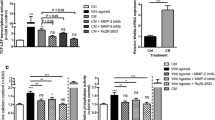
Abstract
Arterial calcification is common in vascular diseases and involves conversion of vascular smooth muscle cells (VSMCs) to an osteoblast phenotype. Clinical studies suggest that the development of atherosclerosis can be promoted by homocysteine (HCY), but the mechanisms remain unclear. Here, we determined whether increases in HCY levels lead to an increase in VSMC calcification and differentiation, and examined the role of an extracellular matrix remodeler, matrix metalloproteinase-2 (MMP-2). Rat VSMCs were exposed to calcification medium in the absence or presence of HCY (10, 100 or 200 μmol/L) or an MMP-2 inhibitor (10−6 or 10−5 mol/L). MTT assays were performed to determine the cytotoxicity of the MMP-2 inhibitor in calcification medium containing 200 μmol/L HCY. Calcification was assessed by measurements of calcium deposition and alkaline phosphatase (ALP) activity as well as von Kossa staining. Expression of osteocalcin, bone morphogenetic protein (BMP)-2, and osteopontin, and MMP-2 was determined by immunoblotting. Calcification medium induced osteogenic differentiation of VSMCs. HCY promoted calcification, increased osteocalcin and BMP-2 expression, and decreased expression of osteopontin. MMP-2 expression was increased by HCY in a dose-dependent manner in VSMCs exposed to both control and calcification medium. The MMP-2 inhibitor decreased the calcium content and ALP activity, and attenuated the osteoblastic phenotype of VSMCs. Vascular calcification and osteogenic differentiation of VSMCs were positively regulated by HCY through increased/restored MMP-2 expression, increased expression of calcification proteins, and decreased anti-calcification protein levels. In summary, MMP-2 inhibition may be a protective strategy against VSMC calcification.







Similar content being viewed by others
References
Takasu J, Budoff MJ, Katz R, Rivera JJ, O’Brien KD, Shavelle DM, Probstfield JL, O’Leary D, Nasir K (2010) Relationship between common carotid intima-media thickness and thoracic aortic calcification: the Multi-Ethnic Study of Atherosclerosis. Atherosclerosis 209:142–146
Huybers S, Bindels RJ (2007) Vascular calcification in chronic kidney disease: new developments in drug therapy. Kidney Int 72:663–665
Ishiyama M, Suzuki E, Katsuda J, Murase H, Tajima Y, Horikawa Y, Goto S, Fujita T, Takeda J (2009) Associations of coronary artery calcification and carotid intima-media thickness with plasma concentrations of vascular calcification inhibitors in type 2 diabetic patients. Diabetes Res Clin Pract 85:189–196
Aboyans V, Ho E, Denenberg JO, Ho LA, Natarajan L, Criqui MH (2008) The association between elevated ankle systolic pressures and peripheral occlusive arterial disease in diabetic and nondiabetic subjects. J Vasc Surg 48:1197–1203
Rosito GA, Massaro JM, Hoffmann U, Ruberg FL, Mahabadi AA, Vasan RS, O’Donnell CJ, Fox CS (2008) Pericardial fat, visceral abdominal fat, cardiovascular disease risk factors, and vascular calcification in a community-based sample: the Framingham Heart Study. Circulation 117:605–613
Dao HH, Essalihi R, Bouvet C, Moreau P (2005) Evolution and modulation of age-related medial elastocalcinosis: impact on large artery stiffness and isolated systolic hypertension. Cardiovasc Res 66:307–317
Kanemaru K, Seya K, Miki I, Motomura S, Furukawa K (2008) Calcification of aortic smooth muscle cells isolated from spontaneously hypertensive rats. J Pharmacol Sci 106:280–286
Doherty TM, Uzui H, Fitzpatrick LA, Tripathi PV, Dunstan CR, Asotra K, Rajavashisth TB (2002) Rationale for the role of osteoclast-like cells in arterial calcification. FASEB J 16:577–582
Demer LL, Tintut Y (1999) Osteopontin between a rock and a hard plaque. Circ Res 84:250–252
Giachelli CM (2001) Ectopic calcification: new concepts in cellular regulation. Z Kardiol 90(Suppl 3):31–37
Parhami F, Morrow AD, Balucan J, Leitinger N, Watson AD, Tintut Y, Berliner JA, Demer LL (1997) Lipid oxidation products have opposite effects on calcifying vascular cell and bone cell differentiation. A possible explanation for the paradox of arterial calcification in osteoporotic patients. Arterioscler Thromb Vasc Biol 17:680–687
Shioi A, Katagi M, Okuno Y, Mori K, Jono S, Koyama H, Nishizawa Y (2002) Induction of bone-type alkaline phosphatase in human vascular smooth muscle cells: roles of tumor necrosis factor-alpha and oncostatin M derived from macrophages. Circ Res 91:9–16
Toborek M, Kopieczna-Grzebieniak E, Drozdz M, Wieczorek M (1995) Increased lipid peroxidation as a mechanism of methionine-induced atherosclerosis in rabbits. Atherosclerosis 115:217–224
Hirose N, Arai Y, Ishii T, Tushima M, Li J (2001) Association of mild hyperhomocysteinemia with aortic calcification in hypercholesterolemic patients. J Atheroscler Thromb 8:91–94
Castro R, Rivera I, Blom HJ, Jakobs C, Tavares de Almeida I (2006) Homocysteine metabolism, hyperhomocysteinaemia and vascular disease: an overview. J Inherit Metab Dis 29:3–20
McCully KS (1969) Vascular pathology of homocysteinemia: implications for the pathogenesis of arteriosclerosis. Am J Pathol 56:111–128
Price PA, Williamson MK, Lothringer JW (1981) Origin of the vitamin K-dependent bone protein found in plasma and its clearance by kidney and bone. J Biol Chem 256:12760–12766
Fallest-Strobl PC, Koch DD, Stein JH, McBride PE (1997) Homocysteine: a new risk factor for atherosclerosis. Am Fam Physician 56(1607–12):1615–1616
Freedman BI, Bowden DW, Ziegler JT, Langefeld CD, Lehtinen AB, Rudock ME, Lenchik L, Hruska KA, Register TC, Carr JJ (2009) Bone morphogenetic protein 7 (BMP7) gene polymorphisms are associated with inverse relationships between vascular calcification and BMD: the Diabetes Heart Study. J Bone Miner Res 24:1719–1727
Tyson KL, Reynolds JL, McNair R, Zhang Q, Weissberg PL, Shanahan CM (2003) Osteo/chondrocytic transcription factors and their target genes exhibit distinct patterns of expression in human arterial calcification. Arterioscler Thromb Vasc Biol 23:489–494
Price PA (1985) Vitamin K-dependent formation of bone Gla protein (osteocalcin) and its function. Vitam Horm 42:65–108
Brown JP, Delmas PD, Malaval L, Edouard C, Chapuy MC, Meunier PJ (1984) Serum bone Gla-protein: a specific marker for bone formation in postmenopausal osteoporosis. Lancet 1:1091–1093
Malluche HH, Faugere MC, Fanti P, Price PA (1984) Plasma levels of bone Gla-protein reflect bone formation in patients on chronic maintenance dialysis. Kidney Int 26:869–874
Minisola S, Rosso R, Romagnoli E, D'Erasmo E, Manfredi G, Damiani C, De Antoni F, Mazzuoli G (1997) Serum osteocalcin and bone mineral density at various skeletal sites: a study performed with three different assays. J Lab Clin Med 129:422–429
Zarjou A, Jeney V, Arosio P, Poli M, Antal-Szalmas P, Agarwal A, Balla G, Balla J (2009) Ferritin prevents calcification and osteoblastic differentiation of vascular smooth muscle cells. J Am Soc Nephrol 20:1254–1263
Scatena M, Liaw L, Giachelli CM (2007) Osteopontin: a multifunctional molecule regulating chronic inflammation and vascular disease. Arterioscler Thromb Vasc Biol 27:2302–2309
Cho HJ, Cho HJ, Kim HS (2009) Osteopontin: a multifunctional protein at the crossroads of inflammation, atherosclerosis, and vascular calcification. Curr Atheroscler Rep 11:206–213
Speer MY, Yang HY, Brabb T, Leaf E, Look A, Lin WL, Frutkin A, Dichek D, Giachelli CM (2009) Smooth muscle cells give rise to osteochondrogenic precursors and chondrocytes in calcifying arteries. Circ Res 104:733–741
Galis ZS, Khatri JJ (2002) Matrix metalloproteinases in vascular remodeling and atherogenesis: the good, the bad, and the ugly. Circ Res 90:251–262
Halpert I, Sires UI, Roby JD, Potter-Perigo S, Wight TN, Shapiro SD, Welgus HG, Wickline SA, Parks WC (1996) Matrilysin is expressed by lipid-laden macrophages at sites of potential rupture in atherosclerotic lesions and localizes to areas of versican deposition, a proteoglycan substrate for the enzyme. Proc Natl Acad Sci USA 93:9748–9753
Nikkari ST, Geary RL, Hatsukami T, Ferguson M, Forough R, Alpers CE, Clowes AW (1996) Expression of collagen, interstitial collagenase, and tissue inhibitor of metalloproteinases-1 in restenosis after carotid endarterectomy. Am J Pathol 148:777–783
Galis ZS, Sukhova GK, Lark MW, Libby P (1994) Increased expression of matrix metalloproteinases and matrix degrading activity in vulnerable regions of human atherosclerotic plaques. J Clin Invest 94:2493–2503
Allaire E, Forough R, Clowes M, Starcher B, Clowes AW (1998) Local overexpression of TIMP-1 prevents aortic aneurysm degeneration and rupture in a rat model. J Clin Invest 102:1413–1420
Petrinec D, Liao S, Holmes DR, Reilly JM, Parks WC, Thompson RW (1996) Doxycycline inhibition of aneurysmal degeneration in an elastase-induced rat model of abdominal aortic aneurysm: preservation of aortic elastin associated with suppressed production of 92 kD gelatinase. J Vasc Surg 23:336–346
Mason DP, Kenagy RD, Hasenstab D, Bowen-Pope DF, Seifert RA, Coats S, Hawkins SM, Clowes AW (1999) Matrix metalloproteinase-9 overexpression enhances vascular smooth muscle cell migration and alters remodeling in the injured rat carotid artery. Circ Res 85:1179–1185
Bendeck MP, Conte M, Zhang M, Nili N, Strauss BH, Farwell SM (2002) Doxycycline modulates smooth muscle cell growth, migration, and matrix remodeling after arterial injury. Am J Pathol 160:1089–1095
Basalyga DM, Simionescu DT, Xiong W, Baxter BT, Starcher BC, Vyavahare NR (2004) Elastin degradation and calcification in an abdominal aorta injury model: role of matrix metalloproteinases. Circulation 110:3480–3487
Campbell JH, Campbell GR (1993) Culture techniques and their applications to studies of vascular smooth muscle. Clin Sci (Lond) 85:501–513
Mune S, Shibata M, Hatamura I, Saji F, Okada T, Maeda Y, Sakaguchi T, Negi S, Shigematsu T (2009) Mechanism of phosphate-induced calcification in rat aortic tissue culture: possible involvement of Pit-1 and apoptosis. Clin Exp Nephrol 13:571–577. doi:10.1007/s10157-009-0208-0
Rungby J, Kassem M, Eriksen EF, Danscher G (1993) The von Kossa reaction for calcium deposits: silver lactate staining increases sensitivity and reduces background. Histochem J 25:446–451
Touyz RM, He Y, Montezano AC, Yao G, Chubanov V, Gudermann T, Callera GE (2006) Differential regulation of transient receptor potential melastatin 6 and 7 cation channels by ANG II in vascular smooth muscle cells from spontaneously hypertensive rats. Am J Physiol Regul Integr Comp Physiol 290:R73–R78
Ketteler M, Biggar PH (2009) Review article: getting the balance right: assessing causes and extent of vascular calcification in chronic kidney disease. Nephrology (Carlton) 14:389–394
Navaneethan SD, Palmer SC, Craig JC, Elder GJ, Strippoli GF (2009) Benefits and harms of phosphate binders in CKD: a systematic review of randomized controlled trials. Am J Kidney Dis 54:619–637
O’Neill WC (2009) Mineral complexes and vascular calcification. Kidney Int 76:915 (author 915-6)
Li X, Giachelli CM (2007) Sodium-dependent phosphate cotransporters and vascular calcification. Curr Opin Nephrol Hypertens 16:325–328
Noonan W, Koch K, Nakane M, Ma J, Dixon D, Bolin A, Reinhart G (2008) Differential effects of vitamin D receptor activators on aortic calcification and pulse wave velocity in uraemic rats. Nephrol Dial Transplant 23:3824–3830
Togawa T, Sengupta S, Chen H, Robinson K, Nonevski I, Majors AK, Jacobsen DW (2000) Mechanisms for the formation of protein-bound homocysteine in human plasma. Biochem Biophys Res Commun 277:668–674
Lee JS, Basalyga DM, Simionescu A, Isenburg JC, Simionescu DT, Vyavahare NR (2006) Elastin calcification in the rat subdermal model is accompanied by up-regulation of degradative and osteogenic cellular responses. Am J Pathol 168:490–498
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, T., Lin, J., Ju, T. et al. Vascular smooth muscle cell differentiation to an osteogenic phenotype involves matrix metalloproteinase-2 modulation by homocysteine. Mol Cell Biochem 406, 139–149 (2015). https://doi.org/10.1007/s11010-015-2432-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-015-2432-0