Abstract
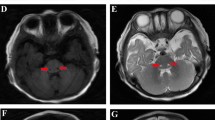
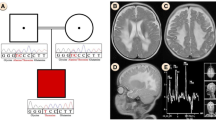
Multiple mitochondrial dysfunction syndrome (MMDS) refers to a class of mitochondrial diseases caused by nuclear gene mutations, which usually begins in early infancy and is classically characterized by markedly impaired neurological development, generalized muscle weakness, lactic acidosis, and hyperglycinemia, cavitating leukoencephalopathy, respiratory failure, as well as early fatality resulted from dysfunction of energy metabolism in multiple systems. So far, six types of MMDS have been identified based on different genotypes, which are caused by mutations in NFU1, BOLA3, IBA57, ISCA2, ISCA1 and PMPCB, respectively. IBA57 encodes a protein involved in the mitochondrial Fe/S cluster assembly process, which plays a vital role in the activity of multiple mitochondrial enzymes. Herein, detailed clinical investigation of 2 Chinese patients from two unrelated families were described, both of them showed mildly delay in developmental milestone before disease onset, the initial symptoms were all presented with acute motor and mental retrogression, and brain MRI showed diffused leukoencephalopathy with cavities, dysplasia of corpus callosum and cerebral atrophy. Exome sequencing revealed three IBA57 variants, one shared variant (c.286T>C) has been previously reported, the remaining two (c.189delC and c.580 A>G) are novel. To enhance the understanding of this rare disease, we further made a literature review about the current progress in clinical, genetic and treatment of the disorder. Due to the rapid progress of MMDS, early awareness is crucial to prompt and proper administration, as well as genetic counseling.


Similar content being viewed by others
Data availability
The raw data are available upon reasonable request.
Code availability
Not applicable.
References
Ajit Bolar N, Vanlander A, Wilbrecht C, Van der Aa N, Smet J, De Paepe B et al (2013) Mutation of the iron-sulfur cluster assembly gene IBA57 causes severe myopathy and encephalopathy. Hum Mol Genet 22:2590–2602
Alfadhel M (2019) Multiple mitochondrial dysfunctions syndrome 4 due to ISCA2 gene defects: a review. Child Neurol Open 6:2329048X19847377
Alfadhel M, Nashabat M, Abu Ali Q, Hundallah K (2017) Mitochondrial iron-sulfur cluster biogenesis from molecular understanding to clinical disease. Neurosciences (Riyadh) 22:4–13
Al-Hassnan Z, Al-Dosary M, Alfadhel M, Faqeih E, Alsagob M, Kenana R et al (2015) ISCA2 mutation causes infantile neurodegenerative mitochondrial disorder. J Med Genet 52:186–194
Ashrafi M, Amanat M, Garshasbi M, Kameli R, Nilipour Y, Heidari M et al (2020) An update on clinical, pathological, diagnostic, and therapeutic perspectives of childhood leukodystrophies. Expert Rev Neurother 20:65–84
Cameron J, Janer A, Levandovskiy V, Mackay N, Rouault T, Tong W et al (2011) Mutations in iron-sulfur cluster scaffold genes NFU1 and BOLA3 cause a fatal deficiency of multiple respiratory chain and 2-oxoacid dehydrogenase enzymes. Am J Hum Genet 89:486–495
Debray F, Stümpfig C, Vanlander A, Dideberg V, Josse C, Caberg J et al (2015) Mutation of the iron-sulfur cluster assembly gene IBA57 causes fatal infantile leukodystrophy. J Inherit Metab Dis 38:1147–1153
Gourdoupis S, Nasta V, Calderone V, Ciofi-Baffoni S, Banci L (2018) IBA57 recruits ISCA2 to Form a [2Fe-2S] cluster-mediated complex. J Am Chem Soc 140:14401–14412
Hamanaka K, Miyatake S, Zerem A, Lev D, Blumkin L, Yokochi K et al (2018) Expanding the phenotype of IBA57 mutations: related leukodystrophy can remain asymptomatic. J Hum Genet 63:1223–1229
Ishiyama A, Sakai C, Matsushima Y, Noguchi S, Mitsuhashi S, Endo Y et al (2017) IBA57 mutations abrogate iron-sulfur cluster assembly leading to cavitating leukoencephalopathy. Neurol Genet 3:e184
Lebigot E, Gaignard P, Dorboz I, Slama A, Rio M, de Lonlay P et al (2017) Impact of mutations within the [Fe-S] cluster or the lipoic acid biosynthesis pathways on mitochondrial protein expression profiles in fibroblasts from patients. Mol Genet Metab 122:85–94
Lerman-Sagie T, Leshinsky-Silver E, Watemberg N, Luckman Y, Lev D (2005) White matter involvement in mitochondrial diseases. Mol Genet Metab 84:127–136
Lill R (2009) Function and biogenesis of iron-sulphur proteins. Nature 460:831–838
Lill R, Freibert S (2020) Mechanisms of mitochondrial iron-sulfur protein biogenesis. Annu Rev Biochem 89:471–499
Liu M, Zhang J, Zhang Z, Zhou L, Jiang Y, Wang J et al (2018) Phenotypic spectrum of mutations in IBA57, a candidate gene for cavitating leukoencephalopathy. Clin Genet 93:235–241
Long J, Best S, Hatem S, Theodorou T, Catton T, Murray S et al (2021) The long and winding road: perspectives of people and parents of children with mitochondrial conditions negotiating management after diagnosis. Orphanet J Rare Dis 16:310
Lossos A, Stümpfig C, Stevanin G, Gaussen M, Zimmerman B, Mundwiller E et al (2015) Fe/S protein assembly gene IBA57 mutation causes hereditary spastic paraplegia. Neurology 84:659–667
Maio N, Rouault T (2020) Outlining the complex pathway of mammalian Fe-S Cluster biogenesis. Trends Biochem Sci 45:411–426
Naidu S, Bibat G, Lin D, Burger P, Barker P, Rosemberg S et al (2005) Progressive cavitating leukoencephalopathy: a novel childhood disease. Ann Neurol 58:929–938
Nasta V, Da Vela S, Gourdoupis S, Ciofi-Baffoni S, Svergun D, Banci L (2019) structural properties of [2Fe-2S] ISCA2-IBA57: a complex of the mitochondrial iron-sulfur cluster assembly machinery. Sci Rep 9:18986
Nizon M, Boutron A, Boddaert N, Slama A, Delpech H, Sardet C et al (2014) Leukoencephalopathy with cysts and hyperglycinemia may result from NFU1 deficiency. Mitochondrion 15:59–64
Parikh S, Goldstein A, Karaa A, Koenig M, Anselm I, Brunel-Guitton C et al (2017) Patient care standards for primary mitochondrial disease: a consensus statement from the Mitochondrial Medicine Society. Genet Med 19. https://doi.org/10.1038/gim.2017.1107
Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J et al (2015) Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med 17:405–424
Saudino G, Suraci D, Nasta V, Ciofi-Baffoni S, Banci L (2021) Molecular basis of multiple mitochondrial dysfunctions syndrome 2 caused by CYS59TYR BOLA3 mutation. Int J Mol Sci 22:4848
Shukla A, Hebbar M, Srivastava A, Kadavigere R, Upadhyai P, Kanthi A et al (2017) Homozygous p.(Glu87Lys) variant in ISCA1 is associated with a multiple mitochondrial dysfunctions syndrome. J Hum Genet 62:723–727
Stehling O, Lill R (2013) The role of mitochondria in cellular iron-sulfur protein biogenesis: mechanisms, connected processes, and diseases. Cold Spring Harb Perspect Biol 5:a011312
Suraci D, Saudino G, Nasta V, Ciofi-Baffoni S, Banci L (2021) ISCA1 orchestrates ISCA2 and NFU1 in the maturation of human mitochondrial [4Fe-4S] proteins. J Mol Biol 433:166924
Tarnopolsky M (2008) The mitochondrial cocktail: rationale for combined nutraceutical therapy in mitochondrial cytopathies. Adv Drug Deliv Rev 60:1561–1567
Toldo I, Nosadini M, Boscardin C, Talenti G, Manara R, Lamantea E et al (2018) Neonatal mitochondrial leukoencephalopathy with brain and spinal involvement and high lactate: expanding the phenotype of ISCA2 gene mutations. Metab Brain Dis 33:805–812
Torraco A, Ardissone A, Invernizzi F, Rizza T, Fiermonte G, Niceta M et al (2017) Novel mutations in IBA57 are associated with leukodystrophy and variable clinical phenotypes. J Neurol 264:102–111
Uziel G, Ghezzi D, Zeviani M (2011) Infantile mitochondrial encephalopathy. Semin Fetal Neonatal Med 16:205–215
Uzunhan T, Çakar N, Seyhan S, Aydin K (2020) A genetic mimic of cerebral palsy: Homozygous NFU1 mutation with marked intrafamilial phenotypic variation. Brain Dev 42:756–761
Vögtle F, Brändl B, Larson A, Pendziwiat M, Friederich M, White S et al (2018) Mutations in PMPCB encoding the catalytic subunit of the mitochondrial Presequence protease cause neurodegeneration in early childhood. Am J Hum Genet 102:557–573
Wachnowsky C, Fidai I, Cowan J (2018) Iron-sulfur cluster biosynthesis and trafficking - impact on human disease conditions. Metallomics 10:9–29
Weiler B, Brück M, Kothe I, Bill E, Lill R, Mühlenhoff U (2020) Mitochondrial [4Fe-4S] protein assembly involves reductive [2Fe-2S] cluster fusion on ISCA1-ISCA2 by electron flow from ferredoxin FDX2. Proc Natl Acad Sci U S A 117:20555–20565
Acknowledgements
We would like to thank the guardians of the patients for the support and cooperation.
Author information
Authors and Affiliations
Contributions
Feixia Zhan: Data acquisition, analysis and interpretation of data, and drafted the manuscript for intellectual content. This work was supported by the grants from the National Natural Science Foundation of China (No.81,870,889 and 82,071,258) and Shanghai Municipal Commission of Health and Family Planning (20184Y0056).
Xiaoli Liu: Funding, data acquisition, interpreted the data, and revised the manuscript for intellectual content.
Ruilong Ni: Data acquisition.
Taotao Liu: Data acquisition.
Yuwen Cao: Data acquisition.
Jingying Wu: Data acquisition.
Wotu Tian: Data acquisition.
Xinghua Luan: Data acquisition, analysis and interpretation of data, and revised the manuscript for intellectual content.
Li Cao: Funding, study design and conceptualization, analysis and interpretation of data, manuscript revision, and supervision.
Corresponding authors
Ethics declarations
Ethics approval
The study was approved by the ethics committee of Shanghai Jiao Tong University Affiliated Sixth People’s Hospital, Shanghai, China. Written informed consent, which also included the consent for the publication of medical information, was obtained from the guardians.
Conflict of interest
All authors in this study have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhan, F., Liu, X., Ni, R. et al. Novel IBA57 mutations in two chinese patients and literature review of multiple mitochondrial dysfunction syndrome. Metab Brain Dis 37, 311–317 (2022). https://doi.org/10.1007/s11011-021-00856-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-021-00856-8