Abstract
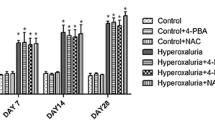
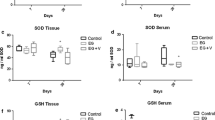
The purpose of the present study was to evaluate the nephro-protective potential of N-acetylcysteine against hyperoxaluria-induced renal mitochondrial dysfunction in rats. Nine days dosing of 0.4 % ethylene glycol +1 % ammonium chloride, developed hyperoxaluria in male wistar rats which resulted in renal injury and dysfunction as supported by increased level of urinary lactate dehydrogenase, calcium, and decreased creatinine clearance. Mitochondrial oxidative strain in hyperoxaluric animals was evident by decreased levels of superoxide dismutase, glutathione peroxidase, glutathione reductase, reduced glutathione, and an increased lipid peroxidation. Declined activities of respiratory chain enzymes and tricarboxylic acid cycle enzymes showed mitochondrial dysfunction in hyperoxaluric animals. N-acetylcysteine (50 mg/kg, i.p.), by virtue of its –SH reviving power, was able to increase the glutathione levels and thus decrease the oxidative stress in renal mitochondria. Hence, mitochondrial damage is, evidently, an essential event in ethylene glycol-induced hyperoxaluria and N-acetylcysteine presented itself as a safe and effective remedy in combating nephrolithiasis.





Similar content being viewed by others
Abbreviations
- COM:
-
Calcium oxalate monohydrate
- DTNB:
-
5,5-Dithiobis-2-nitrobenzoic acid
- EG:
-
Ethylene glycol
- ETC:
-
Electron transport chain
- GR:
-
Glutathione reductase
- GSH:
-
Reduced glutathione
- GPx:
-
Glutathione peroxidase
- i.p.:
-
Intraperitoneal
- LDH:
-
Lactate dehydrogenase
- LPO:
-
Lipid peroxidation
- NP-SH:
-
Non-protein thiols
- MDA:
-
Malondialdehyde
- Mn-SOD:
-
Manganese superoxide dismutase
- NADH:
-
Nicotinamide adenine dinucleotide
- NAC:
-
N-acetylcysteine
- ROS:
-
Reactive oxygen species
- T-SH:
-
Total thiols
References
Brenna I, Dogliotti E, Terranegra A, Raspini B, Soldati L (2013) Nephrolithiasis: nutrition as cause or therapeutic tool. J Transl Med 11:178
López M, Hoppe B (2010) History, epidemiology and regional diversities of urolithiasis. Pediatr Nephrol 25:49–59
Hou J (2013) The role of claudin in hypercalciuric nephrolithiasis. Curr Urol Rep 14:5–12
Robijn S, Hoppe B, Vervaet BA, D’Haese PC, Verhulst A (2011) Hyperoxaluria: a gut-kidney axis & quest. Kidney Int 80:1146–1158
Scheid C, Koul H, Hill WA, Luber-Narod J, Kennington L, Honeyman T, Jonassen J, Menon M (1996) Oxalate toxicity in LLC-PK1 cells: role of free radicals. Kidney Int 49:413–419
Thamilselvan S, Khan SR, Menon M (2003) Oxalate and calcium oxalate mediated free radical toxicity in renal epithelial cells: effect of antioxidants. Urol Res 31:3–9
Jonassen J, Kohjimoto Y, Scheid C, Schmidt M (2005) Oxalate toxicity in renal cells. Urol Res 33:329–339
Cao L-C, Honeyman TW, Cooney R, Kennington L, Scheid CR, Jonassen JA (2004) Mitochondrial dysfunction is a primary event in renal cell oxalate toxicity. Kidney Int 66:1890–1900
Selvam R, Muthukumar A (1997) Effect of depletion of reduced glutathione and its supplementation by glutathione monoester on renal oxalate retention in hyperoxaluric rats. J Nutr Biochem 8:445–450
Khan SR (2005) Hyperoxaluria-induced oxidative stress and antioxidants for renal protection. Urol Res 33:349–357
McMartin KE, Wallace KB (2005) Calcium oxalate monohydrate, a metabolite of ethylene glycol, is toxic for rat renal mitochondrial function. Toxicol Sci 84:195–200
Li X, Fang P, Mai J, Choi ET, Wang H, Yang X (2013) Targeting mitochondrial reactive oxygen species as novel therapy for inflammatory diseases and cancers. J Hematol Oncol 6:19
Khand F, Gordge MP, Robertson W, Noronha-Dutra A, Hothersall J (2002) Mitochondrial superoxide production during oxalate-mediated oxidative stress in renal epithelial cells. Free Radic Biol Med 32:1339–1350
Raza H, John A, Shafarin J (2014) NAC attenuates LPS-induced toxicity in aspirin-sensitized mouse macrophages via suppression of oxidative stress and mitochondrial dysfunction. PLoS ONE 9:e103379
Bijarnia RK, Kaur T, Aggarwal K, Singla S, Tandon C (2008) Modulatory effects of N-acetylcysteine on hyperoxaluric manifestations in rat kidney. Food Chem Toxicol 46:2274–2278
Conesa EL, Valero F, Nadal JC, Fenoy FJ, Lopez B, Arregui B, Salom MG (2001) N-acetyl-l-cysteine improves renal medullary hypoperfusion in acute renal failure. Am J Physiol Regul Integr Comp Physiol 281:R730–R737
Mazzon E, Britti D, De Sarro A, Caputi AP, Cuzzocrea S (2001) Effect of N-acetylcysteine on gentamicin-mediated nephropathy in rats. Eur J Pharmacol 424:75–83
Nitescu N, Ricksten S-E, Marcussen N, Br Haraldsson, Nilsson U, Basu S, Guron G (2006) N-acetylcysteine attenuates kidney injury in rats subjected to renal ischaemia-reperfusion. Nephrol Dial Transplant 21:1240–1247
Park E, Yu KH, Kim DK, Kim S, Sapkota K, Kim S-J, Kim CS, Chun HS (2014) Protective effects of N-acetylcysteine against monosodium glutamate-induced astrocytic cell death. Food Chem Toxicol 67:1–9
Hodgkinson A, Williams A (1972) An improved colorimetric procedure for urine oxalate. Clin Chim Acta 36:127–132
Wright PJ, Plummer DT (1974) The use of urinary enzyme measurements to detect renal damage caused by nephrotoxic compounds. Biochem Pharmacol 23:65–73
Sottocasa GL, Kuylenstierna B, Ernster L, Bergstrand A (1967) An electron-transport system associated with the outer membrane of liver mitochondria a biochemical and morphological study. J Cell Biol 32:415–438
Kirby DM, Thorburn DR, Turnbull DM, Taylor RW (2007) Biochemical assays of respiratory chain complex activity. Methods Cell Biol 80:93–119
Buege JA, Aust SD (1978) Microsomal lipid peroxidation. Methods Enzymol 52:302–310
Kono Y (1978) Generation of superoxide radical during autoxidation of hydroxylamine and an assay for superoxide dismutase. Arch Biochem Biophys 186:189–195
Flohe L, Gunzler WA (1984) Assays of glutathione peroxidase. Methods in enzymology, vol 105. Academic Press, Orlando, pp 114–121
Carlberg I, Mannervik B (1985) Glutathione reductase. Methods Enzymol 113:484–490
Wang H, Joseph JA (1999) Quantifying cellular oxidative stress by dichlorofluorescin assay using microplate reader. Free Radic Biol Med 27:612–616
Sedlak J (1982) Changes of glutathione and protein bound SH-groups concentration in rat adrenals under acute and repeated stress. Endocrinol Exp 16:103–109
Roberts JC, Francetic DJ (1993) The importance of sample preparation and storage in glutathione analysis. Anal Biochem 211:183–187
King TE, Howard RL (1967) Preparations and properties of soluble NADH dehydrogenases from cardiac muscle. Methods Enzymol 10:275–294
King TE, Ohnishi T, Winter DB and Wu J (1976) Biochemical and EPR probes for structure-function studies of iron sulfur centres of succinate dehydrogenase. In: Iron and Copper Proteins. Springer, pp 182-227
Liu Y, Peterson DA, Kimura H, Schubert D (1997) Mechanism of cellular 3-(4,5-dimethyl thiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT) reduction. J Neurochem 69:581–593
Fansler B, Lowenstein J (1969) Aconitase from pig heart: [EC 4.2. 13 Citrate (isocitrate) hydro-lyase]. Methods Enzymol 13:26–30
Bergmeyer H, Bernt E (1974) Isocitrate dehydrogenase. Methods of enzymatic analysis. Verlag Chemie, Weinheim/Bergstr, pp 479–480
Robinson J, Inman L, Sumegi B, Srere PA (1987) Further characterization of the Krebs tricarboxylic acid cycle metabolon. J Biol Chem 262:1786–1790
Huang HS, Ma MC, Chen J, Chen CF (2002) Changes in the oxidant-antioxidant balance in the kidney of rats with nephrolithiasis induced by ethylene glycol. J Urol 167:2584–2593
Khan SR (2006) Renal tubular damage/dysfunction: key to the formation of kidney stones. Urol Res 34:86–91
Schepers MS, van Ballegooijen ES, Bangma CH, Verkoelen CF (2005) Oxalate is toxic to renal tubular cells only at supraphysiologic concentrations. Kidney Int 68:1660–1669
Lenin M, Latha LM, Nagaraj M, Varalakshmi P (2002) Mitigation of free radical toxicity in hyperoxaluric condition by a novel derivative eicosapentaenoate-lipoate. Hum Exp Toxicol 21:153–158
Aggarwal D, Sharma M, Singla SK (2013) The role of natural antioxidants as potential therapeutic agent in Nephrolithiasis. Asian J Pharm Clin Res 6:48–53
Huang H, Chen J, Chen C, Ma M (2006) Vitamin E attenuates crystal formation in rat kidneys: roles of renal tubular cell death and crystallization inhibitors. Kidney Int 70:699–710
Thamilselvan S, Menon M (2005) Vitamin E therapy prevents hyperoxaluria-induced calcium oxalate crystal deposition in the kidney by improving renal tissue antioxidant status. BJU Int 96:117–126
Aggarwal D, Kaushal R, Kaur T, Bijarnia RK, Puri S, Singla SK (2014) The most potent antilithiatic agent ameliorating renal dysfunction and oxidative stress from Bergenia ligulata rhizome. J Ethnopharmacol 158:85–93
Veena CK, Josephine A, Preetha SP, Rajesh NG, Varalakshmi P (2008) Mitochondrial dysfunction in an animal model of hyperoxaluria: a prophylactic approach with fucoidan. Eur J Pharmacol 579:330–336
Ashok P, Koti BC, Vishwanathswamy A (2010) Antiurolithiatic and antioxidant activity of Mimusops elengi on ethylene glycol-induced urolithiasis in rats. Indian J Pharmacol 42:380
Ghaeni FA, Amin B, Hariri AT, Meybodi NT, Hosseinzadeh H (2014) Antilithiatic effects of crocin on ethylene glycol-induced lithiasis in rats. Urolithiasis 42:549–558
Kaur T, Bijarnia RK, Singla SK, Tandon C (2009) In vivo efficacy of Trachyspermum ammi anticalcifying protein in urolithiatic rat model. J Ethnopharmacol 126:459–462
Ueno T, Yamada M, Sugita Y, Ogawa T (2011) N-acetyl cysteine protects TMJ chondrocytes from oxidative stress. J Dent Res 90:353–359
Pigeolet E, Corbisier P, Ae Houbion, Lambert D, Michiels C, Raes M, Zachary M-D, Remacle J (1990) Glutathione peroxidase, superoxide dismutase, and catalase inactivation by peroxides and oxygen derived free radicals. Mech Ageing Dev 51:283–297
Veena CK, Josephine A, Preetha SP, Varalakshmi P (2007) Beneficial role of sulfated polysaccharides from edible seaweed Fucus vesiculosus in experimental hyperoxaluria. Food Chem 100:1552–1559
Das KC, Lewis Molock Y, White CW (1997) Elevation of manganese superoxide dismutase gene expression by thioredoxin. Am J Respir Cell Mol Biol 17:713–726
Tucci P, Cione E, Perri M, Genchi G (2008) All-trans-retinoic acid induces apoptosis in Leydig cells via activation of the mitochondrial death pathway and antioxidant enzyme regulation. J Bioenerg Biomembr 40:315–323
Acknowledgments
The financial assistance provided by the University Grants Commission, New Delhi is gratefully acknowledged.
Conflict of interest
The authors state no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sharma, M., Kaur, T. & Singla, S.K. Protective effects of N-acetylcysteine against hyperoxaluria induced mitochondrial dysfunction in male wistar rats. Mol Cell Biochem 405, 105–114 (2015). https://doi.org/10.1007/s11010-015-2402-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-015-2402-6