Abstract
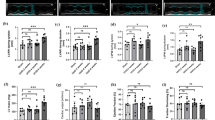
Progressive alteration of the extracellular matrix (ECM) is the characteristic of hypertensive nephropathy (HN). Both mesangial and endothelial cells have the ability to synthesize and degrade ECM components, including collagens through the activation of matrix metalloproteinases (MMPs) in stress conditions, such as in hypertension. On the other hand, hydrogen sulfide (H2S) has been shown to mitigate hypertensive renal matrix remodeling. Surprisingly, whether H2S ameliorates receptor-mediated (urokinase plasminogen activator receptor-associated protein, uPARAP/Endo180) collagen dysregulation in Ang-II hypertension is not clear. The purpose of this study was to determine whether Ang-II alters the expression of Endo180, tissue plasminogen activator (tPA), MMPs, and their tissue inhibitors (TIMPs) leading to the dysregulation of cellular collagen homeostasis and whether H2S mitigates the collagen turnover. Mouse mesangial cells (MCs) and glomerular endothelial cells (MGECs) were treated without or with Ang-II and H2S donor GYY (GYY4137) for 48 h. Cell lysates were analyzed by Western blot and RT-PCR, and cells were analyzed by immunocytochemistry. The results indicated that, while Ang-II differentially expressed MMP-13 and TIMP-1 in MCs and in MGECs, it predominantly decreased tPA, Endo 180, and increased plasminogen activator inhibitor-1 (PAI-1), MMP-14, and collagen IIIA and IV in both the cell types. Interestingly, H2S donor GYY treatment normalized the above changes in both the cell types. We conclude that Ang-II treatment causes ECM remodeling in MCs and MGECs through PAI-1/tPA/Endo180 and MMP/TIMP-dependent collagen remodeling, and H2S treatment mitigates remodeling, in part, by modulating these pathways.











Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Luke RG (1999) Hypertensive nephrosclerosis: pathogenesis and prevalence. Essential hypertension is an important cause of end-stage renal disease. Nephrol Dial Transpl 14:2271–2278
Hill GS (2008) Hypertensive nephrosclerosis. Curr Opin Nephrol Hypertens 17:266–270. https://doi.org/10.1097/MNH.0b013e3282f88a1f
Szijarto IA, Marko L, Filipovic MR, Miljkovic JL, Tabeling C, Tsvetkov D, Wang N, Rabelo LA, Witzenrath M, Diedrich A, Tank J, Akahoshi N, Kamata S, Ishii I, Gollasch M (2018) Cystathionine gamma-lyase produced hydrogen sulfide controls endothelial no bioavailability and blood pressure. Hypertension 71:1210–1217. https://doi.org/10.1161/Hypertensionaha.117.10562
Rose P, Moore PK, Zhu YZ (2017) H2S biosynthesis and catabolism: new insights from molecular studies. Cell Mol Life Sci 74:1391–1412. https://doi.org/10.1007/s00018-016-2406-8
Ishii I, Akahoshi N, Yu XN, Kobayashi Y, Namekata K, Komaki G, Kimura H (2004) Murine cystathionine gamma-lyase: complete cDNA and genomic sequences, promoter activity, tissue distribution and developmental expression. Biochemical Journal 381:113–123. https://doi.org/10.1042/Bj20040243
Kimura H (2011) Hydrogen sulfide: its production, release and functions. Amino Acids 41:113–121. https://doi.org/10.1007/s00726-010-0510-x
Sen U, Sathnur PB, Kundu S, Givvimani S, Coley DM, Mishra PK, Qipshidze N, Tyagi N, Metreveli N, Tyagi SC (2012) Increased endogenous H2S generation by CBS, CSE, and 3MST gene therapy improves ex vivo renovascular relaxation in hyperhomocysteinemia. Am J Physiol Cell Physiol 303:C41–C51. https://doi.org/10.1152/ajpcell.00398.2011
Genovese F, Manresa AA, Leeming DJ, Karsdal MA, Boor P (2014) The extracellular matrix in the kidney: a source of novel non-invasive biomarkers of kidney fibrosis? Fibrogenesis Tissue Repair 7:4. https://doi.org/10.1186/1755-1536-7-4
Gelse K, Poschl E, Aigner T (2003) Collagens—structure, function, and biosynthesis. Adv Drug Deliv Rev 55:1531–1546
Schuppan D, Ruehl M, Somasundaram R, Hahn EG (2001) Matrix as a modulator of hepatic fibrogenesis. Semin Liver Dis 21:351–372. https://doi.org/10.1055/s-2001-17556
Rowe RG, Weiss SJ (2009) Navigating ECM barriers at the invasive front: the cancer cell-stroma interface. Annu Rev Cell Dev Biol 25:567–595. https://doi.org/10.1146/annurev.cellbio.24.110707.175315
Bataller R, Brenner DA (2005) Liver fibrosis. J Clin Invest 115:209–218. https://doi.org/10.1172/JCI24282
Lauer-Fields JL, Juska D, Fields GB (2002) Matrix metalloproteinases and collagen catabolism. Biopolymers 66:19–32. https://doi.org/10.1002/bip.10201
Mott JD, Werb Z (2004) Regulation of matrix biology by matrix metalloproteinases. Curr Opin Cell Biol 16:558–564. https://doi.org/10.1016/j.ceb.2004.07.010
Arpino V, Brock M, Gill SE (2015) The role of TIMPs in regulation of extracellular matrix proteolysis. Matrix Biol 44–46:247–254. https://doi.org/10.1016/j.matbio.2015.03.005
Madsen DH, Jurgensen HJ, Ingvarsen S, Melander MC, Vainer B, Egerod KL, Hald A, Rono B, Madsen CA, Bugge TH, Engelholm LH, Behrendt N (2012) Endocytic collagen degradation: a novel mechanism involved in protection against liver fibrosis. J Pathol 227:94–105. https://doi.org/10.1002/path.3981
Ghosh AK, Vaughan DE (2012) PAI-1 in tissue fibrosis. J Cell Physiol 227:493–507. https://doi.org/10.1002/jcp.22783
Engelholm LH, Ingvarsen S, Jurgensen HJ, Hillig T, Madsen DH, Nielsen BS, Behrendt N (2009) The collagen receptor uPARAP/Endo180. Front Biosci (Landmark Ed) 14:2103–2114
John A, Kundu S, Pushpakumar S, Fordham M, Weber G, Mukhopadhyay M, Sen U (2017) GYY4137, a hydrogen sulfide donor modulates miR194-dependent collagen realignment in diabetic kidney. Sci Rep 7:10924. https://doi.org/10.1038/s41598-017-11256-3
Satoh M, Kashihara N, Yamasaki Y, Maruyama K, Okamoto K, Maeshima Y, Sugiyama H, Sugaya T, Murakami K, Makino H (2001) Renal interstitial fibrosis is reduced in angiotensin II type 1a receptor-deficient mice. J Am Soc Nephrol 12:317–325
Pushpakumar S, Ren L, Kundu S, Gamon A, Tyagi SC, Sen U (2017) Toll-like receptor 4 deficiency reduces oxidative stress and macrophage mediated inflammation in hypertensive kidney. Sci Rep 7:6349. https://doi.org/10.1038/s41598-017-06484-6
Curino AC, Engelholm LH, Yamada SS, Holmbeck K, Lund LR, Molinolo AA, Behrendt N, Nielsen BS, Bugge TH (2005) Intracellular collagen degradation mediated by uPARAP/Endo180 is a major pathway of extracellular matrix turnover during malignancy. J Cell Biol 169:977–985. https://doi.org/10.1083/jcb.200411153
Humphreys BD (2018) Mechanisms of renal fibrosis. Annu Rev Physiol 80:309–326. https://doi.org/10.1146/annurev-physiol-022516-034227
Wolf G (1998) Angiotensin II is involved in the progression of renal disease: importance of non-hemodynamic mechanisms. Nephrologie 19:451–456
Kim S, Iwao H (2000) Molecular and cellular mechanisms of angiotensin II-mediated cardiovascular and renal diseases. Pharmacol Rev 52:11–34
Weir MR, Dzau VJ (1999) The renin-angiotensin-aldosterone system: a specific target for hypertension management. Am J Hypertens 12:205S–213S
Kagami S, Kuhara T, Okada K, Kuroda Y, Border WA, Noble NA (1997) Dual effects of angiotensin II on the plasminogen/plasmin system in rat mesangial cells. Kidney Int 51:664–671
Pushpakumar S, Kundu S, Pryor T, Givvimani S, Lederer E, Tyagi SC, Sen U (2013) Angiotensin-II induced hypertension and renovascular remodelling in tissue inhibitor of metalloproteinase 2 knockout mice. J Hypertens 31:2270–2281. https://doi.org/10.1097/HJH.0b013e3283649b33. (discussion 2281)
Ichikawi I, Harris RC (1991) Angiotensin actions in the kidney: renewed insight into the old hormone. Kidney Int 40:583–596
Wolf G, Neilson EG (1993) Angiotensin II as a renal growth factor. J Am Soc Nephrol 3:1531–1540
Egido J (1996) Vasoactive hormones and renal sclerosis. Kidney Int 49:578–597
Ruiz-Ortega M, Egido J (1997) Angiotensin II modulates cell growth-related events and synthesis of matrix proteins in renal interstitial fibroblasts. Kidney Int 52:1497–1510
Mezzano SA, Ruiz-Ortega M, Egido J (2001) Angiotensin II and renal fibrosis. Hypertension 38:635–638
Frenay ARS, Snijder PM, Koning AM, Bachtler M, Pasch A, Kwakernaak AJ, van den Berg E, Bos EM, Hillebrands JL, Navis G, Leuvenink HGD, van Goor H (2014) Hydrogen sulfide attenuates angiotensin II-induced hypertension, proteinuria and renal damage. Nitric Oxide Biol Chem 39:S23–S24. https://doi.org/10.1016/j.niox.2014.03.075
Holwerda KM, Burke SD, Faas MM, Zsengeller Z, Stillman IE, Kang PM, van Goor H, McCurley A, Jaffe IZ, Karumanchi SA, Lely AT (2014) Hydrogen sulfide attenuates sFlt1-induced hypertension and renal damage by upregulating vascular endothelial growth factor. J Am Soc Nephrol 25:717–725. https://doi.org/10.1681/Asn.2013030291
Madhusudhan T, Kerlin BA, Isermann B (2016) The emerging role of coagulation proteases in kidney disease. Nat Rev Nephrol 12:94–109. https://doi.org/10.1038/nrneph.2015.177
Svenningsen P, Hinrichs GR, Zachar R, Ydegaard R, Jensen BL (2017) Physiology and pathophysiology of the plasminogen system in the kidney. Pflug Arch 469:1415–1423. https://doi.org/10.1007/s00424-017-2014-y
Eddy AA (2002) Plasminogen activator inhibitor-1 and the kidney. Am J Physiol Renal Physiol 283:F209–F220. https://doi.org/10.1152/ajprenal.00032.2002
Zhang G, Kernan KA, Collins SJ, Cai X, Lopez-Guisa JM, Degen JL, Shvil Y, Eddy AA (2007) Plasmin(ogen) promotes renal interstitial fibrosis by promoting epithelial-to-mesenchymal transition: role of plasmin-activated signals. J Am Soc Nephrol 18:846–859. https://doi.org/10.1681/ASN.2006080886
Olson JA Jr, Shiverick KT, Ogilvie S, Buhi WC, Raizada MK (1991) Angiotensin II induces secretion of plasminogen activator inhibitor 1 and a tissue metalloprotease inhibitor-related protein from rat brain astrocytes. Proc Natl Acad Sci USA 88:1928–1932
Zelezna B, Rydzewski B, Lu D, Olson JA, Shiverick KT, Tang W, Sumners C, Raizada MK (1992) Angiotensin-II induction of plasminogen activator inhibitor-1 gene expression in astroglial cells of normotensive and spontaneously hypertensive rat brain. Mol Endocrinol 6:2009–2017. https://doi.org/10.1210/mend.6.12.1491687
Vaughan DE, Lazos SA, Tong K (1995) Angiotensin II regulates the expression of plasminogen activator inhibitor-1 in cultured endothelial cells. A potential link between the renin-angiotensin system and thrombosis. J Clin Invest 95:995–1001. https://doi.org/10.1172/JCI117809
Zhao W, Chen SS, Chen Y, Ahokas RA, Sun Y (2008) Kidney fibrosis in hypertensive rats: role of oxidative stress. Am J Nephrol 28:548–554. https://doi.org/10.1159/000115289
Tharaux PL, Chatziantoniou C, Fakhouri F, Dussaule JC (2000) Angiotensin II activates collagen I gene through a mechanism involving the MAP/ER kinase pathway. Hypertension 36:330–336
Boffa JJ, Lu Y, Placier S, Stefanski A, Dussaule JC, Chatziantoniou C (2003) Regression of renal vascular and glomerular fibrosis: role of angiotensin II receptor antagonism and matrix metalloproteinases. J Am Soc Nephrol 14:1132–1144
Song K, Wang F, Li Q, Shi YB, Zheng HF, Peng HJ, Shen HY, Liu CF, Hu LF (2014) Hydrogen sulfide inhibits the renal fibrosis of obstructive nephropathy. Kidney Int 85:1318–1329. https://doi.org/10.1038/ki.2013.449
Melander MC, Jurgensen HJ, Madsen DH, Engelholm LH, Behrendt N (2015) The collagen receptor uPARAP/Endo180 in tissue degradation and cancer. Int J Oncol 47:1177–1188. https://doi.org/10.3892/ijo.2015.3120
Behrendt N (2004) The urokinase receptor (uPAR) and the uPAR-associated protein (uPARAP/Endo180): membrane proteins engaged in matrix turnover during tissue remodeling. Biol Chem 385:103–136. https://doi.org/10.1515/BC.2004.031
Zhang G, Eddy AA (2008) Urokinase and its receptors in chronic kidney disease. Front Biosci 13:5462–5478
Messaritou G, East L, Roghi C, Isacke CM, Yarwood H (2009) Membrane type-1 matrix metalloproteinase activity is regulated by the endocytic collagen receptor Endo180. J Cell Sci 122:4042–4048
Cheng Z, Limbu MH, Wang Z, Liu J, Liu L, Zhang X, Chen P, Liu B (2017) MMP-2 and 9 in chronic kidney disease. Int J Mol Sci. https://doi.org/10.3390/ijms18040776
Itoh Y, Seiki M (2006) MT1-MMP: a potent modifier of pericellular microenvironment. J Cell Physiol 206:1–8. https://doi.org/10.1002/jcp.20431
Engelholm LH, Nielsen BS, Netzel-Arnett S, Solberg H, Chen XD, Lopez Garcia JM, Lopez-Otin C, Young MF, Birkedal-Hansen H, Dano K, Lund LR, Behrendt N, Bugge TH (2001) The urokinase plasminogen activator receptor-associated protein/endo180 is coexpressed with its interaction partners urokinase plasminogen activator receptor and matrix metalloprotease-13 during osteogenesis. Lab Invest 81:1403–1414. https://doi.org/10.1038/labinvest.3780354
Carmeliet P, Moons L, Lijnen R, Baes M, Lemaitre V, Tipping P, Drew A, Eeckhout Y, Shapiro S, Lupu F, Collen D (1997) Urokinase-generated plasmin activates matrix metalloproteinases during aneurysm formation. Nat Genet 17:439–444. https://doi.org/10.1038/ng1297-439
Pushpakumar SB, Kundu S, Metreveli N, Sen U (2013) Folic acid mitigates angiotensin-II-induced blood pressure and renal remodeling. PLoS ONE 8:e83813. https://doi.org/10.1371/journal.pone.0083813
Zakiyanov O, Kalousova M, Zima T, Tesar V (2019) Matrix metalloproteinases In renal diseases: a critical appraisal. Kidney Blood Press Res 44:298–330. https://doi.org/10.1159/000499876
Ren J, Zhang J, Rudemiller NP, Griffiths R, Wen Y, Lu X, Privratsky JR, Gunn MD, Crowley SD (2019) Twist1 in Infiltrating macrophages attenuates kidney fibrosis via matrix metallopeptidase 13-mediated matrix degradation. J Am Soc Nephrol 30:1674–1685. https://doi.org/10.1681/ASN.2018121253
Cai G, Zhang X, Hong Q, Shao F, Shang X, Fu B, Feng Z, Lin H, Wang J, Shi S, Yin Z, Chen X (2008) Tissue inhibitor of metalloproteinase-1 exacerbated renal interstitial fibrosis through enhancing inflammation. Nephrol Dial Transpl 23:1861–1875. https://doi.org/10.1093/ndt/gfm666
Acknowledgements
We thank Naira Metreveli for her technical assistance related to cell culture experiments and the acquisition of confocal images. This work was supported in part by National Institutes of Health Grants, DK104653 and DK116591 (to U.S.) and American Heart Association Scientist Development Grant, 15SDG25840013 (to S.P.)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest, financial or otherwise.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Majumder, S., Amin, M., Pushpakumar, S. et al. Collagen receptor- and metalloproteinase-dependent hypertensive stress response in mesangial and glomerular endothelial cells. Mol Cell Biochem 466, 1–15 (2020). https://doi.org/10.1007/s11010-019-03680-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-019-03680-y