Abstract
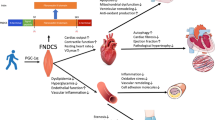
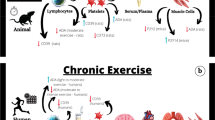
Mechanosensitive ion channels are widely distributed in the heart, lung, bladder and other tissues, and plays an important role in exercise-induced cardiovascular function promotion. By reviewing the PubMed databases, the results were summarized using the terms “Exercise/Sport”, “Piezo1”, “Transient receptor potential (TRP)” and “Cardiovascular” as the keywords, 124-related papers screened were sorted and reviewed. The results showed that: (1) Piezo1 and TRP channels play an important role in regulating blood pressure and the development of cardiovascular diseases such as atherosclerosis, myocardial infarction, and cardiac fibrosis; (2) Exercise promotes cardiac health, inhibits the development of pathological heart to heart failure, regulating the changes in the characterization of Piezo1 and TRP channels; (3) Piezo1 activates downstream signaling pathways with very broad pathways, such as AKT/eNOS, NF-κB, p38MAPK and HIPPO-YAP signaling pathways. Piezo1 and Irisin regulate nuclear localization of YAP and are hypothesized to act synergistically to regulate tissue mechanical properties of the cardiovascular system and (4) The cardioprotective effects of exercise through the TRP family are mostly accomplished through Ca2+ and involve many signaling pathways. TRP channels exert their important cardioprotective effects by reducing the TRPC3-Nox2 complex and mediating Irisin-induced Ca2+ influx through TRPV4. It is proposed that exercise stimulates the mechanosensitive cation channel Piezo1 and TRP channels, which exerts cardioprotective effects. The activation of Piezo1 and TRP channels and their downstream targets to exert cardioprotective function by exercise may provide a theoretical basis for the prevention of cardiovascular diseases and the rehabilitation of clinical patients.




Similar content being viewed by others
Data availability
Enquiries about data availability should be directed to the authors.
References
Kefauver JM, Ward AB, Patapoutian A (2020) Discoveries in structure and physiology of mechanically activated ion channels. Nature 587:567–576. https://doi.org/10.1038/s41586-020-2933-1
Brohawn SG (2015) How ion channels sense mechanical force: insights from mechanosensitive K2P channels TRAAK, TREK1, and TREK2. Ann N Y Acad Sci 1352:20–32. https://doi.org/10.1111/nyas.12874
Brohawn SG, Su Z, MacKinnon R (2014) Mechanosensitivity is mediated directly by the lipid membrane in TRAAK and TREK1 K+ channels. Proc Natl Acad Sci USA 111:3614–3619. https://doi.org/10.1073/pnas.1320768111
Startek JB, Boonen B, Talavera K, Meseguer V (2019) TRP channels as sensors of chemically-induced changes in cell membrane mechanical properties. Int J Mol Sci. https://doi.org/10.3390/ijms20020371
Servin-Vences MR, Moroni M, Lewin GR, Poole K (2017) Direct measurement of TRPV4 and PIEZO1 activity reveals multiple mechanotransduction pathways in chondrocytes. Elife. https://doi.org/10.7554/eLife.21074
Ben-Shahar Y (2011) Sensory functions for degenerin/epithelial sodium channels (DEG/ENaC). Adv Genet 76:1–26. https://doi.org/10.1016/B978-0-12-386481-9.00001-8
Lin SH, Cheng YR, Banks RW, Min MY, Bewick GS, Chen CC (2016) Evidence for the involvement of ASIC3 in sensory mechanotransduction in proprioceptors. Nat Commun 7:11460. https://doi.org/10.1038/ncomms11460
Knoepp F, Ashley Z, Barth D, Baldin JP, Jennings M, Kazantseva M, Saw EL, Katare R, Alvarez de la Rosa D, Weissmann N, Fronius M (2020) Shear force sensing of epithelial Na(+) channel (ENaC) relies on N-glycosylated asparagines in the palm and knuckle domains of alphaENaC. Proc Natl Acad Sci USA 117:717–726. https://doi.org/10.1073/pnas.1911243117
Coste B, Xiao B, Santos JS, Syeda R, Grandl J, Spencer KS, Kim SE, Schmidt M, Mathur J, Dubin AE, Montal M, Patapoutian A (2012) Piezo proteins are pore-forming subunits of mechanically activated channels. Nature 483:176–181. https://doi.org/10.1038/nature10812
Ge J, Li W, Zhao Q, Li N, Chen M, Zhi P, Li R, Gao N, Xiao B, Yang M (2015) Architecture of the mammalian mechanosensitive Piezo1 channel. Nature 527:64–69. https://doi.org/10.1038/nature15247
Wang S, Chennupati R, Kaur H, Iring A, Wettschureck N, Offermanns S (2016) Endothelial cation channel PIEZO1 controls blood pressure by mediating flow-induced ATP release. J Clin Invest 126:4527–4536. https://doi.org/10.1172/JCI87343
Wehrwein EA, Joyner MJ (2013) Regulation of blood pressure by the arterial baroreflex and autonomic nervous system. Handb Clin Neurol 117:89–102. https://doi.org/10.1016/B978-0-444-53491-0.00008-0
Zeng WZ, Marshall KL, Min S, Daou I, Chapleau MW, Abboud FM, Liberles SD, Patapoutian A (2018) PIEZOs mediate neuronal sensing of blood pressure and the baroreceptor reflex. Science 362:464–467. https://doi.org/10.1126/science.aau6324
Baratchi S, Khoshmanesh K, Woodman OL, Potocnik S, Peter K, McIntyre P (2017) Molecular sensors of blood flow in endothelial cells. Trends Mol Med 23:850–868. https://doi.org/10.1016/j.molmed.2017.07.007
Li J, Hou B, Tumova S, Muraki K, Bruns A, Ludlow MJ, Sedo A, Hyman AJ, McKeown L, Young RS, Yuldasheva NY, Majeed Y, Wilson LA, Rode B, Bailey MA, Kim HR, Fu Z, Carter DA, Bilton J, Imrie H, Ajuh P, Dear TN, Cubbon RM, Kearney MT, Prasad RK, Evans PC, Ainscough JF, Beech DJ (2014) Piezo1 integration of vascular architecture with physiological force. Nature 515:279–282. https://doi.org/10.1038/nature13701
Ranade SS, Qiu Z, Woo SH, Hur SS, Murthy SE, Cahalan SM, Xu J, Mathur J, Bandell M, Coste B, Li YS, Chien S, Patapoutian A (2014) Piezo1, a mechanically activated ion channel, is required for vascular development in mice. Proc Natl Acad Sci USA 111:10347–10352. https://doi.org/10.1073/pnas.1409233111
Retailleau K, Duprat F, Arhatte M, Ranade SS, Peyronnet R, Martins JR, Jodar M, Moro C, Offermanns S, Feng Y, Demolombe S, Patel A, Honore E (2015) Piezo1 in smooth muscle cells is involved in hypertension-dependent arterial remodeling. Cell Rep 13:1161–1171. https://doi.org/10.1016/j.celrep.2015.09.072
Kang H, Hong Z, Zhong M, Klomp J, Bayless KJ, Mehta D, Karginov AV, Hu G, Malik AB (2019) Piezo1 mediates angiogenesis through activation of MT1-MMP signaling. Am J Physiol Cell Physiol 316:C92–C103. https://doi.org/10.1152/ajpcell.00346.2018
Beech DJ (2018) Endothelial Piezo1 channels as sensors of exercise. J Physiol 596:979–984. https://doi.org/10.1113/JP274396
Douguet D, Patel A, Xu A, Vanhoutte PM, Honore E (2019) Piezo ion channels in cardiovascular mechanobiology. Trends Pharmacol Sci 40:956–970. https://doi.org/10.1016/j.tips.2019.10.002
Gimbrone MA Jr, Garcia-Cardena G (2016) Endothelial cell dysfunction and the pathobiology of atherosclerosis. Circ Res 118:620–636. https://doi.org/10.1161/CIRCRESAHA.115.306301
Carmeliet E (1999) Cardiac ionic currents and acute ischemia: from channels to arrhythmias. Physiol Rev 79:917–1017. https://doi.org/10.1152/physrev.1999.79.3.917
Dutta S, Minchole A, Quinn TA, Rodriguez B (2017) Electrophysiological properties of computational human ventricular cell action potential models under acute ischemic conditions. Prog Biophys Mol Biol 129:40–52. https://doi.org/10.1016/j.pbiomolbio.2017.02.007
Tran K, Smith NP, Loiselle DS, Crampin EJ (2009) A thermodynamic model of the cardiac sarcoplasmic/endoplasmic Ca(2+) (SERCA) pump. Biophys J 96:2029–2042. https://doi.org/10.1016/j.bpj.2008.11.045
Kormos A, Nagy N, Acsai K, Vaczi K, Agoston S, Pollesello P, Levijoki J, Szentandrassy N, Papp JG, Varro A, Toth A (2014) Efficacy of selective NCX inhibition by ORM-10103 during simulated ischemia/reperfusion. Eur J Pharmacol 740:539–551. https://doi.org/10.1016/j.ejphar.2014.06.033
Clasen L, Angendohr S, Becher S, Bartsch B, Enkel S, Meyer C, Kelm M, Makimoto H, Klocker N (2023) Cardiac ischemia and reperfusion in mice: a comprehensive hemodynamic, electrocardiographic and electrophysiological characterization. Sci Rep 13:5693. https://doi.org/10.1038/s41598-023-32346-5
Jiang F, Yin K, Wu K, Zhang M, Wang S, Cheng H, Zhou Z, Xiao B (2021) The mechanosensitive Piezo1 channel mediates heart mechano-chemo transduction. Nat Commun 12:869. https://doi.org/10.1038/s41467-021-21178-4
Murthy SE, Dubin AE, Patapoutian A (2017) Piezos thrive under pressure: mechanically activated ion channels in health and disease. Nat Rev Mol Cell Biol 18:771–783. https://doi.org/10.1038/nrm.2017.92
Liang J, Huang B, Yuan G, Chen Y, Liang F, Zeng H, Zheng S, Cao L, Geng D, Zhou S (2017) Stretch-activated channel Piezo1 is up-regulated in failure heart and cardiomyocyte stimulated by AngII. Am J Transl Res 9:2945–2955
Niu L, Cheng B, Huang G, Nan K, Han S, Ren H, Liu N, Li Y, Genin GM, Xu F (2022) A positive mechanobiological feedback loop controls bistable switching of cardiac fibroblast phenotype. Cell Discov 8:84. https://doi.org/10.1038/s41421-022-00427-w
Blythe NM, Muraki K, Ludlow MJ, Stylianidis V, Gilbert HTJ, Evans EL, Cuthbertson K, Foster R, Swift J, Li J, Drinkhill MJ, van Nieuwenhoven FA, Porter KE, Beech DJ, Turner NA (2019) Mechanically activated Piezo1 channels of cardiac fibroblasts stimulate p38 mitogen-activated protein kinase activity and interleukin-6 secretion. J Biol Chem 294:17395–17408. https://doi.org/10.1074/jbc.RA119.009167
Albarran-Juarez J, Iring A, Wang S, Joseph S, Grimm M, Strilic B, Wettschureck N, Althoff TF, Offermanns S (2018) Piezo1 and G(q)/G(11) promote endothelial inflammation depending on flow pattern and integrin activation. J Exp Med 215:2655–2672. https://doi.org/10.1084/jem.20180483
Zhang Y, Su SA, Li W, Ma Y, Shen J, Wang Y, Shen Y, Chen J, Ji Y, Xie Y, Ma H, Xiang M (2021) Piezo1-mediated mechanotransduction promotes cardiac hypertrophy by impairing calcium homeostasis to activate calpain/calcineurin signaling. Hypertension 78:647–660. https://doi.org/10.1161/HYPERTENSIONAHA.121.17177
Faucherre A, Moha Ou Maati H, Nasr N, Pinard A, Theron A, Odelin G, Desvignes JP, Salgado D, Collod-Beroud G, Avierinos JF, Lebon G, Zaffran S, Jopling C (2020) Piezo1 is required for outflow tract and aortic valve development. J Mol Cell Cardiol 143:51–62. https://doi.org/10.1016/j.yjmcc.2020.03.013
Emig R, Knodt W, Krussig MJ, Zgierski-Johnston CM, Gorka O, Gross O, Kohl P, Ravens U, Peyronnet R (2021) Piezo1 channels contribute to the regulation of human atrial fibroblast mechanical properties and matrix stiffness sensing. Cells. https://doi.org/10.3390/cells10030663
Braidotti N, Chen SN, Long CS, Cojoc D, Sbaizero O (2022) Piezo1 channel as a potential target for hindering cardiac fibrotic remodeling. Int J Mol Sci. https://doi.org/10.3390/ijms23158065
Lim GB (2022) Piezo1 senses pressure overload and initiates cardiac hypertrophy. Nat Rev Cardiol 19:503. https://doi.org/10.1038/s41569-022-00746-1
Bartoli F, Evans EL, Blythe NM, Stewart L, Chuntharpursat-Bon E, Debant M, Musialowski KE, Lichtenstein L, Parsonage G, Futers TS, Turner NA, Beech DJ (2022) Global PIEZO1 gain-of-function mutation causes cardiac hypertrophy and fibrosis in mice. Cells. https://doi.org/10.3390/cells11071199
MacKay CE, Floen M, Leo MD, Hasan R, Garrud TAC, Fernandez-Pena C, Singh P, Malik KU, Jaggar JH (2022) A plasma membrane-localized polycystin-1/polycystin-2 complex in endothelial cells elicits vasodilation. Elife. https://doi.org/10.7554/eLife.74765
MacKay CE, Leo MD, Fernandez-Pena C, Hasan R, Yin W, Mata-Daboin A, Bulley S, Gammons J, Mancarella S, Jaggar JH (2020) Intravascular flow stimulates PKD2 (polycystin-2) channels in endothelial cells to reduce blood pressure. Elife. https://doi.org/10.7554/eLife.56655
Volk T, Schwoerer AP, Thiessen S, Schultz JH, Ehmke H (2003) A polycystin-2-like large conductance cation channel in rat left ventricular myocytes. Cardiovasc Res 58:76–88. https://doi.org/10.1016/s0008-6363(02)00858-1
Wu G, Markowitz GS, Li L, D’Agati VD, Factor SM, Geng L, Tibara S, Tuchman J, Cai Y, Park JH, van Adelsberg J, Hou H Jr, Kucherlapati R, Edelmann W, Somlo S (2000) Cardiac defects and renal failure in mice with targeted mutations in Pkd2. Nat Genet 24:75–78. https://doi.org/10.1038/71724
Hof T, Chaigne S, Recalde A, Salle L, Brette F, Guinamard R (2019) Transient receptor potential channels in cardiac health and disease. Nat Rev Cardiol 16:344–360. https://doi.org/10.1038/s41569-018-0145-2
Numaga-Tomita T, Nishida M (2020) TRPC channels in cardiac plasticity. Cells. https://doi.org/10.3390/cells9020454
Camacho Londono JE, Tian Q, Hammer K, Schroder L, Camacho Londono J, Reil JC, He T, Oberhofer M, Mannebach S, Mathar I, Philipp SE, Tabellion W, Schweda F, Dietrich A, Kaestner L, Laufs U, Birnbaumer L, Flockerzi V, Freichel M, Lipp P (2015) A background Ca2+ entry pathway mediated by TRPC1/TRPC4 is critical for development of pathological cardiac remodelling. Eur Heart J 36:2257–2266. https://doi.org/10.1093/eurheartj/ehv250
He X, Li S, Liu B, Susperreguy S, Formoso K, Yao J, Kang J, Shi A, Birnbaumer L, Liao Y (2017) Major contribution of the 3/6/7 class of TRPC channels to myocardial ischemia/reperfusion and cellular hypoxia/reoxygenation injuries. Proc Natl Acad Sci USA 114:E4582–E4591. https://doi.org/10.1073/pnas.1621384114
Numaga-Tomita T, Oda S, Nishiyama K, Tanaka T, Nishimura A, Nishida M (2019) TRPC channels in exercise-mimetic therapy. Pflugers Arch 471:507–517. https://doi.org/10.1007/s00424-018-2211-3
Sabourin J, Bartoli F, Antigny F, Gomez AM, Benitah JP (2016) Transient receptor potential canonical (TRPC)/Orai1-dependent store-operated Ca2+ channels: new targets of aldosterone in cardiomyocytes. J Biol Chem 291:13394–13409. https://doi.org/10.1074/jbc.M115.693911
Kitajima N, Watanabe K, Morimoto S, Sato Y, Kiyonaka S, Hoshijima M, Ikeda Y, Nakaya M, Ide T, Mori Y, Kurose H, Nishida M (2011) TRPC3-mediated Ca2+ influx contributes to Rac1-mediated production of reactive oxygen species in MLP-deficient mouse hearts. Biochem Biophys Res Commun 409:108–113. https://doi.org/10.1016/j.bbrc.2011.04.124
Bon RS, Wright DJ, Beech DJ, Sukumar P (2022) Pharmacology of TRPC channels and its potential in cardiovascular and metabolic medicine. Annu Rev Pharmacol Toxicol 62:427–446. https://doi.org/10.1146/annurev-pharmtox-030121-122314
Kumar B, Dreja K, Shah SS, Cheong A, Xu SZ, Sukumar P, Naylor J, Forte A, Cipollaro M, McHugh D, Kingston PA, Heagerty AM, Munsch CM, Bergdahl A, Hultgardh-Nilsson A, Gomez MF, Porter KE, Hellstrand P, Beech DJ (2006) Upregulated TRPC1 channel in vascular injury in vivo and its role in human neointimal hyperplasia. Circ Res 98:557–563. https://doi.org/10.1161/01.RES.0000204724.29685.db
Liu DY, Thilo F, Scholze A, Wittstock A, Zhao ZG, Harteneck C, Zidek W, Zhu ZM, Tepel M (2007) Increased store-operated and 1-oleoyl-2-acetyl-sn-glycerol-induced calcium influx in monocytes is mediated by transient receptor potential canonical channels in human essential hypertension. J Hypertens 25:799–808. https://doi.org/10.1097/HJH.0b013e32803cae2b
Thilo F, Loddenkemper C, Berg E, Zidek W, Tepel M (2009) Increased TRPC3 expression in vascular endothelium of patients with malignant hypertension. Mod Pathol 22:426–430. https://doi.org/10.1038/modpathol.2008.200
Zhang S, Patel HH, Murray F, Remillard CV, Schach C, Thistlethwaite PA, Insel PA, Yuan JX (2007) Pulmonary artery smooth muscle cells from normal subjects and IPAH patients show divergent cAMP-mediated effects on TRPC expression and capacitative Ca2+ entry. Am J Physiol Lung Cell Mol Physiol 292:L1202–L1210. https://doi.org/10.1152/ajplung.00214.2006
Guinamard R, Demion M, Magaud C, Potreau D, Bois P (2006) Functional expression of the TRPM4 cationic current in ventricular cardiomyocytes from spontaneously hypertensive rats. Hypertension 48:587–594. https://doi.org/10.1161/01.HYP.0000237864.65019.a5
Zhao R, Liu X, Qi Z, Yao X, Tsang SY (2021) TRPV1 channels regulate the automaticity of embryonic stem cell-derived cardiomyocytes through stimulating the Na(+) /Ca(2+) exchanger current. J Cell Physiol 236:6806–6823. https://doi.org/10.1002/jcp.30369
Hong J, Lisco AM, Rudebush TL, Yu L, Gao L, Kitzerow O, Zucker IH, Wang HJ (2020) Identification of cardiac expression pattern of transient receptor potential vanilloid type 1 (TRPV1) receptor using a transgenic reporter mouse model. Neurosci Lett 737:135320. https://doi.org/10.1016/j.neulet.2020.135320
Zhong B, Rubinstein J, Ma S, Wang DH (2018) Genetic ablation of TRPV1 exacerbates pressure overload-induced cardiac hypertrophy. Biomed Pharmacother 99:261–270. https://doi.org/10.1016/j.biopha.2018.01.065
Aguettaz E, Bois P, Cognard C, Sebille S (2017) Stretch-activated TRPV2 channels: role in mediating cardiopathies. Prog Biophys Mol Biol 130:273–280. https://doi.org/10.1016/j.pbiomolbio.2017.05.007
Zhang Q, Qi H, Cao Y, Shi P, Song C, Ba L, Chen Y, Gao J, Li S, Li B, Sun H (2018) Activation of transient receptor potential vanilloid 3 channel (TRPV3) aggravated pathological cardiac hypertrophy via calcineurin/NFATc3 pathway in rats. J Cell Mol Med 22:6055–6067. https://doi.org/10.1111/jcmm.13880
Qi H, Ren J, Zhang Q, Cao Y, Ba L, Song C, Shi P, Fu B, Sun H (2019) MiR-103 inhibiting cardiac hypertrophy through inactivation of myocardial cell autophagy via targeting TRPV3 channel in rat hearts. J Cell Mol Med 23:1926–1939. https://doi.org/10.1111/jcmm.14095
Liu Y, Qi H, Shi P, Zhang Q, Li S, Wang Y, Cao Y, Chen Y, Ba L, Gao J, Huang W, Sun H (2018) Transient receptor potential vanilloid-3 (TRPV3) activation plays a central role in cardiac fibrosis induced by pressure overload in rats via TGF-beta(1) pathway. Naunyn Schmiedebergs Arch Pharmacol 391:131–143. https://doi.org/10.1007/s00210-017-1443-7
Peixoto-Neves D, Wang Q, Leal-Cardoso JH, Rossoni LV, Jaggar JH (2015) Eugenol dilates mesenteric arteries and reduces systemic BP by activating endothelial cell TRPV4 channels. Br J Pharmacol 172:3484–3494. https://doi.org/10.1111/bph.13156
Zhang DX, Mendoza SA, Bubolz AH, Mizuno A, Ge ZD, Li R, Warltier DC, Suzuki M, Gutterman DD (2009) Transient receptor potential vanilloid type 4-deficient mice exhibit impaired endothelium-dependent relaxation induced by acetylcholine in vitro and in vivo. Hypertension 53:532–538. https://doi.org/10.1161/HYPERTENSIONAHA.108.127100
Guo S, Huang Y, Zhang Y, Huang H, Hong S, Liu T (2020) Impacts of exercise interventions on different diseases and organ functions in mice. J Sport Health Sci 9:53–73. https://doi.org/10.1016/j.jshs.2019.07.004
Polyak A, Topal L, Zombori-Toth N, Toth N, Prorok J, Kohajda Z, Deri S, Demeter-Haludka V, Hegyi P, Venglovecz V, Agoston G, Husti Z, Gazdag P, Szlovak J, Arpadffy-Lovas T, Naveed M, Sarusi A, Jost N, Virag L, Nagy N, Baczko I, Farkas AS, Varro A (2023) Cardiac electrophysiological remodeling associated with enhanced arrhythmia susceptibility in a canine model of elite exercise. Elife. https://doi.org/10.7554/eLife.80710
Maron BJ, Pelliccia A (2006) The heart of trained athletes: cardiac remodelling and the risks of sports, including sudden death. Circulation 114:1633–1644. https://doi.org/10.1161/CIRCULATIONAHA.106.613562
Gazdag P, Oravecz K, Acsai K, Demeter-Haludka V, Ordog B, Szlovak J, Kohajda Z, Polyak A, Barta BA, Olah A, Radovits T, Merkely B, Papp JG, Baczko I, Varro A, Nagy N, Prorok J (2020) Increased Ca(2+) content of the sarcoplasmic reticulum provides arrhythmogenic trigger source in swimming-induced rat athlete’s heart model. Sci Rep 10:19596. https://doi.org/10.1038/s41598-020-76496-2
Pelliccia A, Day S, Olivotto I (2023) Leisure-time and competitive sport participation: a changing paradigm for HCM patients. Eur J Prev Cardiol. https://doi.org/10.1093/eurjpc/zwad011
Rode B, Shi J, Endesh N, Drinkhill MJ, Webster PJ, Lotteau SJ, Bailey MA, Yuldasheva NY, Ludlow MJ, Cubbon RM, Li J, Futers TS, Morley L, Gaunt HJ, Marszalek K, Viswambharan H, Cuthbertson K, Baxter PD, Foster R, Sukumar P, Weightman A, Calaghan SC, Wheatcroft SB, Kearney MT, Beech DJ (2017) Piezo1 channels sense whole body physical activity to reset cardiovascular homeostasis and enhance performance. Nat Commun 8:350. https://doi.org/10.1038/s41467-017-00429-3
Bartoli F, Debant M, Chuntharpursat-Bon E, Evans EL, Musialowski KE, Parsonage G, Morley LC, Futers TS, Sukumar P, Bowen TS, Kearney MT, Lichtenstein L, Roberts LD, Beech DJ (2022) Endothelial Piezo1 sustains muscle capillary density and contributes to physical activity. J Clin Invest. https://doi.org/10.1172/JCI141775
Wong TY, Juang WC, Tsai CT, Tseng CJ, Lee WH, Chang SN, Cheng PW (2018) Mechanical stretching simulates cardiac physiology and pathology through mechanosensor Piezo1. J Clin Med. https://doi.org/10.3390/jcm7110410
Eisner DA, Caldwell JL, Kistamas K, Trafford AW (2017) Calcium and excitation-contraction coupling in the heart. Circ Res 121:181–195. https://doi.org/10.1161/CIRCRESAHA.117.310230
Jiang Y, Yang X, Jiang J, Xiao B (2021) Structural designs and mechanogating mechanisms of the mechanosensitive Piezo channels. Trends Biochem Sci 46:472–488. https://doi.org/10.1016/j.tibs.2021.01.008
Sciancalepore M, Massaria G, Tramer F, Zacchi P, Lorenzon P, Bernareggi A (2022) A preliminary study on the role of Piezo1 channels in myokine release from cultured mouse myotubes. Biochem Biophys Res Commun 623:148–153. https://doi.org/10.1016/j.bbrc.2022.07.059
Smith JR, Hart CR, Ramos PA, Akinsanya JG, Lanza IR, Joyner MJ, Curry TB, Olson TP (2020) Metabo- and mechanoreceptor expression in human heart failure: relationships with the locomotor muscle afferent influence on exercise responses. Exp Physiol 105:809–818. https://doi.org/10.1113/EP088353
Rubinstein J, Lasko VM, Koch SE, Singh VP, Carreira V, Robbins N, Patel AR, Jiang M, Bidwell P, Kranias EG, Jones WK, Lorenz JN (2014) Novel role of transient receptor potential vanilloid 2 in the regulation of cardiac performance. Am J Physiol Heart Circ Physiol 306:H574–H584. https://doi.org/10.1152/ajpheart.00854.2013
Naticchioni M, Karani R, Smith MA, Onusko E, Robbins N, Jiang M, Radzyukevich T, Fulford L, Gao X, Apel R, Heiny J, Rubinstein J, Koch SE (2015) Transient receptor potential vanilloid 2 regulates myocardial response to exercise. PLoS ONE 10:e0136901. https://doi.org/10.1371/journal.pone.0136901
Cooper MA, Ryals JM, Wu PY, Wright KD, Walter KR, Wright DE (2017) Modulation of diet-induced mechanical allodynia by metabolic parameters and inflammation. J Peripher Nerv Syst 22:39–46. https://doi.org/10.1111/jns.12199
Ye L, Xu M, Hu M, Zhang H, Tan X, Li Q, Shen B, Huang J (2018) TRPV4 is involved in irisin-induced endothelium-dependent vasodilation. Biochem Biophys Res Commun 495:41–45. https://doi.org/10.1016/j.bbrc.2017.10.160
Li X, Han L, Nookaew I, Mannen E, Silva MJ, Almeida M, Xiong J (2019) Stimulation of Piezo1 by mechanical signals promotes bone anabolism. Elife. https://doi.org/10.7554/eLife.49631
Bosutti A, Giniatullin A, Odnoshivkina Y, Giudice L, Malm T, Sciancalepore M, Giniatullin R, D’Andrea P, Lorenzon P, Bernareggi A (2021) “Time window” effect of Yoda1-evoked Piezo1 channel activity during mouse skeletal muscle differentiation. Acta Physiol (Oxf) 233:e13702. https://doi.org/10.1111/apha.13702
Obi S, Nakajima T, Hasegawa T, Kikuchi H, Oguri G, Takahashi M, Nakamura F, Yamasoba T, Sakuma M, Toyoda S, Tei C, Inoue T (1985) Heat induces interleukin-6 in skeletal muscle cells via TRPV1/PKC/CREB pathways. J Appl Physiol 122:683–694. https://doi.org/10.1152/japplphysiol.00139.2016
Fujii N, Kenny GP, McGarr GW, Amano T, Honda Y, Kondo N, Nishiyasu T (2021) TRPV4 channel blockade does not modulate skin vasodilation and sweating during hyperthermia or cutaneous postocclusive reactive and thermal hyperemia. Am J Physiol Regul Integr Comp Physiol 320:R563–R573. https://doi.org/10.1152/ajpregu.00123.2020
Krout D, Schaar A, Sun Y, Sukumaran P, Roemmich JN, Singh BB, Claycombe-Larson KJ (2017) The TRPC1 Ca(2+)-permeable channel inhibits exercise-induced protection against high-fat diet-induced obesity and type II diabetes. J Biol Chem 292:20799–20807. https://doi.org/10.1074/jbc.M117.809954
Craighead DH, Shank SW, Gottschall JS, Passe DH, Murray B, Alexander LM, Kenney WL (2017) Ingestion of transient receptor potential channel agonists attenuates exercise-induced muscle cramps. Muscle Nerve 56:379–385. https://doi.org/10.1002/mus.25611
Iring A, Jin YJ, Albarran-Juarez J, Siragusa M, Wang S, Dancs PT, Nakayama A, Tonack S, Chen M, Kunne C, Sokol AM, Gunther S, Martinez A, Fleming I, Wettschureck N, Graumann J, Weinstein LS, Offermanns S (2019) Shear stress-induced endothelial adrenomedullin signaling regulates vascular tone and blood pressure. J Clin Invest 129:2775–2791. https://doi.org/10.1172/JCI123825
Cinar E, Zhou S, DeCourcey J, Wang Y, Waugh RE, Wan J (2015) Piezo1 regulates mechanotransductive release of ATP from human RBCs. Proc Natl Acad Sci U S A 112:11783–11788. https://doi.org/10.1073/pnas.1507309112
Fleming I (2010) Molecular mechanisms underlying the activation of eNOS. Pflugers Arch 459:793–806. https://doi.org/10.1007/s00424-009-0767-7
Davies PF (2009) Hemodynamic shear stress and the endothelium in cardiovascular pathophysiology. Nat Clin Pract Cardiovasc Med 6:16–26. https://doi.org/10.1038/ncpcardio1397
Wang C, Baker BM, Chen CS, Schwartz MA (2013) Endothelial cell sensing of flow direction. Arterioscler Thromb Vasc Biol 33:2130–2136. https://doi.org/10.1161/ATVBAHA.113.301826
Boo YC, Hwang J, Sykes M, Michell BJ, Kemp BE, Lum H, Jo H (2002) Shear stress stimulates phosphorylation of eNOS at Ser(635) by a protein kinase A-dependent mechanism. Am J Physiol Heart Circ Physiol 283:H1819–H1828. https://doi.org/10.1152/ajpheart.00214.2002
Molkentin JD, Bugg D, Ghearing N, Dorn LE, Kim P, Sargent MA, Gunaje J, Otsu K, Davis J (2017) Fibroblast-specific genetic manipulation of p38 mitogen-activated protein kinase in vivo reveals its central regulatory role in fibrosis. Circulation 136:549–561. https://doi.org/10.1161/CIRCULATIONAHA.116.026238
Lai A, Thurgood P, Cox CD, Chheang C, Peter K, Jaworowski A, Khoshmanesh K, Baratchi S (2022) Piezo1 response to shear stress is controlled by the components of the extracellular matrix. ACS Appl Mater Interfaces 14:40559–40568. https://doi.org/10.1021/acsami.2c09169
Wang KC, Yeh YT, Nguyen P, Limqueco E, Lopez J, Thorossian S, Guan KL, Li YJ, Chien S (2016) Flow-dependent YAP/TAZ activities regulate endothelial phenotypes and atherosclerosis. Proc Natl Acad Sci USA 113:11525–11530. https://doi.org/10.1073/pnas.1613121113
Mia MM, Cibi DM, Ghani S, Singh A, Tee N, Sivakumar V, Bogireddi H, Cook SA, Mao J, Singh MK (2022) Loss of Yap/Taz in cardiac fibroblasts attenuates adverse remodelling and improves cardiac function. Cardiovasc Res 118:1785–1804. https://doi.org/10.1093/cvr/cvab205
Yang Y, Wang D, Zhang C, Yang W, Li C, Gao Z, Pei K, Li Y (2022) Piezo1 mediates endothelial atherogenic inflammatory responses via regulation of YAP/TAZ activation. Hum Cell 35:51–62. https://doi.org/10.1007/s13577-021-00600-5
Xiong Y, Dong L, Bai Y, Tang H, Li S, Luo D, Liu F, Bai J, Yang S, Song X (2022) Piezo1 activation facilitates ovarian cancer metastasis via Hippo/YAP signaling axis. Channels (Austin) 16:159–166. https://doi.org/10.1080/19336950.2022.2099381
Wang L, You X, Lotinun S, Zhang L, Wu N, Zou W (2020) Mechanical sensing protein PIEZO1 regulates bone homeostasis via osteoblast-osteoclast crosstalk. Nat Commun 11:282. https://doi.org/10.1038/s41467-019-14146-6
Pathak MM, Nourse JL, Tran T, Hwe J, Arulmoli J, Le DT, Bernardis E, Flanagan LA, Tombola F (2014) Stretch-activated ion channel Piezo1 directs lineage choice in human neural stem cells. Proc Natl Acad Sci USA 111:16148–16153. https://doi.org/10.1073/pnas.1409802111
Liu S, Xu X, Fang Z, Ning Y, Deng B, Pan X, He Y, Yang Z, Huang K, Li J (2021) Piezo1 impairs hepatocellular tumor growth via deregulation of the MAPK-mediated YAP signaling pathway. Cell Calcium 95:102367. https://doi.org/10.1016/j.ceca.2021.102367
Panciera T, Azzolin L, Cordenonsi M, Piccolo S (2017) Mechanobiology of YAP and TAZ in physiology and disease. Nat Rev Mol Cell Biol 18:758–770. https://doi.org/10.1038/nrm.2017.87
Wang L, Luo JY, Li B, Tian XY, Chen LJ, Huang Y, Liu J, Deng D, Lau CW, Wan S, Ai D, Mak KK, Tong KK, Kwan KM, Wang N, Chiu JJ, Zhu Y, Huang Y (2016) Integrin-YAP/TAZ-JNK cascade mediates atheroprotective effect of unidirectional shear flow. Nature 540:579–582. https://doi.org/10.1038/nature20602
He L, Si G, Huang J, Samuel ADT, Perrimon N (2018) Mechanical regulation of stem-cell differentiation by the stretch-activated Piezo channel. Nature 555:103–106. https://doi.org/10.1038/nature25744
Nakajima H, Yamamoto K, Agarwala S, Terai K, Fukui H, Fukuhara S, Ando K, Miyazaki T, Yokota Y, Schmelzer E, Belting HG, Affolter M, Lecaudey V, Mochizuki N (2017) Flow-dependent endothelial YAP regulation contributes to vessel maintenance. Dev Cell 40(523–536):e6. https://doi.org/10.1016/j.devcel.2017.02.019
Duchemin AL, Vignes H, Vermot J (2019) Mechanically activated piezo channels modulate outflow tract valve development through the Yap1 and Klf2-Notch signaling axis. Elife. https://doi.org/10.7554/eLife.44706
Mohri Z, Del Rio HA, Krams R (2017) The emerging role of YAP/TAZ in mechanotransduction. J Thorac Dis 9:E507–E509. https://doi.org/10.21037/jtd.2017.03.179
Tang Z, Wei X, Li T, Wu H, Xiao X, Hao Y, Li S, Hou W, Shi L, Li X, Guo Z (2021) Three-dimensionally printed Ti2448 with low stiffness enhanced angiogenesis and osteogenesis by regulating macrophage polarization via Piezo1/YAP signaling axis. Front Cell Dev Biol 9:750948. https://doi.org/10.3389/fcell.2021.750948
Nabel EG, Braunwald E (2012) A tale of coronary artery disease and myocardial infarction. N Engl J Med 366:54–63. https://doi.org/10.1056/NEJMra1112570
He L, Nguyen NB, Ardehali R, Zhou B (2020) Heart regeneration by endogenous stem cells and cardiomyocyte proliferation: controversy, fallacy, and progress. Circulation 142:275–291. https://doi.org/10.1161/CIRCULATIONAHA.119.045566
Xu L, Chen Z, Geng T, Ru B, Wan Q, Zhang J, Li S, Cai W (2022) Irisin promotes the proliferation and tenogenic differentiation of rat tendon-derived stem/progenitor cells via activating YAP/TAZ. In Vitro Cell Dev Biol Anim 58:658–668. https://doi.org/10.1007/s11626-022-00699-2
Yang Y, Geng T, Samara A, Olstad OK, He J, Agger AE, Skallerud BH, Landin MA, Heyward CA, Pullisaar H, Reseland JE (2023) Recombinant irisin enhances the extracellular matrix formation, remodeling potential, and differentiation of human periodontal ligament cells cultured in 3D. J Periodontal Res 58:336–349. https://doi.org/10.1111/jre.13094
Song K, Nam YJ, Luo X, Qi X, Tan W, Huang GN, Acharya A, Smith CL, Tallquist MD, Neilson EG, Hill JA, Bassel-Duby R, Olson EN (2012) Heart repair by reprogramming non-myocytes with cardiac transcription factors. Nature 485:599–604. https://doi.org/10.1038/nature11139
Huang C, Tu W, Fu Y, Wang J, Xie X (2018) Chemical-induced cardiac reprogramming in vivo. Cell Res 28:686–689. https://doi.org/10.1038/s41422-018-0036-4
Yu Q, Kou W, Xu X, Zhou S, Luan P, Xu X, Li H, Zhuang J, Wang J, Zhao Y, Xu Y, Peng W (2019) FNDC5/Irisin inhibits pathological cardiac hypertrophy. Clin Sci (Lond) 133:611–627. https://doi.org/10.1042/CS20190016
Li H, Qin S, Liang Q, Xi Y, Bo W, Cai M, Tian Z (2021) Exercise training enhances myocardial mitophagy and improves cardiac function via Irisin/FNDC5-PINK1/Parkin pathway in MI mice. Biomedicines. https://doi.org/10.3390/biomedicines9060701
Zhang M, Xu Y, Jiang L (2019) Irisin attenuates oxidized low-density lipoprotein impaired angiogenesis through AKT/mTOR/S6K1/Nrf2 pathway. J Cell Physiol 234:18951–18962. https://doi.org/10.1002/jcp.28535
Maak S, Norheim F, Drevon CA, Erickson HP (2021) Progress and challenges in the biology of FNDC5 and Irisin. Endocr Rev 42:436–456. https://doi.org/10.1210/endrev/bnab003
Liu S, Cui F, Ning K, Wang Z, Fu P, Wang D, Xu H (2022) Role of irisin in physiology and pathology. Front Endocrinol (Lausanne) 13:962968. https://doi.org/10.3389/fendo.2022.962968
Shinge SAU, Zhang D, Din AU, Yu F, Nie Y (2022) Emerging Piezo1 signaling in inflammation and atherosclerosis; a potential therapeutic target. Int J Biol Sci 18:923–941. https://doi.org/10.7150/ijbs.63819
Chen T, Lin Y, Wu Z, Shi H, Hu W, Li S, Que Y, Qiu J, Li P, Qiu X, Liang T, Wang X, Gao B, Zhou H, Deng Z, Chen Y, Zhu Y, Peng Y, Liang A, Gao W, Huang D (2022) Irisin ameliorates intervertebral disc degeneration by activating LATS/YAP/CTGF signaling. Oxid Med Cell Longev 2022:9684062. https://doi.org/10.1155/2022/9684062
Inoue K, Fujie S, Hasegawa N, Horii N, Uchida M, Iemitsu K, Sanada K, Hamaoka T, Iemitsu M (2020) Aerobic exercise training-induced irisin secretion is associated with the reduction of arterial stiffness via nitric oxide production in adults with obesity. Appl Physiol Nutr Metab 45:715–722. https://doi.org/10.1139/apnm-2019-0602
Zhang Y, Mu Q, Zhou Z, Song H, Zhang Y, Wu F, Jiang M, Wang F, Zhang W, Li L, Shao L, Wang X, Li S, Yang L, Wu Q, Zhang M, Tang D (2016) Protective effect of irisin on atherosclerosis via suppressing oxidized low density lipoprotein induced vascular inflammation and endothelial dysfunction. PLoS ONE 11:e0158038. https://doi.org/10.1371/journal.pone.0158038
Shimauchi T, Numaga-Tomita T, Ito T, Nishimura A, Matsukane R, Oda S, Hoka S, Ide T, Koitabashi N, Uchida K, Sumimoto H, Mori Y, Nishida M (2017) TRPC3-Nox2 complex mediates doxorubicin-induced myocardial atrophy. JCI Insight. https://doi.org/10.1172/jci.insight.93358
Funding
This work was supported by National Natural Science Foundation of China Grants (31900828 to Y.X. and 32171128 to Z.T.) and the Fundamental Research Funds for the Central Universities (GK202301013 to Y.X.).
Author information
Authors and Affiliations
Contributions
XD, YX and ZT established the theme of the review and constructed the framework; XD and YX searched the papers and did analysis; XD, RL and YX drafted manuscript; XD and YX prepared the figures; XD, RL, YX and ZT revised manuscript and approved the final version.
Corresponding author
Ethics declarations
Competing interests
The authors declare that there are no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Duan, X., Liu, R., Xi, Y. et al. The mechanisms of exercise improving cardiovascular function by stimulating Piezo1 and TRP ion channels: a systemic review. Mol Cell Biochem (2024). https://doi.org/10.1007/s11010-024-05000-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11010-024-05000-5