Abstract
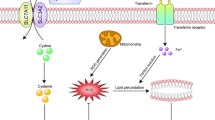
Unlike other death forms, such as autophagy, necrosis, and apoptosis, ferroptosis is a novel type of programmed cell death with iron-dependent properties. Esteroxygenase affects the content of unsaturated fatty acids and promotes lipid peroxidation. In addition, GSH can cause the reduction of GPX4, which can cause ferroptosis. P53 and its signaling pathways also regulate ferroptosis. Recent studies have confirmed that ferroptosis also promotes the death of RGC. The progressive loss of RGC is one of the pathological features of glaucoma, indicating that ferroptosis may be related to the onset of glaucoma. Down-regulation of GPX4 leads to the loss of nerve cells, which suggests that ferroptosis may also be related to diseases related to optic nerve damage. At present, ferroptosis has been extensively researched and advanced in systemic diseases, such as cardiovascular diseases, gastrointestinal tumors such as stomach, liver, and pancreas, and brain diseases. This review focuses on the research progress of ferroptosis in ophthalmic diseases, especially glaucoma and optic nerve damage.
Similar content being viewed by others
Data availability
These authors have not disclosed any data availability.
References
Hirschhorn T, Stockwell BR (2019) The development of the concept of ferroptosis. Free Radic Biol Med 133:130–143
Liu J, Zhang C, Wang J, Hu W, Feng Z (2020) The regulation of ferroptosis by tumor suppressor p53 and its pathway. Int J Mol Sci 21(21):8387
Bertrand RL (2017) Iron accumulation, glutathione depletion, and lipid peroxidation must occur simultaneously during ferroptosis and are mutually amplifying events. Med Hypotheses 101:69–74
Stockwell BR, Friedmann Angeli JP, Bayir H, Bush AI, Conrad M, Dixon SJ, Fulda S, Gascón S, Hatzios SK, Kagan VE, Noel K, Jiang X, Linkermann A, Murphy ME, Overholtzer M, Oyagi A, Pagnussat GC, Park J, Ran Q, Rosenfeld CS, Salnikow K, Tang D, Torti FM, Torti SV, Toyokuni S, Woerpel KA, Zhang DD (2017) Ferroptosis: a regulated cell death nexus linking metabolism, redox biology, and disease. Cell 171(2):273–285
Ng SW, Norwitz SG, Norwitz ER (2019) The impact of iron overload and ferroptosis on reproductive disorders in humans: implications for preeclampsia. Int J Mol Sci 20(13):3283
Gao M, Monian P, Quadri N, Ramasamy R, Jiang X (2015) Glutaminolysis and transferrin regulate ferroptosis. Mol Cell 59(2):298–308
Mandal PK, Seiler A, Perisic T, Kölle P, Banjac Canak A, Förster H, Weiss N, Kremmer E, Lieberman MW, Bannai S, Kuhlencordt P, Sato H, Bornkamm GW, Conrad M (2010) System x(c)- and thioredoxin reductase 1 cooperatively rescue glutathione deficiency. J Biol Chem 285(29):22244–22253
Aoyama K, Nakaki T (2015) Glutathione in cellular redox homeostasis: association with the excitatory amino acid carrier 1 (EAAC1). Molecules 20(5):8742–8758
Stockwell BR, Jiang X, Gu W (2020) Emerging mechanisms and disease relevance of ferroptosis. Trends Cell Biol 30(6):478–490
Conrad M, Friedmann Angeli JP (2015) Glutathione peroxidase 4 (Gpx4) and ferroptosis: what’s so special about it? Mol Cell Oncol 2(3):e995047
Liu DS, Duong CP, Haupt S, Montgomery KG, House CM, Azar WJ, Pearson HB, Fisher OM, Read M, Guerra GR, Haupt Y, Cullinane C, Wiman KG, Abrahmsen L, Phillips WA, Clemons NJ (2017) Inhibiting the system xc-/glutathione axis selectively targets cancers with mutant-p53 accumulation. Nat Commun 8:14844
Li W, Li W, Leng Y, Xiong Y, Xia Z (2020) Ferroptosis is involved in diabetes myocardial ischemia/reperfusion injury through endoplasmic reticulum stress. DNA Cell Biol 39(2):210–225
Fang X, Wang H, Han D, Xie E, Yang X, Wei J, Gu S, Gao F, Zhu N, Yin X, Cheng Q, Zhang P, Dai W, Chen J, Yang F, Yang HT, Linkermann A, Gu W, Min J, Wang F (2019) Ferroptosis as a target for protection against cardiomyopathy. Proc Natl Acad Sci USA 116(7):2672–2680
Wang J, Deng B, Liu Q, Huang Y, Chen W, Li J, Zhou Z, Zhang L, Liang B, He J, Chen Z, Yan C, Yang Z, Xian S, Wang L (2020) Pyroptosis and ferroptosis induced by mixed lineage kinase 3 (MLK3) signaling in cardiomyocytes are essential for myocardial fibrosis in response to pressure overload. Cell Death Dis 11(7):574
Yin Z, Ding G, Chen X, Qin X, Xu H, Zeng B, Ren J, Zheng Q, Wang S (2020) Beclin1 haploinsufficiency rescues low ambient temperature-induced cardiac remodeling and contractile dysfunction through inhibition of ferroptosis and mitochondrial injury. Metabolism 113:154397
Yang M, Chen P, Liu J, Zhu S, Kroemer G, Klionsky DJ, Lotze MT, Zeh HJ, Kang R, Tang D (2019) Clockophagy is a novel selective autophagy process favoring ferroptosis. Sci Adv. https://doi.org/10.1126/sciadv.aaw2238
Cancel LM, Ebong EE, Mensah S, Hirschberg C, Tarbell JM (2016) Endothelial glycocalyx, apoptosis and inflammation in an atherosclerotic mouse model. Atherosclerosis 252:136–146
Tuo QZ, Lei P, Jackman KA, Li XL, Xiong H, Li XL, Liuyang ZY, Roisman L, Zhang ST, Ayton S, Wang Q, Crouch PJ, Ganio K, Wang XC, Pei L, Adlard PA, Lu YM, Cappai R, Wang JZ, Liu R, Bush AI (2017) Tau-mediated iron export prevents ferroptotic damage after ischemic stroke. Mol Psychiatry 22(11):1520–1530
Massie A, Schallier A, Kim SW, Fernando R, Kobayashi S, Beck H, De Bundel D, Vermoesen K, Bannai S, Smolders I, Conrad M, Plesnila N, Sato H, Michotte Y (2011) Dopaminergic neurons of system x(c)−-deficient mice are highly protected against 6-hydroxydopamine-induced toxicity. FASEB J 25(4):1359–1369
Van Do B, Gouel F, Jonneaux A, Timmerman K, Gelé P, Pétrault M, Bastide M, Laloux C, Moreau C, Bordet R, Devos D, Devedjian JC (2016) Ferroptosis, a newly characterized form of cell death in Parkinson’s disease that is regulated by PKC. Neurobiol Dis 94:169–178
Fonseca-Nunes A, Jakszyn P, Agudo A (2014) Iron and cancer risk: a systematic review and meta-analysis of the epidemiological evidence. Cancer Epidemiol Biomark Prev 23(1):12–31
Taira N, Nihira K, Yamaguchi T, Miki Y, Yoshida K (2007) DYRK2 is targeted to the nucleus and controls p53 via Ser46 phosphorylation in the apoptotic response to DNA damage. Mol Cell 25(5):725–738
Jennis M, Kung CP, Basu S, Budina-Kolomets A, Leu JI, Khaku S, Scott JP, Cai KQ, Campbell MR, Porter DK, Wang X, Bell DA, Li X, Garlick DS, Liu Q, Hollstein M, George DL, Murphy ME (2016) An African-specific polymorphism in the TP53 gene impairs p53 tumor suppressor function in a mouse model. Genes Dev 30(8):918–930
Sun X, Ou Z, Chen R, Niu X, Chen D, Kang R, Tang D (2016) Activation of the p62-Keap1-NRF2 pathway protects against ferroptosis in hepatocellular carcinoma cells. Hepatology 63(1):173–184
Badgley MA, Kremer DM, Maurer HC, DelGiorno KE, Lee HJ, Purohit V, Sagalovskiy IR, Ma A, Kapilian J, Firl CEM, Decker AR, Sastra SA, Palermo CF, Andrade LR, Sajjakulnukit P, Zhang L, Tolstyka ZP, Hirschhorn T, Lamb C, Liu T, Gu W, Seeley ES, Stone E, Georgiou G, Manor U, Iuga A, Wahl GM, Stockwell BR, Lyssiotis CA, Olive KP (2020) Cysteine depletion induces pancreatic tumor ferroptosis in mice. Science 368(6486):85–89
Eling N, Reuter L, Hazin J, Hamacher-Brady A, Brady NR (2015) Identification of artesunate as a specific activator of ferroptosis in pancreatic cancer cells. Oncoscience 2(5):517–532
Hou W, Xie Y, Song X, Sun X, Lotze MT, Zeh HJ 3rd, Kang R, Tang D (2016) Autophagy promotes ferroptosis by degradation of ferritin. Autophagy 12(8):1425–1428
Ishimoto T, Nagano O, Yae T, Tamada M, Motohara T, Oshima H, Oshima M, Ikeda T, Asaba R, Yagi H, Masuko T, Shimizu T, Ishikawa T, Kai K, Takahashi E, Imamura Y, Baba Y, Ohmura M, Suematsu M, Baba H, Saya H (2011) CD44 variant regulates redox status in cancer cells by stabilizing the xCT subunit of system xc(−) and thereby promotes tumor growth. Cancer Cell 19(3):387–400
Ye Z, Liu W, Zhuo Q, Hu Q, Liu M, Sun Q, Zhang Z, Fan G, Xu W, Ji S, Yu X, Qin Y, Xu X (2020) Ferroptosis: final destination for cancer? Cell Prolif 53(3):e12761
Sharfuddin AA, Molitoris BA (2011) Pathophysiology of ischemic acute kidney injury. Nat Rev Nephrol 7(4):189–200
Linkermann A, Bräsen JH, Darding M, Jin MK, Sanz AB, Heller JO, De Zen F, Weinlich R, Ortiz A, Walczak H, Weinberg JM, Green DR, Kunzendorf U, Krautwald S (2013) Two independent pathways of regulated necrosis mediate ischemia-reperfusion injury. Proc Natl Acad Sci USA 110(29):12024–12029
Jang HR, Rabb H (2009) The innate immune response in ischemic acute kidney injury. Clin Immunol 130(1):41–50
Peng JJ, Song WT, Yao F, Zhang X, Peng J, Luo XJ, Xia XB (2020) Involvement of regulated necrosis in blinding diseases: focus on necroptosis and ferroptosis. Exp Eye Res 191:107922
Ruan Y, Jiang S, Musayeva A, Pfeiffer N, Gericke A (2021) Corneal epithelial stem cells-physiology, pathophysiology and therapeutic options. Cells 10(9):2302
Bath C (2013) Human corneal epithelial subpopulations: oxygen dependent ex vivo expansion and transcriptional profiling. Acta Ophthalmol 91(4):1–34
Sakai O, Uchida T, Imai H, Ueta T (2016) Glutathione peroxidase 4 plays an important role in oxidative homeostasis and wound repair in corneal epithelial cells. FEBS Open Bio 6(12):1238–1247
Chou R, Dana T, Bougatsos C, Grusing S, Blazina I (2016) Screening for impaired visual acuity in older adults: updated evidence report and systematic review for the US preventive services task force. JAMA 315(9):915–933
Cao JY, Dixon SJ (2016) Mechanisms of ferroptosis. Cell Mol Life Sci 73(11–12):2195–2209
Zhao T, Guo X, Sun Y (2021) Iron accumulation and lipid peroxidation in the aging retina: implication of ferroptosis in age-related macular degeneration. Aging Dis 12(2):529–551
Hernández-Zimbrón LF, Zamora-Alvarado R, Ochoa-De la Paz L, Velez-Montoya R, Zenteno E, Gulias-Cañizo R, Quiroz-Mercado H, Gonzalez-Salinas R (2018) Age-related macular degeneration: new paradigms for treatment and management of AMD. Oxid Med Cell Longev 2018:8374647
Totsuka K, Ueta T, Uchida T, Roggia MF, Nakagawa S, Vavvas DG, Honjo M, Aihara M (2019) Oxidative stress induces ferroptotic cell death in retinal pigment epithelial cells. Exp Eye Res 181:316–324
Ola MS, Alhomida AS, LaNoue KF (2019) Gabapentin attenuates oxidative stress and apoptosis in the diabetic rat retina. Neurotox Res 36(1):81–90
Zhang YH, Wang DW, Xu SF, Zhang S, Fan YG, Yang YY, Guo SQ, Wang S, Guo T, Wang ZY, Guo C (2018) α-Lipoic acid improves abnormal behavior by mitigation of oxidative stress, inflammation, ferroptosis, and tauopathy in P301S Tau transgenic mice. Redox Biol 14:535–548
Ning A, Cui J, To E, Ashe KH, Matsubara J (2008) Amyloid-beta deposits lead to retinal degeneration in a mouse model of Alzheimer disease. Invest Ophthalmol Vis Sci 49(11):5136–5143
Yang X, Huo F, Liu B, Liu J, Chen T, Li J, Zhu Z, Lv B (2017) Crocin inhibits oxidative stress and pro-inflammatory response of microglial cells associated with diabetic retinopathy through the activation of PI3K/Akt signaling pathway. J Mol Neurosci 61(4):581–589
He J, Li Z, Xia P, Shi A, FuChen X, Zhang J, Yu P (2022) Ferroptosis and ferritinophagy in diabetes complications. Mol Metab 60:101470
Sun Y, Zheng Y, Wang C, Liu Y (2018) Glutathione depletion induces ferroptosis, autophagy, and premature cell senescence in retinal pigment epithelial cells. Cell Death Dis 9(7):753
Zhang J, Sheng S, Wang W, Dai J, Zhong Y, Ren J, Jiang K, Li S, Bian X, Liu L (2022) Molecular mechanisms of iron mediated programmed cell death and its roles in eye diseases. Front Nutr 9:844757
Wang D, Tang L, Zhang Y, Ge G, Jiang X, Mo Y, Wu P, Deng X, Li L, Zuo S, Yan Q, Zhang S, Wang F, Shi L, Li X, Xiang B, Zhou M, Liao Q, Guo C, Zeng Z, Xiong W, Gong Z (2022) Regulatory pathways and drugs associated with ferroptosis in tumors. Cell Death Dis 13(6):544
Jelinek A, Heyder L, Daude M, Plessner M, Krippner S, Grosse R, Diederich WE, Culmsee C (2018) Mitochondrial rescue prevents glutathione peroxidase-dependent ferroptosis. Free Radic Biol Med 117:45–57
Kuganesan N, Dlamini S, Tillekeratne LMV, Taylor WR (2021) Tumor suppressor p53 promotes ferroptosis in oxidative stress conditions independent of modulation of ferroptosis by p21, CDKs, RB, and E2F. J Biol Chem 297(6):101365
Tham YC, Li X, Wong TY, Quigley HA, Aung T, Cheng CY (2014) Global prevalence of glaucoma and projections of glaucoma burden through 2040: a systematic review and meta-analysis. Ophthalmology 121(11):2081–2090
Burton MJ, Ramke J, Marques AP, Bourne RRA, Congdon N, Jones I, Ah Tong BAM, Arunga S, Bachani D, Bascaran C, Bastawrous A, Blanchet K, Braithwaite T, Buchan JC, Cairns J, Cama A, Chagunda M, Chuluunkhuu C, Cooper A, Crofts-Lawrence J, Dean WH, Denniston AK, Ehrlich JR, Emerson PM, Evans JR, Frick KD, Friedman DS, Furtado JM, Gichangi MM, Gichuhi S, Gilbert SS, Gurung R, Habtamu E, Holland P, Jonas JB, Keane PA, Keay L, Khanna RC, Khaw PT, Kuper H, Kyari F, Lansingh VC, Mactaggart I, Mafwiri MM, Mathenge W, McCormick I, Morjaria P, Mowatt L, Muirhead D, Murthy GVS, Mwangi N, Patel DB, Peto T, Qureshi BM, Salomão SR, Sarah V, Shilio BR, Solomon AW, Swenor BK, Taylor HR, Wang N, Webson A, West SK, Wong TY, Wormald R, Yasmin S, Yusufu M, Silva JC, Resnikoff S, Ravilla T, Gilbert CE, Foster A, Faal HB (2021) The lancet global health commission on global eye health: vision beyond 2020. Lancet Glob Health 9(4):e489–e551
Quigley HA (1996) Number of people with glaucoma worldwide. Br J Ophthalmol 80(5):389–393
Jonas JB, Budde WM, Panda-Jonas S (1999) Ophthalmoscopic evaluation of the optic nerve head. Surv Ophthalmol 43(4):293–320
Lambiase A, Aloe L, Centofanti M, Parisi V, Báo SN, Mantelli F, Colafrancesco V, Manni GL, Bucci MG, Bonini S, Levi-Montalcini R (2009) Experimental and clinical evidence of neuroprotection by nerve growth factor eye drops: implications for glaucoma. Proc Natl Acad Sci USA 106(32):13469–13474
Tezel G (2021) Multifactorial pathogenic processes of retinal ganglion cell degeneration in glaucoma towards multi-target strategies for broader treatment effects. Cells 10(6):1372
McMonnies C (2018) Reactive oxygen species, oxidative stress, glaucoma and hyperbaric oxygen therapy. J Optom 11(1):3–9
Jünemann AG, Stopa P, Michalke B, Chaudhri A, Reulbach U, Huchzermeyer C, Schlötzer-Schrehardt U, Kruse FE, Zrenner E, Rejdak R (2013) Levels of aqueous humor trace elements in patients with non-exsudative age-related macular degeneration: a case-control study. PLoS ONE 8(2):e56734
Negoro S, Kunisada K, Tone E, Funamoto M, Oh H, Kishimoto T, Yamauchi-Takihara K (2000) Activation of JAK/STAT pathway transduces cytoprotective signal in rat acute myocardial infarction. Cardiovasc Res 47(4):797–805
Kuehn MH, Fingert JH, Kwon YH (2005) Retinal ganglion cell death in glaucoma: mechanisms and neuroprotective strategies. Ophthalmol Clin N Am 18(3):383–395
Lukasiewicz PD (2005) Synaptic mechanisms that shape visual signaling at the inner retina. Prog Brain Res 147:205–218
Kamińska A, Romano GL, Rejdak R, Zweifel S, Fiedorowicz M, Rejdak M, Bajka A, Amato R, Bucolo C, Avitabile T, Drago F, Toro MD (2021) Influence of trace elements on neurodegenerative diseases of the eye-the glaucoma model. Int J Mol Sci 22(9):4323
Vernazza S, Oddone F, Tirendi S, Bassi AM (2021) Risk factors for retinal ganglion cell distress in glaucoma and neuroprotective potential intervention. Int J Mol Sci 22(15):7994
Ramdas WD (2018) The relation between dietary intake and glaucoma: a systematic review. Acta Ophthalmol 96(6):550–556
Gospe SM III, Bhatti MT (2018) Orbital anatomy. Int Ophthalmol Clin 58(2):5–23
Hayek G, Mercier P, Fournier HD (2006) Anatomy of the orbit and its surgical approach. Adv Tech Stand Neurosurg 31:35–71
Pai SB, Nagarjun MN (2017) A neurosurgical perspective to approaches to the orbit: a cadaveric study. Neurol India 65(5):1094–1101
Chan EW, Li X, Tham YC, Liao J, Wong TY, Aung T, Cheng CY (2016) Glaucoma in Asia: regional prevalence variations and future projections. Br J Ophthalmol 100(1):78–85
Acknowledgements
Not applicable.
Funding
These authors have not disclosed any funding.
Author information
Authors and Affiliations
Contributions
SH analyzed the data. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
Not applicable.
Consent for publication
Not applicable.
Consent to participate
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huang, S., Liu, K., Su, Y. et al. Research progress of ferroptosis in glaucoma and optic nerve damage. Mol Cell Biochem 478, 721–727 (2023). https://doi.org/10.1007/s11010-022-04545-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-022-04545-7