Abstract
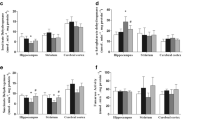
Tyrosinemia type II is a rare autosomal recessive disease caused by deficiency of hepatic tyrosine aminotransferase and is associated with neurologic and development difficulties in numerous patients. Considering that the mechanisms underlying the neurological dysfunction in hypertyrosinemic patients are poorly known and that high concentrations of tyrosine provoke mitochondrial dysfunction and oxidative stress, in the present study we investigated the in vivo influence of antioxidants (N-acetylcysteine, NAC; and deferoxamine, DFX) administration on the inhibitory effects on parameters of energy metabolism in cerebral cortex, hippocampus and striatum of rats, provoked by chronic administration of L.-tyrosine. Our results showed that chronic administration of L.-tyrosine results in a marked decrease in the activity of citrate synthase in all the analyzed structures and succinate dehydrogenase activities in hippocampus and striatum, and that antioxidants administration can prevent this inhibition in hippocampus and striatum. Moreover, chronic administration of L.-tyrosine inhibited the activity of complex I, II-III and IV in the striatum, which can be prevented by antioxidant treatment. However, the co-administration of NAC plus DFX could not prevent the inhibition of creatine kinase activity in the striatum. In conclusion, the present study demonstrates that the administration of antioxidants NAC and DFX attenuates the L.-tyrosine effects on enzymes of the Krebs cycle and the mitochondrial respiratory chain, suggesting that impairment of energy metabolism can be involved with oxidative stress. These results also indicate a possible neuroprotective role for NAC and DFX as a potential adjuvant therapy to the patients with Tyrosinemia type II.



Similar content being viewed by others
References
Aguirre P, Urrutia P, Tapia V, Villa M, Paris I, Segura-Aguilar J, Nunez MT (2012) The dopamine metabolite aminochrome inhibits mitochondrial complex I and modifies the expression of iron transporters DMT1 and FPN1. Biometals : an international journal on the role of metal ions in biology, biochemistry, and medicine 25:795–803. doi:10.1007/s10534-012-9525-y
Arent CO et al (2012) Synergist effects of n-acetylcysteine and deferoxamine treatment on behavioral and oxidative parameters induced by chronic mild stress in rats. Neurochem Int 61:1072–1080. doi:10.1016/j.neuint.2012.07.024
Asanuma M, Miyazaki I, Ogawa N (2003) Dopamine- or L-DOPA-induced neurotoxicity: the role of dopamine quinone formation and tyrosinase in a model of Parkinson's disease. Neurotox Res 5:165–176
Balansky R, Izzotti A, Scatolini L, D'Agostini F, De Flora S (1996) Induction by carcinogens and chemoprevention by N-acetylcysteine of adducts to mitochondrial DNA in rat organs. Cancer Res 56:1642–1647
Barbosa PR et al (2010) Inhibition of mitochondrial respiratory chain in the brain of rats after renal ischemia is prevented by N-acetylcysteine and deferoxamine. Metab Brain Dis 25:219–225. doi:10.1007/s11011-010-9187-9
Bavarsad Shahripour R, Harrigan MR, Alexandrov AV (2014) N-acetylcysteine (NAC) in neurological disorders: mechanisms of action and therapeutic opportunities. Brain and behavior 4:108–122. doi:10.1002/brb3.208
Bongiovanni R, Yamamoto BK, Simpson C, Jaskiw GE (2003) Pharmacokinetics of systemically administered tyrosine: a comparison of serum, brain tissue and in vivo microdialysate levels in the rat. J Neurochem 87:310–317
Cassina A, Radi R (1996) Differential inhibitory action of nitric oxide and peroxynitrite on mitochondrial electron transport. Arch Biochem Biophys 328:309–316. doi:10.1006/abbi.1996.0178
Cassol OJ Jr et al (2010) Effects of N-acetylcysteine/deferoxamine, taurine and RC-3095 on respiratory chain complexes and creatine kinase activities in rat brain after sepsis. Neurochem Res 35:515–521. doi:10.1007/s11064-009-0089-3
Cetinkaya A, Bulbuloglu E, Kurutas EB, Ciralik H, Kantarceken B, Buyukbese MA (2005) Beneficial effects of N-acetylcysteine on acetic acid-induced colitis in rats. Tohoku J Exp Med 206:131–139
Chen L et al (2008) Unregulated cytosolic dopamine causes neurodegeneration associated with oxidative stress in mice. The Journal of neuroscience : the official journal of the Society for Neuroscience 28:425–433. doi:10.1523/JNEUROSCI.3602-07.2008
Cocco T, Sgobbo P, Clemente M, Lopriore B, Grattagliano I, Di Paola M (2005) Villani G. Tissue-specific changes of mitochondrial functions in aged rats: effect of a long-term dietary treatment with N-acetylcysteine Free radical biology & medicine 38:796–805. doi:10.1016/j.freeradbiomed.2004.11.034
Cremona T, Kearney EB (1965) Studies on the respiratory chain-linked reduced nicotinamide adenine dinucleotide dehydrogenase. X. Reactions with sulfhydryl reagents. J Biol Chem 240:3645–3652
Cuzzocrea S, Mazzon E, Costantino G, Serraino I, De Sarro A, Caputi AP (2000) Effects of n-acetylcysteine in a rat model of ischemia and reperfusion injury. Cardiovasc Res 47:537–548
Damiani CR et al (2007) Oxidative stress and metabolism in animal model of colitis induced by dextran sulfate sodium. J Gastroenterol Hepatol 22:1846–1851. doi:10.1111/j.1440-1746.2007.04890.x
de Andrade RB, Gemelli T, Rojas DB, Funchal C, Dutra-Filho CS, Wannmacher CM (2011) Tyrosine inhibits creatine kinase activity in cerebral cortex of young rats. Metab Brain Dis 26:221–227. doi:10.1007/s11011-011-9255-9
de Andrade RB, Gemelli T, Rojas DB, Funchal C, Dutra-Filho CS, Wannmacher CM (2012) Tyrosine impairs enzymes of energy metabolism in cerebral cortex of rats. Mol Cell Biochem 364:253–261. doi:10.1007/s11010-012-1225-y
de Andrade RB et al (2015) Creatine and pyruvate prevent the alterations caused by tyrosine on parameters of oxidative stress and enzyme activities of phosphoryltransfer network in cerebral cortex of Wistar rats. Mol Neurobiol 51:1184–1194. doi:10.1007/s12035-014-8791-9
De Flora S, Izzotti A, D'Agostini F, Balansky RM (2001) Mechanisms of N-acetylcysteine in the prevention of DNA damage and cancer, with special reference to smoking-related end-points. Carcinogenesis 22:999–1013
De Pra SD et al (2014) L-tyrosine induces DNA damage in brain and blood of rats. Neurochem Res 39:202–207. doi:10.1007/s11064-013-1207-9
De Vries N, De Flora S (1993) N-acetyl-l-cysteine. J Cell Biochem 53(Supplement 17F):270–277
Di-Pietro PB et al (2008) Inhibition of brain creatine kinase activity after renal ischemia is attenuated by N-acetylcysteine and deferoxamine administration. Neurosci Lett 434:139–143. doi:10.1016/j.neulet.2008.01.051
During MJ, Acworth IN, Wurtman RJ (1989) Dopamine release in rat striatum: physiological coupling to tyrosine supply. J Neurochem 52:1449–1454
Ferrari G, Yan CY, Greene LA (1995) N-acetylcysteine (D- and L-stereoisomers) prevents apoptotic death of neuronal cells. The Journal of neuroscience : the official journal of the Society for Neuroscience 15:2857–2866
Ferreira GK et al (2012) L-tyrosine administration increases acetylcholinesterase activity in rats. Neurochem Int 61:1370–1374. doi:10.1016/j.neuint.2012.09.017
Ferreira GK et al (2013a) Effect of acute and chronic administration of L-tyrosine on nerve growth factor levels in rat brain. Neurochem Res 38:1742–1746. doi:10.1007/s11064-013-1078-0
Ferreira GK et al (2013b) Effect of L-tyrosine in vitro and in vivo on energy metabolism parameters in brain and liver of young rats. Neurotox Res 23:327–335. doi:10.1007/s12640-012-9345-4
Ferreira GK et al (2014) An evaluation of the effects of acute and chronic L-tyrosine administration on BDNF levels and BDNF mRNA expression in the rat brain. Mol Neurobiol 49:734–740. doi:10.1007/s12035-013-8552-1
Ferreira GK et al (2015) The characterization of neuroenergetic effects of chronic L-tyrosine administration in young rats: evidence for striatal susceptibility. Metab Brain Dis 30:215–221. doi:10.1007/s11011-014-9615-3
Fischer JC et al (1985) Differential investigation of the capacity of succinate oxidation in human skeletal muscle. Clinica chimica acta; international journal of clinical chemistry 153:23–36
Fries GR, Kapczinski F (2011) N-acetylcysteine as a mitochondrial enhancer: a new class of psychoactive drugs? Rev Bras Psiquiatr 33:321–322
Gluck M, Ehrhart J, Jayatilleke E, Zeevalk GD (2002) Inhibition of brain mitochondrial respiration by dopamine: involvement of H(2)O(2) and hydroxyl radicals but not glutathione-protein-mixed disulfides. J Neurochem 82:66–74
Goldsmith LA, Kang E, Bienfang DC, Jimbow K, Gerald P, Baden HP (1973) Tyrosinemia with plantar and palmar keratosis and keratitis. J Pediatr 83:798–805
Halliwell B (2011) Free radicals and antioxidants - quo vadis? Trends Pharmacol Sci 32:125–130. doi:10.1016/j.tips.2010.12.002
Halliwell B, Lee CY (2010) Using isoprostanes as biomarkers of oxidative stress: some rarely considered issues. Antioxid Redox Signal 13:145–156. doi:10.1089/ars.2009.2934
Hart AM, Terenghi G, Kellerth JO, Wiberg M (2004) Sensory neuroprotection, mitochondrial preservation, and therapeutic potential of N-acetyl-cysteine after nerve injury. Neuroscience 125:91–101. doi:10.1016/j.neuroscience.2003.12.040
Held PK (2006) Disorders of tyrosine catabolism. Mol Genet Metab 88:103–106
Hughes BP (1962) A method for the estimation of serum creatine kinase and its use in comparing creatine kinase and aldolase activity in normal and pathological sera. Clinica chimica acta; international journal of clinical chemistry 7:597–603
Khan M et al (2004) Administration of N-acetylcysteine after focal cerebral ischemia protects brain and reduces inflammation in a rat model of experimental stroke. J Neurosci Res 76:519–527. doi:10.1002/jnr.20087
Khan FH, Sen T, Maiti AK, Jana S, Chatterjee U, Chakrabarti S (2005) Inhibition of rat brain mitochondrial electron transport chain activity by dopamine oxidation products during extended in vitro incubation: implications for Parkinson's disease. Biochim Biophys Acta 1741:65–74. doi:10.1016/j.bbadis.2005.03.013
LaVoie MJ, Hastings TG (1999) Dopamine quinone formation and protein modification associated with the striatal neurotoxicity of methamphetamine: evidence against a role for extracellular dopamine. The Journal of neuroscience : the official journal of the Society for Neuroscience 19:1484–1491
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Macedo LG et al (2013) Effect of acute administration of L-tyrosine on oxidative stress parameters in brain of young rats. Neurochem Res 38:2625–2630. doi:10.1007/s11064-013-1180-3
Macsai MS, Schwartz TL, Hinkle D, Hummel MB, Mulhern MG, Rootman D (2001) Tyrosinemia type II: nine cases of ocular signs and symptoms. Am J Ophthalmol 132:522–527
Martinez Banaclocha M (2000) N-acetylcysteine elicited increase in complex I activity in synaptic mitochondria from aged mice: implications for treatment of Parkinson's disease. Brain Res 859:173–175
Martinez Banaclocha M, Martinez N (1999) N-acetylcysteine elicited increase in cytochrome c oxidase activity in mice synaptic mitochondria. Brain Res 842:249–251
Martinez M, Martinez N, Hernandez AI, Ferrandiz ML (1999) Hypothesis: can N-acetylcysteine be beneficial in Parkinson's disease? Life Sci 64:1253–1257
Mayer M, Noble M (1994) N-acetyl-L-cysteine is a pluripotent protector against cell death and enhancer of trophic factor-mediated cell survival in vitro. Proc Natl Acad Sci U S A 91:7496–7500
Mitchell GA, Grompe M, Lambert M, Tanguay RM (2001) Hypertyrosinemia. In: Scriver CR, Beaudet AL, Sly WS, Valle D (eds) The metabolic and molecular bases of inherited disease, vol 8. Mc Graw-Hill, New York, pp. 1977–1982
Morre MC, Hefti F, Wurtman RJ (1980) Regional tyrosine levels in rat brain after tyrosine administration. J Neural Transm 49:45–50
Munoz AM, Rey P, Soto-Otero R, Guerra MJ, Labandeira-Garcia JL (2004) Systemic administration of N-acetylcysteine protects dopaminergic neurons against 6-hydroxydopamine-induced degeneration. J Neurosci Res 76:551–562. doi:10.1002/jnr.20107
Pan J et al. (2009) Blockade of the translocation and activation of c-Jun N-terminal kinase 3 (JNK3) attenuates dopaminergic neuronal damage in mouse model of Parkinson's disease Neurochemistry international 54:418-425 doi:10.1016/j.neuint.2009.01.013
Pinho RA, Silveira PC, Silva LA, Luiz Streck E, Dal-Pizzol F, Moreira JCF (2005) N-acetylcysteine and deferoxamine reduce pulmonary oxidative stress and inflammation in rats after coal dust exposure. Environ Res 99:355–360. doi:10.1016/j.envres.2005.03.005
Ramos AC et al (2013) Acute administration of l-tyrosine alters energetic metabolism of hippocampus and striatum of infant rats. International journal of developmental neuroscience : the official journal of the International Society for Developmental Neuroscience 31:303–307. doi:10.1016/j.ijdevneu.2013.03.005
Rees JN, Florang VR, Anderson DG, Doorn JA (2007) Lipid peroxidation products inhibit dopamine catabolism yielding aberrant levels of a reactive intermediate. Chem Res Toxicol 20:1536–1542. doi:10.1021/tx700248y
Ritter C, Andrades ME, Reinke A, Menna-Barreto S, Moreira JC, Dal-Pizzol F (2004) Treatment with N-acetylcysteine plus deferoxamine protects rats against oxidative stress and improves survival in sepsis. Crit Care Med 32:342–349. doi:10.1097/01.CCM.0000109454.13145.CA
Russo PA, Mitchell GA, Tanguay RM (2001) Tyrosinemia: a review. Pediatric and developmental pathology : the official journal of the Society for Pediatric Pathology and the Paediatric Pathology Society 4:212–221
Rustin P, Chretien D, Bourgeron T, Gerard B, Rotig A, Saudubray JM, Munnich A (1994) Biochemical and molecular investigations in respiratory chain deficiencies. Clinica chimica acta; international journal of clinical chemistry 228:35–51
Sagrista ML, Garcia AE, Africa De Madariaga M, Mora M (2002) Antioxidant and pro-oxidant effect of the thiolic compounds N-acetyl-L-cysteine and glutathione against free radical-induced lipid peroxidation. Free Radic Res 36:329–340
Scaini G et al (2012a) Evaluation of acetylcholinesterase in an animal model of maple syrup urine disease. Mol Neurobiol 45:279–286. doi:10.1007/s12035-012-8243-3
Scaini G et al (2012b) DNA damage in an animal model of maple syrup urine disease. Mol Genet Metab 106:169–174. doi:10.1016/j.ymgme.2012.04.009
Scaini G et al (2012c) Antioxidant administration prevents memory impairment in an animal model of maple syrup urine disease. Behav Brain Res 231:92–96. doi:10.1016/j.bbr.2012.03.004
Scaini G et al (2013a) Chronic administration of branched-chain amino acids impairs spatial memory and increases brain-derived neurotrophic factor in a rat model. J Inherit Metab Dis 36:721–730. doi:10.1007/s10545-012-9549-z
Scaini G et al (2013b) Acute and chronic administration of the branched-chain amino acids decreases nerve growth factor in rat hippocampus. Mol Neurobiol 48:581–589. doi:10.1007/s12035-013-8447-1
Scaini G et al (2014) Behavioral responses in rats submitted to chronic administration of branched-chain amino acids. JIMD reports 13:159–167. doi:10.1007/8904_2013_274
Scott CR (2006) The genetic tyrosinemias. Am J Med Genet C: Semin Med Genet 142C:121–126. doi:10.1002/ajmg.c.30092
Sener RN (2005) Tyrosinemia: computed tomography, magnetic resonance imaging, diffusion magnetic resonance imaging, and proton spectroscopy findings in the brain. J Comput Assist Tomogr 29:323–325
Sgaravatti AM et al (2008) Tyrosine promotes oxidative stress in cerebral cortex of young rats. International journal of developmental neuroscience: the official journal of the International Society for Developmental Neuroscience 26:551–559. doi:10.1016/j.ijdevneu.2008.05.007
Sgaravatti AM et al (2009) Tyrosine administration decreases glutathione and stimulates lipid and protein oxidation in rat cerebral cortex. Metab Brain Dis 24:415–425. doi:10.1007/s11011-009-9153-6
Shasi Vardhan K, Pratap Rudra MP, Rao SL (1997) Inhibition of tyrosine aminotransferase by beta-N-oxalyl-L-alpha,beta-diaminopropionic acid, the Lathyrus sativus neurotoxin. J Neurochem 68:2477–2484
Shen XM, Li H, Dryhurst G (2000) Oxidative metabolites of 5-S-cysteinyldopamine inhibit the alpha-ketoglutarate dehydrogenase complex: possible relevance to the pathogenesis of Parkinson's disease. J Neural Transm (Vienna, Austria : 1996) 107:959–978
Sprong RC, Winkelhuyzen-Janssen AM, Aarsman CJ, van Oirschot JF, van der Bruggen T, van Asbeck BS (1998) Low-dose N-acetylcysteine protects rats against endotoxin-mediated oxidative stress, but high-dose increases mortality. Am J Respir Crit Care Med 157:1283–1293. doi:10.1164/ajrccm.157.4.9508063
Srere PA (1969) Citrate synthase. Methods Enzymol 13:3–11. doi:10.1016/0076-6879(69)13005-0
Valikhani M, Akhyani M, Jafari AK, Barzegari M, Toosi S (2006) Oculocutaneous tyrosinaemia or tyrosinaemia type 2: a case report. Journal of the European Academy of Dermatology and Venereology : JEADV 20:591–594. doi:10.1111/j.1468-3083.2006.01572.x
Wurtman RJ, Fernstrom JD (1976) Control of brain neurotransmitter synthesis by precursor availability and nutritional state. Biochem Pharmacol 25:1691–1696
Wurtman RJ, Hefti F, Melamed E (1980) Precursor control of neurotransmitter synthesis. Pharmacol Rev 32:315–335
Wurtman R, Caballero B, Salzman E (1988) Facilitation of levodopa-induced dyskinesias by dietary carbohydrates. N Engl J Med 319:1288–1289. doi:10.1056/NEJM198811103191915
Xiong Y, Peterson PL, Lee CP (1999) Effect of N-acetylcysteine on mitochondrial function following traumatic brain injury in rats. J Neurotrauma 16:1067–1082
Acknowledgements
Laboratory of Bioenergetics (Brazil) are one of the centers of the National Institute for Molecular Medicine (INCT-MM) and one of the members of the Center of Excellence in Applied Neurosciences of Santa Catarina (NENASC). This research was supported by grants from CNPq (ELS), FAPESC (ELS), and UNESC (ELS). ELS is a 1D CNPq Research Fellow.
Author information
Authors and Affiliations
Corresponding author
Additional information
Brena Teodorak and Giselli Scaini contributed equally to this work.
Rights and permissions
About this article
Cite this article
Teodorak, B.P., Scaini, G., Carvalho-Silva, M. et al. Antioxidants reverse the changes in energy metabolism of rat brain after chronic administration of L.-tyrosine. Metab Brain Dis 32, 557–564 (2017). https://doi.org/10.1007/s11011-016-9936-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-016-9936-5