Abstract
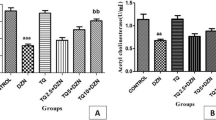
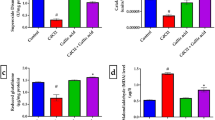
Choline (Ch) is an essential nutrient that seems to be involved in a wide variety of metabolic reactions and functions that affect the nervous system, while thioacetamide (TAA) is a well-known hepatotoxic agent. The induction of prolonged Ch-deprivation (CD) in rats receiving TAA (through the drinking water) provides an experimental model of mild progressive hepatotoxicity that could simulate commonly-presented cases in clinical practice. In this respect, the aim of this study was to investigate the effects of a 30-day dietary CD and/or TAA administration (300 mg/L of drinking water) on the serum total antioxidant status (TAS) and the activities of brain acetylcholinesterase (AChE), Na+,K+-ATPase and Mg2+-ATPase of adult rats. Twenty male Wistar rats were divided into four groups: A (control), B (CD), C (TAA), D (CD+TAA). Dietary CD was provoked through the administration of Ch-deficient diet. Rats were sacrificed by decapitation at the end of the 30-day experimental period and whole brain enzymes were determined spectrophotometrically. Serum TAS was found significantly lowered by CD (−11% vs Control, p < 0.01) and CD+TAA administration (−19% vs Control, p < 0.001), but was not significantly altered due to TAA administration. The rat brain AChE activity was found significantly increased by TAA administration (+11% vs Control, p < 0.01), as well as by CD+TAA administration (+14% vs Control, p < 0.01). However, AChE was not found to be significantly altered by the 30-day dietary CD. On the other hand, CD caused a significant increase in brain Na+,K+-ATPase activity (+16% vs Control, p < 0.05) and had no significant effect on Mg2+-ATPase. Exposure to TAA had no significant effect on Na+,K+-ATPase, but inhibited Mg2+-ATPase (−20% vs Control, p < 0.05). When administered to CD rats, TAA caused a significant decrease in Na+,K+-ATPase activity (−41% vs Control, p < 0.001), but Mg2+-ATPase activity was maintained into control levels. Our data revealed that an adult-onset 30-day dietary-induced CD had no effect on AChE activity. Treatment with TAA not only reversed the stimulatory effect of CD on adult rat brain Na+,K+-ATPase, but caused a dramatic decrease in its activity (−41%). Previous studies have linked this inhibition with metabolic phenomena related to TAA-induced fulminant hepatic failure and encephalopathy. Our data suggest that CD (at least under the examined 30-day period) is an unfavorable background for the effect of TAA-induced hepatic damage on Na+,K+-ATPase activity (an enzyme involved in neuronal excitability, metabolic energy production and neurotransmission).

Similar content being viewed by others
Notes
Ingridients of the Ch-deficient diet: sucrose, coconut oil, starch wheat, dextrine, extracted peanut meal, soy protein, corn oil, dicalcium phosphate, cellulose, potassium citrate, sodium chloride, magnesium oxide, L-cystine, vitamin A, vitamin D3, vitamin E (alpha-tocopherol), copper (copper sulfate pentahydrate), selenium (sodium selenite). Analysis: protein (12%), fat (16%), fiber (2%), ash (3.5%).
References
Avni Y, Shirin H, Aeed H, Shamurov M, Birkenfeld S, Bruck R (2004) Thioacetamide-induced hepatic damage in a rat nutritional model of steatohepatitis. Hepatol Res 30:141–147
Blusztajn JK, Zeisel SH, Wurtman RJ (1985) Developmental changes in the activity of phosphatibylethanolamine N-methyltransferases in rat brain. Biochem J 232:505–511
Bogdanski DF, Tissari A, Brodie BB (1968) Role of sodium, potassium, ouabain and reserpine in uptake, storage and metabolism of biogenic amines in synaptosomes. Life Sci 7:419–428
Bowler K, Tirri R (1974) The temperature characteristics of synaptic membrane ATPases from immature and adult rat brain. J Neurochem 23:611–613
Buchman AL, Dubin MD, Moukarzel AA, Jenden DJ, Roch M, Rice KM, Gornbein J, Ament ME (1995) Choline deficiency: a cause of hepatic steatosis during parenteral nutrition that can be reversed with intravenous choline supplementation. Hepatology 22:1399–1403
Canty DJ, Zeisel SH (1994) Lecithin and choline in human health and disease. Nutr Rev 52:327–339
Cermak JM, Holler T, Jackson DA, Blusztajn JK (1998) Prenatal availability of choline modifies development of the hippocampal cholinergic system. FASEB J 12:349–357
Cohen EL, Wurtman RJ (1976) Brain acetylcholine: control by dietary choline. Science 191:561–562
Davis CD, Uthus EO (2004) DNA methylation, cancer susceptibility, and nutrient interactions. Exp Biol Med (Maywood) 229:988–995
Dwivedi Y, Rastogi R, Garg NK, Dhawan BN (1993) Perfusion with picroliv reverses biochemical changes induced in livers of rats toxicated with galactosamine or thioacetamide. Planta Med 59:418–420
EEC Council (1986) EEC Council Directive 86/609/EEC of 24 November 1986 on the approximation of laws, regulations and administrative provisions of the Member States regarding the protection of animals used for experimental and other scientific purposes. Official Journal of the European Union L358:1–28
Ellman GL, Courtney KD, Andres V Jr, Featherstone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7:88–95
Fernández I, Fontana L, Gil A, Ríosc A, Torres MI (2005) Dietary supplementation with monounsaturated and long-chain polyunsaturated fatty acids influences the liver structural recovery and hepatocyte binuclearity in female Wistar rats in experimental cirrhosis induced by thioacetamide. Exp Toxicol Pathol 57:65–75
Fisher MC, Zeisel SH, Mar MH, Sadler TW (2001) Inhibitors of choline uptake and metabolism cause developmental abnormalities in neurulating mouse embryos. Teratology 64:114–122
Hernandez J (1987) Brain Na+, K+-ATPase activity possibly regulated by a specific serotonin receptor. Brain Res 408:399–402
Kilbinger H, Kruel MR (1981) Choline inhibits acetylcholine release via presynaptic muscarinic receptors. Naunyn-Schmiedeberg’s Arch Pharmacol 316:131–134
Konstandi M, Segos D, Galanopoulou P, Theocharis S, Zarros A, Lang MA, Marselos M, Liapi C (2009) Effects of choline-deprivation on paracetamol- or phenobarbital-induced rat liver metabolic response. J Appl Toxicol 29:101–109
Kouniniotou-Krontiri P, Tsakiris S (1989) Time dependence of Li+ action on acetylcholinesterase activity in correlation with spontaneous quantal release of acetylcholine in rat diaphragm. Jnp J Physiol 39:429–440
Lees GJ, Lehmann A, Sandberg M, Hamberger A (1990) The neurotoxicity of ouabain, a sodium-potassium ATPase inhibitor, in the rat hippocampus. Neurosci Lett 120:159–162
Liapi C, Feskou I, Zarros A, Galanopoulou P, Tsakiris S (2007) Effects of gestational and lactational choline deprivation on brain antioxidant status, acetylcholinesterase, (Na+, K+)- and Mg2+-ATPase activities in offspring rats. Clin Chem Lab Med 45:651–656
Liapi C, Feskou I, Zarros A, Carageorgiou H, Galanopoulou P, Tsakiris S (2008) Equilibrated diet restores the effects of early age choline-deficient feeding on rat brain antioxidant status and enzyme activities: the role of homocysteine, L-phenylalanine and L-alanine. Metab Brain Dis 23:289–301
Lombardi B, Pani P, Schlunk FF (1968) Choline-deficiency fatty liver: impaired release of hepatic triglycerides. J Lipid Res 9:437–446
Lowry OH, Rosebrough NJ, Farr AL, Randale RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Mata M, Fink DJ, Gainer H, Smith CB, Davidsen L, Savakis H, Schwartz WJ, Sokoloff L (1980) Activity-dependent energy metabolism in rat posterior pituitary primarily reflects sodium pump activity. J Neurochem 34:213–215
Molloy AM, Mills JL, Cox C, Daly SF, Conley M, Brody LC, Kirke PN, Scott JM, Ueland PM (2005) Choline and homocysteine interrelations in umbilical cord and maternal plasma at delivery. Am J Clin Nutr 82:836–842
Nikolaev V, Kerimova M, Naydenova E, Ivanov E, Adjarov D (1988) Biochemical changes in the rat after chronic thioacetamide intoxication. Toxicology 48:81–85
Noda S, Masumi S, Moriyama M, Kannan Y, Ohta M, Sugano T, Yamate J (1996) Population of hepatic macrophages and response of perfused liver to platelet-activating factor during production of thioacetamide-induced cirrhosis in rats. Hepatology 24:412–418
Pérez MJ, Suárez A, Gómez-Capilla JA, Sánchez-Medina F, Gil A (2002) Dietary nucleotide supplementation reduces thioacetamide-induced liver fibrosis in rats. J Nutr 132:652–657
Pérez MJ, Sánchez-Medina F, Torres M, Gil A, Suárez A (2004) Dietary nucleotides enhance the liver redox state and protein synthesis in cirrhotic rats. J Nutr 134:2504–2508
Rozhanets VV, Promyslov MS, Gabrielian NI, Shcherbaneva OI (1979) Inhibitory effect of cerebrospinal fluid on the activity of sodium-, potassium-ATPase in animal brain synaptic membranes. Vopr Med Khim 25:71–74
Sanui H, Rubin H (1982) The role of magnesium in cell proliferation and transformation. In: Boynton AL, McKochan WL, Whitfield JP (eds) Ions, cell proliferation and cancer. Academic Press, New York, pp 517–537
Sastry BS, Phillis JW (1977) Antagonism of biogenic amine-induced depression of cerebral cortical neurones by Na+, K+-ATPase inhibitors. Can J Physiol Pharmacol 55:170–180
Streck EL, Zugno AI, Tagliari B, Sarkis JJF, Wajner M, Wannmacher CMD, Wyse ATS (2002) On the mechanism of the inhibition of Na+, K+-ATPase activity caused by homocysteine. Int J Dev Neurosci 20:77–81
Swann AC (1984) (Na+, K+)-adenosine triphosphatase regulation by the sympathetic nervous system: effects of noradrenergic stimulation and lesion in vivo. J Pharmacol Exp Ther 228:304–311
Tsakiris S (2001) Effects of L-phenylalanine on acetylcholinesterase and Na+, K+-ATPase activities in adult and aged rat brain. Mech Ageing Dev 122:491–501
Tsakiris S, Angelogianni P, Schulpis KH, Behrakis P (2000) Protective effect of L-cysteine and glutathione on rat brain Na+,K+-ATPase inhibition induced by free radicals. Z Naturforsch [C] 55:271–277
Yen CN, Maitra A, Lee KF, Jan YY, Chen MF (2004) Thioacetamide-induced intestinal-type cholangiocarcinoma in rat: an animal model recapitulating the multi-stage progression of human cholangiocarcinoma. Carcinogenesis 25:631–636
Zarros A, Theocharis S, Skandali N, Tsakiris S (2008) Effects of fulminant hepatic encephalopathy on the adult rat brain antioxidant status and the activities of acetylcholinesterase, (Na+, K+)- and Mg2+-ATPase: comparison of the enzymes’ response to in vitro treatment with ammonia. Metab Brain Dis 23:255–264
Zeisel SH (1981) Dietary choline: biochemistry, physiology, and pharmacology. Annu Rev Nutr 1:95–121
Zeisel SH (2000) Choline: an essential nutrient for humans. Nutrition 16:669–671
Zeisel SH (2005) Choline, homocysteine, and pregnancy. Am J Clin Nutr 82:719–720
Acknowledgments
This study was funded by the University of Athens. Many thanks are expressed to the medical students Marianna Almpani and Vassilios Memtsas for their assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liapi, C., Al-Humadi, H., Zarros, A. et al. Combined thirty-day exposure to thioacetamide and choline-deprivation alters serum antioxidant status and crucial brain enzyme activities in adult rats. Metab Brain Dis 24, 441–451 (2009). https://doi.org/10.1007/s11011-009-9147-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-009-9147-4