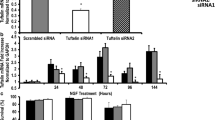
Human nerve growth factor-β (hNGF-β) plays a major role in the development and survival of the central and peripheral nervous systems; therefore, it can, in principle, be used as a therapeutic agent for the treatment of neurodegenerative diseases. As was shown, hNGF-β stimulates neuronal differentiation in vitro and regulates the expression of neuron-related genes. In our study, expression of the ATP1B1, Clusterin, MAO-A, and DUSP6 genes was evaluated in hNGF-treated PC12 cells. At first, hNGF-β protein was generated in a pET39b/BL21 (DE3) bacterial expression system, and biological activity of this protein was confirmed by differentiation of cultured PC12 cells. These cells were treated by the addition of recombinant hNGF-β protein in a final dose of 50 ng/ml diluted in RPMI 1640. Then total RNA was extracted from harvested PC12 cells at day 7 after treatment and used to analyze the expression levels of the above-mentioned genes by real-time PCR. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as a reference gene. It was found that purified hNGF-β protein migrated as a clear band of ~37 kDa in SDS-PAGE. Moreover, differentiation of PC12 cells was observed 7 days after hNGF-β treatment. Expression of the ATP1B1 and DUSP6 genes was upregulated (230 and 193%), respectively after addition of hNGF-β protein. In contrast, the MAO-A and Clusterin genes showed practically no change in their expression, as compared to untreated cells. Generally, our study demonstrated that the treatment of PC12 cells with hNGF-β protein induces neural morphological modifications and differences in the expression of certain neuron-related genes. Changes in the expression of these genes will be applied in an experimental model.
Similar content being viewed by others
References
L. A. Greene, “Nerve growth factor prevents the death and stimulates the neuronal differentiation of clonal PC12 pheochromocytoma cells in serum-free medium,” J. Cell Biol., 78 No. 3, 747-755 (1978).
Z. S. M. Hajihassan, M. R. Bazl, and H. Eftekhary, “Expression of human nerve growth factor β and bacterial protein disulfide isomerase (DsbA) as a fusion protein (DsbA:: hNGF) significantly enhances periplasmic production of hNGF-β in Escherichia coli,” Roman. Biotechnol. Lett., 21, No. 5, 11850 (2016).
P. Gholami Tilko, Z. Hajihassan, and H. Moghimi, “Optimization of recombinant β-NGF expression in Escherichia coli using response surface methodology,” Prep. Biochem. Biotechnol., 47, No. 4, 406-413 (2017).
C. Wiesmann and A. DeVos, “Nerve growth factor: structure and function,” Cell. Mol. Life Sci., 58, Nos. 5/6, 748-759 (2001).
L. A. Greene and A. S. Tischler, “Establishment of a noradrenergic clonal line of rat adrenal pheochromocytoma cells which respond to nerve growth factor,” Proc. Natl. Acad. Sci. USA, 73, No. 7, 2424-2428 (1976).
K. P. Das, T. M. Freudenrich, and W. R. Mundy, “Assessment of PC12 cell differentiation and neurite growth: a comparison of morphological and neurochemical measures,” Neurotoxicol. Teratol., 26, No. 3, 397-406 (2004).
L. A. Greene and G. Rein, “Release, storage and uptake of catecholamines by a clonal cell line of nerve growth factor (NGF) responsive pheochromocytoma cells,” Brain Res., 129, No. 2, 247-263 (1977).
B. Rudkin, P. Lazarovici, B. Levi, et al., “Cell cyclespecific action of nerve growth factor in PC12 cells: Differentiation without proliferation,” EMBO J., 8, No. 11, 3319-3325 (1989).
T. Yang-Feng, J. Schneider, V. Lindgren, et al., “Chromosomal localization of human Na+, K+-ATPase α- and β-subunit genes,” Genomics, 2, No. 2, 128-138 (1988).
B. Pratscher, C. Friedrich, W. Goger, et al., “Characterization of NKIP: A novel, Na+/K+-ATPase interacting protein mediates neural differentiation and apoptosis,” Exp. Cell Res., 1, No. 314, 463-477 (2008).
C. Gutacker, G. Klock, D. Patrick, and K. B. Claudia, “Nerve growth factor and epidermal growth factor stimulate clusterin gene expression in PC12 cells,” Biochem. J., 339, No. 3, 759-766 (1999).
G. S. De Zutter and R. J. Davis, “Pro-apoptotic gene expression mediated by the p38 mitogen-activated protein kinase signal transduction pathway,” Proc. Natl. Acad. Sci. USA, 98, No. 11, 6168-6173 (2001).
J. Sambrook, E. F. Fritschi, and T. Maniatis, Molecular Cloning: A Laboratory Manual, Cold Spring Harbor Laboratory Press, New York (1989).
M. W. Pfaffl, “A new mathematical model for relative quantification in real time RT-PCR,” Nucleic Acids Res., 29, No. 9, e45 (2001).
K. H. Lee, C. J. Ryu, H. J. Hong, et al., “CDNA microarray analysis of nerve growth factor-regulated gene expression profile in rat PC12 cells,” Neurochem. Res., 30, No. 4, 533-540 (2005).
J. H. Kim, T. Y. Lee, K. H. Yoo, et al., “Expression profile identifies novel genes involved in neuronal differentiation,” BMB Rep., 41, No. 2, 146-152 (2008).
K. I. Patterson, T. Brummer, P. M. O’brien, and R. J. Daly, “Dual-specificity phosphatases: critical regulators with diverse cellular targets,” Biochem. J., 418, No. 3, 475-489 (2009).
T. F. Dijkmans, T. G. Schouten, J. T. Kamphorst, et al., “Temporal and functional dynamics of the transcriptome during nerve growth factor-induced differentiation,” J. Neurochem., 105, No. 6, 2388-2403 (2008).
I. Arisi, M. D’Onofrio, R. Brandi, et al., “ProNGF/NGF mixtures induce gene expression changes in PC12 cells that neither singly produces,” BMC Neurosci., 15, 48 (2014).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Agi, E., Namvar, A., Moradi, S. et al. Expression of the Neuron-Related Genes in hNGF-β-Treated PC12 Cells. Neurophysiology 50, 392–397 (2018). https://doi.org/10.1007/s11062-019-09770-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11062-019-09770-3