Abstract
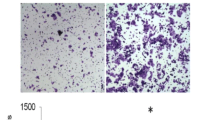
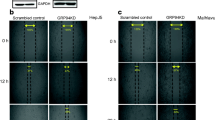
The major characteristics of pancreatic cancer are its excessive local invasion and early systemic dissemination. The glucose-regulated protein is over-expressed in many human cancers including pancreatic cancer and correlated with invasion and metastasis in many cancers. To investigate the effect of Grp78 on the invasion of pancreatic cancer, we used western blot and Transwell assay. We found Grp78 is expressed at lower levels in capan-2 and higher expressed in MiaPaCa-2 cells, and Grp78 expression levels were correlated with the invasion potentials of tumor cells. Then,we increased the expression of Grp78 in capan-2 cells and decreased the expression of Grp78 in MiaPaCa-2 cells. We found that over-expression of Grp78 caused significant increase in the expression of TIMP-1, TIMP-2, MMP-14, MMP-2, and MMP-9 in Capan-2 cells. Consistently, knockdown of Grp78 decreased the expression of them in MiaPaCa-2 cells. Gelatin zymography showed Grp78 over-expression stimulated the activities of MMP-2 and MMP-9, while GRP78 knockdown reduced the activities of MMP-2 and MMP-9. Cytoskeleton staining showed that knockdown of Grp78 caused a marked increase in cytoskeleton F-actin stress fibers in MiaPaCa-2 cells. Consistently, GRP78 knockdown hyperactivated RhoA and inhibited significantly Rac activity. Grp78 over-expression decreases the RhoA and stimulated Rac activity. We also found that Grp78 modulated FAK and JNK signaling pathways. Over-expression of GRP78 in Capan-2 activated FAK and JNK. Finally, we demonstrated that knockdown of FAK by shRNA in combination with blockade of JNK signaling pathway with SP600125 completely inhibited GRP78-induced cancer cell invasion. GRP78 is involved in the regulation of pancreatic cancer invasion. FAK and JNK are the key downstream effectors of GRP78.






Similar content being viewed by others
References
American Cancer Society. Cancer facts & figures 2010: see page 4 for incidence estimates, and page 19 for survival percentages”
Keleg Shereen, Büchler Peter, Ludwig Roman et al (2003) Invasion and metastasis in pancreatic cancer. Mol Cancer 2:14
Lee AS (2001) The glucose-regulated proteins: stress induction and clinical applications. Trends Biochem Sci 26:504–510
Thornton M, Aslam MA et al (2013) The unfolded protein response regulator GRP78 is a novel predictive biomarker in colorectal cancer. Int J Cancer 133:1408–1418
Wu CT, Wang WC, Chen MF et al (2013) Glucose-regulated protein 78 mediates hormone-independent prostate cancer progression and metastasis through maspin and COX-2 expression. Tumour Biol 35:195–204
Zhu X, Zhang J, Fan W et al (2013) The rs391957 variant cis-regulating oncogene GRP78 expression contributes to the risk of hepatocellular carcinoma. Carcinogenesis 34:1273–1280
Lee S, Terry D, Hurst DR et al (2011) Protein signatures in human MDA-MB-231 breast cancer cells indicating a more invasive phenotype following knockdown of human Endometase/Matrilysin-2 by siRNA. J Cancer 2:165–176
Zhang J, Jiang Y, Jia Z et al (2006) Association of elevated GRP78 expression with increased lymph node metastasis and poor prognosis in patients with gastric cancer. Clin Exp Metastasis 23:401–410
Gonzalez-Gronow M, Cuchacovich M, Llanos C et al (2006) Prostate cancer cell proliferation in vitro is modulated by antibodies against glucose-regulated protein 78 isolated from patient serum. Cancer Res 66:11424–11431
Cook KL, Shajahan AN, Wärri A et al (2012) Glucose-regulated protein 78 controls cross-talk between apoptosis and autophagy to determine antiestrogen responsiveness. Cancer Res 72:3337–3349
Su R, Li Z, Li H et al (2010) Grp78 promotes the invasion of hepatocellular carcinoma. BMC Cancer 10:20
Chen CC, Sureshbabul M, Chen HW et al (2013) Curcumin suppresses metastasis via Sp-1, FAK inhibition, and E-cadherin upregulation in colorectal cancer. Evid Based Complement Alternat Med 2013:541695–541712
Vicente-Manzanares M, Choi CK, Horwitz AR (2009) Integrins in cell migration. J Cell Sci 122:199–206
Morimura S, Takahashi K (2011) Rac1 and stathmin but not EB1 are required for invasion of breast cancer cells in response to IGF-I. Int J Cell Biol 2011:615912
Li Z, Zhang L, Zhao Y et al (2013) Cell-surface GRP78 facilitates colorectal cancer cell migration and invasion. Int J Biochem Cell Biol 45:987–994
Li H, Song H, Luo J et al (2012) Knockdown of glucose-regulated protein 78 decreases the invasion, metalloproteinase expression and ECM degradation in hepatocellular carcinoma cells. J Exp Clin Cancer Res 31:39
Kang J, Zhao G, Lin T, Tang S, Xu G, Hu S et al (2013) A peptide derived from phage display library exhibits anti-tumor activity by targeting GRP78 in gastric cancer multidrug resistance cells. Cancer Lett 339:247–259
Tsai HY, Yang YF, Wu AT et al (2013) Endoplasmic reticulum ribosome-binding protein 1 (RRBP1) overexpression is frequently found in lung cancer patients and alleviates intracellular stress-induced apoptosis through the enhancement of GRP78. Oncogene 32(41):4921–4931
Grkovic S, O’Reilly VC, Han S et al (2013) IGFBP-3 binds GRP78, stimulates autophagy and promotes the survival of breast cancer cells exposed to adverse microenvironments. Oncogene 32:2412–2420
Cáceres LC, Bonacci GR, Sánchez MC et al (2010) Chiabrando1: activated α2 macroglobulin induces matrix metalloproteinase 9 expression by low-density lipoprotein receptor-related protein 1 through MAPK-ERK1/2 and NF-κB activation in macrophage-derived cell lines. J Cell Biochem 111:607–617
Khan MM, Simizu S, Suzuki T et al (2012) Protein disulfide isomerase-mediated disulfide bonds regulate the gelatinolytic activity and secretion of matrix metalloproteinase-9. Exp Cell Res 318:904–914
Chen L, Sun B, Zhang S et al (2009) Influence of microenvironments on microcirculation patterns and tumor invasion-related protein expression in melanoma. Oncol Rep 21:917–923
Khyrul WA, LaLonde DP, Brown MC et al (2004) The integrin-linked kinase regulates cell morphology and motility in a rho-associated kinase-dependent manner. J Biol Chem 279:54131–54139
Wei L, Zhou W, Wang L et al (2000) beta(1)-integrin and PI 3-kinase regulate RhoA-dependent activation of skeletal alpha-actin promoter in myoblasts. Am J Physiol Heart Circ Physiol 278:H1736–H1743
Conflict of interest
We declare that we have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Additional information
Yuan X P and Dong Ming have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Yuan, X.P., Dong, M., Li, X. et al. GRP78 promotes the invasion of pancreatic cancer cells by FAK and JNK. Mol Cell Biochem 398, 55–62 (2015). https://doi.org/10.1007/s11010-014-2204-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-014-2204-2