Abstract
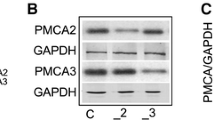
Cellular calcium homeostasis is controlled predominantly by the plasma membrane calcium pump (PMCA). From four PMCA isoforms, PMCA1 and PMCA4 are ubiquitous, while PMCA2 and PMCA3 are found in excitable cells. We have previously shown that suppression of neuron-specific PMCAs in non-differentiated PC12 cells changed the cell morphology and triggered neuritogenesis. Using the microarrays, real-time PCR and immunodetection, we analyzed the effect of PMCA2 or PMCA3 reduction in PC12 cells on gene expression, with emphasis on calmodulin (CaM), neuromodulin (GAP43) and MAP kinases. In PMCA-suppressed lines total CaM increased, and the calm I and calm II genes appeared to be responsible for this effect. mRNA and protein levels of GAP43 were increased, however, the amount of phosphorylated form was lower than in control cells. Localization of CaM/GAP43 and CaM/pGAP43 differed between control and PMCA-reduced cells. In both PMCA-modified lines, amounts of ERK1/2 increased. While pERK1 decreased, the pERK2 level was similar in all examined lines. PMCA suppression did not change the p38 amount, but the p-p38 diminished. JNK2 protein decreased in both PMCA-reduced cells without changes in pJNK level. Microarray analysis revealed distinct expression patterns of certain genes involved in the regulation of cell cycle, proliferation, migration, differentiation, apoptosis and cell signaling. Suppression of neuron-specific PMCA isoforms affected the phenotype of PC12 cells enabling adaptation to the sustained increase in cytosolic Ca2+ concentration. This is the first report showing function of PMCA2 and PMCA3 isoforms in the regulation of signaling pathways in PC12 cells.






Similar content being viewed by others
Abbreviations
- CaM:
-
Calmodulin
- CREB:
-
cAMP Response element-binding
- ERK1 and ERK2:
-
Extracellular signal-regulated kinase 1 and 2
- GAP43:
-
Growth-associated protein 43, neuromodulin
- JNK2:
-
c-Jun N-terminal kinase 2
- NFAT:
-
Nuclear factor of activated T-cells
- NGF:
-
Nerve growth factor
- p38 MAPK:
-
p38 Mitogen-activated protein kinase
- PMCA:
-
Plasma membrane Ca2+-ATPase
References
Brini M, Carafoli E (2000) Calcium signalling: a historical account, recent developments and future perspectives. Cell Mol Life Sci 57:354–370
Cerella C, Diederich M, Ghibelli L (2010) The dual role of calcium as messenger and stressor in cell damage, death, and survival. Int J Cell Biol 2010:546163. doi:10.1155/2010/546163
Strehler EE, Zacharias DA (2001) Role of alternative splicing in generating isoform diversity among plasma membrane calcium pumps. Physiol Rev 81:21–50
Brandt P, Neve RL (1992) Expression of plasma membrane calcium-pumping ATPase mRNAs in developing rat brain and adult brain subregions: evidence for stage-specific expression. J Neurochem 59:1566–1569
Strehler EE, Filoteo AG, Penniston JT, Caride AJ (2007) Plasma-membrane Ca2+ pumps: structural diversity as the basis for functional versatility. Biochem Soc Trans 35:919–922
Di Leva F, Domi T, Fedrizzi L, Lim D, Carafoli E (2008) The plasma membrane Ca2+ ATPase of animal cells: structure, function and regulation. Arch Biochem Biophys 476:65–74
Elwess NL, Filoteo AG, Enyedi A, Penniston JT (1997) Plasma membrane Ca2+ pump isoforms 2a and 2b are unusually responsive to calmodulin and Ca2+. J Biol Chem 272:17981–17986
Gnegy ME (1993) Calmodulin in neurotransmitter and hormone action. Annu Rev Pharmacol Toxicol 33:45–70
Garcia ML, Usachev YM, Thayer SA, Strehler EE, Windebank AJ (2001) Plasma membrane calcium ATPase plays a role in reducing Ca2+-mediated cytotoxicity in PC12 cells. J Neurosci Res 64:661–669
Keller D, Grover AK (2000) Nerve growth factor treatment alters Ca2+ pump levels in PC12 cells. Neuroreport 11:65–68
Zylinska L, Kozaczuk A, Szemraj J, Kargas C, Kowalska I (2007) Functional importance of PMCA isoforms in growth and development of PC12 cells. Ann N Y Acad Sci 1099:254–269
Kosiorek M, Podszywalow-Bartnicka P, Zylinska L, Zablocki K, Pikula S (2011) Interaction of plasma membrane Ca2+-ATPase isoform 4 with calcineurin A: implications for catecholamine secretion by PC12 cells. Biochem Biophys Res Commun 411:235–240. doi:10.1016/j.bbrc.2011.06.098
Beckel-Mitchener AC, Miera A, Keller R, Perrone-Bizzozero NI (2002) Poly(A) tail length-dependent stabilization of GAP-43 mRNA by the RNA-binding protein HuD. J Biol Chem 277:27996–28002
Sobczak M, Boczek T, Ferenc B, Taha J, Kozaczuk A, Wiktorska M, Sacewicz-Hofman I, Niewiarowska J, Zylinska L (2010) Functional characteristic of PC12 cells with reduced microsomal glutathione transferase 1. Acta Biochim Pol 57:589–596
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25:402–408
Weinman J, Della Gaspera B, Dautigny A, Pham Dinh D, Wang J, Nojima H, Weinman S (1991) Developmental regulation of calmodulin gene expression in rat brain and skeletal muscle. Cell Regul 2:819–826
Kornhauser JM, Cowan CW, Shaywitz AJ, Dolmetsch RE, Griffith EC, Hu LS, Haddad C, Xia Z, Greenberg ME (2002) CREB transcriptional activity in neurons is regulated by multiple, calcium-specific phosphorylation events. Neuron 34:221–233
Cocas LA, Miyoshi G, Carney RS, Sousa VH, Hirata T, Jones KR, Fishell G, Huntsman MM, Corbin JG (2009) Emx1-lineage progenitors differentially contribute to neural diversity in the striatum and amygdale. J Neurosci 29:15933–15946
Stühmer T, Anderson SA, Ekker M, Rubenstein JL (2002) Ectopic expression of the Dlx genes induces glutamic acid decarboxylase and Dlx expression. Development 129:245–252
Lönn P, Zaia K, Israelsson C, Althini S, Usoskin D, Kylberg A, Ebendal T (2005) BMP enhances transcriptional responses to NGF during PC12 cell differentiation. Neurochem Res 30:753–765
Dehmelt L, Halpain S (2005) The MAP2/Tau family of microtubule-associated proteins. Genome Biol 6:204. doi:10.1186/gb-2004-6-1-204
Bronstein JM, Chen K, Tiwari-Woodruff S, Kornblum HI (2000) Developmental expression of OSP/claudin-11. J Neurosci Res 60:284–290
Kageyama R, Ohtsuka T, Kobayashi T (2008) Roles of Hes genes in neural development. Dev Growth Differ 50(Suppl 1):S97–S103
Holmes GP, Negus K, Burridge L, Raman S, Algar E, Yamada T, Little MH (1998) Distinct but overlapping expression patterns of two vertebrate slit homologs implies functional roles in CNS development and organogenesis. Mech Dev 79:57–72
Chedotal A, Kerjan G, Moreau-Fauvarque C (2005) The brain within the tumor: new roles for axon guidance molecules in cancers. Cell Death Differ 12:1044–1056
Kano Y, Otsuka F, Takeda M, Suzuki J, Inagaki K, Miyoshi T, Miyamoto M, Otani H, Ogura T, Makino H (2005) Regulatory roles of bone morphogenetic proteins and glucocorticoids in catecholamine production by rat pheochromocytoma cells. Endocrinology 146:5332–5340
Parra M, Verdin E (2010) Regulatory signal transduction pathways for class IIa histone deacetylases. Curr Opin Pharmacol 10:454–460
Lim ACB, Qu D, Qi RZ (2003) Protein-protein interactions in Cdk5 regulation and function. Neurosignals 12:230–238
Jessberger S, Gage FH, Eisch AJ, Lagace DC (2009) Making a neuron: Cdk5 in embryonic and adult neurogenesis. Trends Neurosci 32:575–582
Zheng YL, Li BS, Kanungo J, Kesavapany S, Amin N, Grant P, Pant HC (2007) Cdk5 Modulation of mitogen-activated protein kinase signaling regulates neuronal survival. Mol Biol Cell 18:404–413
Gleeson JG, Lin PT, Flanagan LA, Walsh CA (1999) Doublecortin is a microtubule-associated protein and is expressed widely by migrating neurons. Neuron 23:257–271
Shmueli O, Gdalyahu A, Sorokina K, Nevo E, Avivi A, Reiner O (2001) DCX in PC12 cells: CREB-mediated transcription and neurite outgrowth. Hum Mol Genet 10:1061–1070
Shafer TJ, Atchison WD (1991) Transmitter, ion channel and receptor properties of pheochromocytoma (PC12) cells: a model for neurotoxicological studies. Neurotoxicology 12:473–492
Arcuri C, Bianchi R, Brozzi F, Donato R (2005) S100B increases proliferation in PC12 neuronal cells and reduces their responsiveness to nerve growth factor via Akt activation. J Biol Chem 280:4402–4414
Palfi A, Kortvely E, Fekete E, Kovacs B, Varszegi S, Gulya K (2002) Differential calmodulin gene expression in the rodent brain. Life Sci 70:2829–2855
Toutenhoofd SL, Strehler EE (2000) The calmodulin multigene family as a unique case of genetic redundancy: multiple levels of regulation to provide spatial and temporal control of calmodulin pools? Cell Calcium 28:83–96
Yamniuk AP, Vogel HJ (2004) Calmodulin’s flexibility allows for promiscuity in its interactions with target proteins and peptides. Mol Biotechnol 27:33–57
Denny JB (2006) Molecular mechanisms, biological actions, and neuropharmacology of the growth-associated protein GAP-43. Curr Neuropharmacol 4:293–304
Benowitz LI, Routtenerg A (1997) GAP-43: an intrinsic determinant of neuronal development and plasticity. Trends Neurosci 20:84–91
Ng YP, Wu Z, Wise H, Tsim KW, Wong YH, Ip NY (2009) Differential and synergistic effect of nerve growth factor and cAMP on the regulation of early response genes during neuronal differentiation. Neurosignals 17:111–120
Moore DL, Goldberg JL (2011) Multiple transcription factor families regulate axon growth and regeneration. Dev Neurobiol (in press). doi:10.1002/dneu.20934
Klee CB (1991) Concerted regulation of protein phosphorylation and dephosphorylation by calmodulin. Neurochem Res 16:1059–1065
Hogan PG, Chen L, Nardone J, Rao A (2003) Transcriptional regulation by calcium, calcineurin, and NFAT. Genes Dev 17:2205–2232
Holton M, Yang D, Wang W, Mohamed TM, Neyses L, Armesilla AL (2007) The interaction between endogenous calcineurin and the plasma membrane calcium-dependent ATPase is isoform specific in breast cancer cells. FEBS Lett 581:4115–4119
Buch MH, Pickard A, Rodriguez A, Gillies S, Maass AH, Emerson M, Cartwright EJ, Williams JC, Oceandy D, Redondo JM, Neyses L, Armesilla AL (2005) The sarcolemmal calcium pump inhibits the calcineurin/nuclear factor of activated T-cell pathway via interaction with the calcineurin A catalytic subunit. J Biol Chem 280:29479–29487
Guerini D, Wang X, Li L, Genazzani A, Carafoli E (2000) Calcineurin controls the expression of isoform 4CII of the plasma membrane Ca2+ pump in neurons. J Biol Chem 275:3706–3712
Sanna B, Bueno OF, Dai YS, Wilkins BJ, Molkentin JD (2005) Direct and indirect interactions between calcineurin-NFAT and MEK1-extracellular signal-regulated kinase 1/2 signaling pathways regulate cardiac gene expression and cellular growth. Mol Cell Biol 25:865–878
Aramburu J, Rao A, Klee CB (2000) Calcineurin: from structure to function. Curr Top Cell Regul 36:237–295
Junttila MR, Li S-P, Westermarck J (2008) Phosphatase-mediated crosstalk between MAPK signaling pathways in the regulation of cell survival. FASEB J 22:954–965
Muroi Y, Ishii T, Teramoto K, Hori M, Nishimura M (2004) Calcineurin contributes to the enhancing effect of adenosine on nerve growth factor-induced neurite outgrowth via the decreased duration of p38 mitogen-activated protein kinase phosphorylation. J Pharmacol Sci 95:124–131
Vaudry D, Stork PJ, Lazarovici P, Eiden LE (2002) Signaling pathways for PC12 cell differentiation: making the right connections. Science 296:1648–1649
Egea J, Espinet C, Comella JX (1998) Calmodulin modulates mitogen-activated protein kinase activation in response to membrane depolarization in PC12 cells. J Neurochem 70:2554–2564
Zheng F, Luo Y, Wang H (2009) Regulation of brain-derived neurotrophic factor-mediated transcription of the immediate early gene Arc by intracellular calcium and calmodulin. J Neurosci Res 87:380–392
Agell N, Bachs O, Rocamora N, Villalonga P (2002) Modulation of the Ras/Raf/MEK/ERK pathway by Ca2+, and calmodulin. Cell Signal 14:649–654
Iwasaki S, Iguchi M, Watanabe K, Hoshino R, Tsujimoto M, Kohno M (1999) Specific activation of the p38 mitogen-activated protein kinase signaling pathway and induction of neurite outgrowth in PC12 cells by bone morphogenetic protein-2. J Biol Chem 274:26503–26510
Acknowledgments
We are grateful to Eleonora Welhan for her excellent technical assistance. Supported by the grant 2P05A 03529 from the Ministry of Education and Science, Poland and grants 502-03/6-086/502-64-003 and 503/6-086-02/503-01 from Medical University of Lodz, Poland.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Boczek, T., Kozaczuk, A., Ferenc, B. et al. Gene expression pattern in PC12 cells with reduced PMCA2 or PMCA3 isoform: selective up-regulation of calmodulin and neuromodulin. Mol Cell Biochem 360, 89–102 (2012). https://doi.org/10.1007/s11010-011-1047-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-011-1047-3