara LK, Chico LW, Van Eldik LJ, Watterson DM (2007) A novel p38α MAPK inhibitor suppresses brain proinflammatory cytokine up-regulation and attenuates synaptic dysfunction and behavioral deficits in an Alzheimer's disease mouse model. J Neuroinflammation 4:1–14. https://doi.org/10.1186/1742-2094-4-21
Nicodeme E, Jeffrey KL, Schaefer U, Beinke S, Dewell S, Chung C-w, Chandwani R, Marazzi I, Wilson P, Coste H (2010) Suppression of inflammation by a synthetic histone mimic. Nature 468:1119–1123. https://doi.org/10.1038/nature09589
Nikseresht Z, Ahangar N, Badrikoohi M, Babaei P (2021) Synergistic enhancing-memory effect of D-serine and RU360, a mitochondrial calcium uniporter blocker in rat model of Alzheimer's disease. Behav Brain Res 409:113307. https://doi.org/10.1016/j.bbr.2021.113307
Oberheim NA, Takano T, Han X, He W, Lin JH, Wang F, Xu Q, Wyatt JD, Pilcher W, Ojemann JG (2009) Uniquely hominid features of adult human astrocytes. J Neurosci 29:3276–3287. https://doi.org/10.1523/JNEUROSCI.4707-08.2009
Olmos G, Lladó J (2014) Tumor necrosis factor alpha: a link between neuroinflammation and excitotoxicity. Mediat Inflamm 2014. https://doi.org/10.1155/2014/861231
Paulsen RE, Contestabile A, Villani L, Fonnum F (1987) An in vivo model for studying function of brain tissue temporarily devoid of glial cell metabolism: the use of fluorocitrate. J Neurochem 48:1377–1385. https://doi.org/10.1111/j.1471-4159.1987.tb05674.x
Paxinos G, Watson C (2006) The rat brain in stereotaxic coordinates: hard cover edition. Elsevier, Amsterdam
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT–PCR. Nucleic Acids Res 29:e45–e45. https://doi.org/10.1093/nar/29.9.e45
Pickering M, O’Connor JJ (2007) Pro-inflammatory cytokines and their effects in the dentate gyrus. Prog Brain Res 163:339–354. https://doi.org/10.1016/S0079-6123(07)63020-9
Rao JS, Kellom M, Kim H-W, Rapoport SI, Reese EA (2012) Neuroinflammation and synaptic loss. Neurochem Res 37:903–910. https://doi.org/10.1007/s11064-012-0708-2
Reiner DJ, Mietlicki-Baase EG, McGrath LE, Zimmer DJ, Bence KK, Sousa GL, Konanur VR, Krawczyk J, Burk DH, Kanoski SE (2016) Astrocytes regulate GLP-1 receptor-mediated effects on energy balance. J Neurosci 36:3531–3540. https://doi.org/10.1523/JNEUROSCI.3579-15.2016
Ren W-J, Liu Y, Zhou L-J, Li W, Zhong Y, Pang R-P, Xin W-J, Wei X-H, Wang J, Zhu H-Q (2011) Peripheral nerve injury leads to working memory deficits and dysfunction of the hippocampus by upregulation of TNF-α in rodents. Neuropsychopharmacology 36:979–992. https://doi.org/10.1038/npp.2010.236
Sartor GC, Malvezzi AM, Kumar A, Andrade NS, Wiedner HJ, Vilca SJ, Janczura KJ, Bagheri A, Al-Ali H, Powell SK (2019) Enhancement of BDNF expression and memory by HDAC inhibition requires BET bromodomain reader proteins. J Neurosci 39:612–626. https://doi.org/10.1523/JNEUROSCI.1604-18.2018
Schmitt U, Tanimoto N, Seeliger M, Schaeffel F, Leube R (2009) Detection of behavioral alterations and learning deficits in mice lacking synaptophysin. Neuroscience 162:234–243. https://doi.org/10.1016/j.neuroscience.2009.04.046
Shang X-L, Wang Q-B, Liu X-P, Yao X-Q, Cao F-Y, Wang Q, Zhang J-Y, Wang J-Z, Liu G-P (2015) Fluorocitrate induced the alterations of memory-related proteins and tau hyperphosphorylation in SD rats. Neurosci Lett 584:230–235. https://doi.org/10.1016/j.neulet.2014.10.036
Shao CY, Mirra SS, Sait HB, Sacktor TC, Sigurdsson EM (2011) Postsynaptic degeneration as revealed by PSD-95 reduction occurs after advanced Aβ and tau pathology in transgenic mouse models of Alzheimer’s disease. Acta Neuropathol 122:285–292. https://doi.org/10.1007/s00401-011-0843-x
Sharifzadeh M, Esmaeili-Bandboni A, Emami MR, Naeini F, Zarezadeh M, Javanbakht MH (2022) The effects of all trans retinoic acid, vitamin D3 and their combination on plasma levels of miRNA-125a-5p, miRNA-34a, and miRNA-126 in an experimental model of diabetes. Avicenna J Phytomed 12. https://doi.org/10.22038/ajp.2021.18598
Shi J, Vakoc CR (2014) The mechanisms behind the therapeutic activity of BET bromodomain inhibition. Mol Cell 54:728–736. https://doi.org/10.1016/j.molcel.2014.05.016
Shioda S, Funahashi H, Nakajo S, Yada T, Maruta O, Nakai Y (1998) Immunohistochemical localization of leptin receptor in the rat brain. Neurosci Lett 243:41–44. https://doi.org/10.1016/S0304-3940(98)00082-2
Smit T, Deshayes NA, Borchelt DR, Kamphuis W, Middeldorp J, Hol EM (2021) Reactive astrocytes as treatment targets in Alzheimer's disease—systematic review of studies using the APPswePS1dE9 mouse model. Glia 69:1852–1881. https://doi.org/10.1002/glia.23981
Sobrinho CR, Gonçalves CM, Takakura AC, Mulkey DK, Moreira TS (2017) Fluorocitrate-mediated depolarization of astrocytes in the retrotrapezoid nucleus stimulates breathing. J Neurophysiol 118:1690–1697. https://doi.org/10.1152/jn.00032.2017
Swanson RA, Graham SH (1994) Fluorocitrate and fluoroacetate effects on astrocyte metabolism in vitro. Brain Res 664:94–100. https://doi.org/10.1016/0006-8993(94)91958-5
Swerdlow RH (2018) Mitochondria and mitochondrial cascades in Alzheimer’s disease. J Alzheimers Dis 62:1403–1416. https://doi.org/10.3233/JAD-170585
Tang BL (2009) Neuronal protein trafficking associated with Alzheimer disease: from APP and BACE1 to glutamate receptors. Cell Adhes Migr 3:118–128. https://doi.org/10.4161/cam.3.1.7254
Tichauer J, Saud K, Von Bernhardi R (2007) Modulation by astrocytes of microglial cell-mediated neuroinflammation: effect on the activation of microglial signaling pathways. Neuroimmunomodulation 14:168–174. https://doi.org/10.1159/000110642
Valtorta F, Pennuto M, Bonanomi D, Benfenati F (2004) Synaptophysin: leading actor or walk-on role in synaptic vesicle exocytosis? Bioessays 26:445–453. https://doi.org/10.1002/bies.20012
Wall AM, Mukandala G, Greig NH, O'Connor JJ (2015) Tumor necrosis factor-α potentiates long-term potentiation in the rat dentate gyrus after acute hypoxia. J Neurosci Res 93:815–829. https://doi.org/10.1002/jnr.23540
Wang L, Li C-C, Wang G-W, Cai J-X (2009) The effects of centrally administered fluorocitrate via inhibiting glial cells on working memory in rats. Sci China Ser C Life Sci 52:701–709. https://doi.org/10.1007/s11427-009-0101-9
Wang H, Huang W, Liang M, Shi Y, Zhang C, Li Q, Liu M, Shou Y, Yin H, Zhu X (2018) (+)-JQ1 attenuated LPS-induced microglial inflammation via MAPK/NFκB signaling. Cell Biosci 8:1–15. https://doi.org/10.1186/s13578-018-0258-7
Wang W, Zhao F, Ma X, Perry G, Zhu X (2020) Mitochondria dysfunction in the pathogenesis of Alzheimer’s disease: recent advances. Mol Neurodegener 15:1–22. https://doi.org/10.1186/s13024-020-00376-6
Wu Z, Guo Z, Gearing M, Chen G (2014) Tonic inhibition in dentate gyrus impairs long-term potentiation and memory in an Alzheimer’s disease model. Nat Commun 5:1–13. https://doi.org/10.1038/ncomms5159
Xiao Y, Liang L, Huang M, Qiu Q, Zeng S, Shi M, Zou Y, Ye Y, Yang X, Xu H (2016) Bromodomain and extra-terminal domain bromodomain inhibition prevents synovial inflammation via blocking IkappaB kinase-dependent NF-kappaB activation in rheumatoid fibroblast-like synoviocytes. Rheumatology (Oxford) 55:173–184. https://doi.org/10.1093/rheumatology/kev312
Xu W, Schlüter OM, Steiner P, Czervionke BL, Sabatini B, Malenka RC (2008) Molecular dissociation of the role of PSD-95 in regulating synaptic strength and LTD. Neuron 57:248–262. https://doi.org/10.1016/j.neuron.2007.11.027
Zhang Y, Sloan SA, Clarke LE, Caneda C, Plaza CA, Blumenthal PD, Vogel H, Steinberg GK, Edwards MS, Li G (2016) Purification and characterization of progenitor and mature human astrocytes reveals transcriptional and functional differences with mouse. Neuron 89:37–53. https://doi.org/10.1016/j.neuron.2015.11.013
Zhang X, Shen X, Dong J, Liu W-C, Song M, Sun Y, Shu H, Towse C-L, Liu W, Liu C-F (2019) Inhibition of reactive astrocytes with fluorocitrate ameliorates learning and memory impairment through upregulating CRTC1 and synaptophysin in ischemic stroke rats. Cell Mol Neurobiol 39:1151–1163. https://doi.org/10.1007/s10571-019-00709-0
Zhou B, Mu J, Gong Y, Lu C, Zhao Y, He T, Qin Z (2017) Brd4 inhibition attenuates unilateral ureteral obstruction-induced fibrosis by blocking TGF-beta-mediated Nox4 expression. Redox Biol 11:390–402. https://doi.org/10.1016/j.redox.2016.12.031
Acknowledgments
The authors thank Dr. Nozhat Zebardast for her technical support and Hamid Morrovati for his cooperation in animal handling during surgery.
Funding
This study was supported by Grants from Research Council of Guilan University of Medical Sciences (Grant No. GUMS 98032706).
Author information
Authors and Affiliations
Contributions
PB designed and supervised the experiment, prepared first draft and revised manuscript. AE performed and analyzed the real-time and western blotting, also actively cooperated in manuscript revising. RN and MB performed the behavioral experiments and analyzed data. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Authors declare no conflict of interest
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nikkar, R., Esmaeili-bandboni, A., Badrikoohi, M. et al. Effects of inhibiting astrocytes and BET/BRD4 chromatin reader on spatial memory and synaptic proteins in rats with Alzheimer’s disease. Metab Brain Dis 37, 1119–1131 (2022). https://doi.org/10.1007/s11011-022-00940-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-022-00940-7