Abstract
Context
Natal dispersal critically influences eco-evolutionary dynamics and the persistence of spatially structured populations. As both short- and long-distance movements contribute to population persistence in fragmented landscapes, understanding dispersal requires assessing phenotypic and environmental effects on a wide range of distances.
Objectives
To assess phenotypic and environmental correlates of dispersal movements in fragmented landscapes.
Methods
We radio-tracked juvenile middle spotted woodpeckers in fragmented landscapes to assess phenotypic and environmental effects on emigration age, transfer duration (in days), and transfer distances.
Results
Large fledglings and those in good condition emigrated earlier than smaller individuals and those in worse condition. Birds in better condition also reduced transfer duration. Overall, females dispersed earlier, remained shorter at transfer and moved further than males. However, while females increased transfer distances with increasing connectivity, males increased distances with decreasing connectivity. Emigration age increased with decreasing patch size and increasing patch quality, and with decreasing population density in patches with soft edges. Both transfer duration and distance increased with decreasing population density.
Conclusions
The correlations between phenotypic traits of fledglings and their posterior movements suggest that early-life conditions influenced dispersal through carry-over effects. Early emigration from low-quality and high-populated patches can be a behavioural mechanism to quickly escape adverse natal conditions, but population density effects were modulated by edge hardness. Finally, because reductions in connectivity led to similar transfer distances between sexes through a reduction in female distances, a lack of sex-biased dispersal can be a previously overlooked effect of habitat isolation that may alter eco-evolutionary dynamics.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Natal dispersal (hereafter ‘dispersal’), defined as the movement of an individual from birth site to the first breeding site (Greenwood 1980), is a crucial process influencing individual fitness, gene flow, population dynamics and species distributions (Hanski and Gilpin, 1997; Clobert et al. 2001; Bowler and Benton 2005). Even though high dispersal rates may increase simultaneous extinctions by synchronizing the dynamics of local populations (Heino et al. 1997), immigration may rescue declining or sink populations from extinction (Brown and Kodric-Brown 1977; Watkinson and Sutherland 1995), or promote the (re)colonization of empty patches thereby counteracting the extinctions in other patches in fragmented landscapes (Hanski and Gilpin 1997; Robles and Ciudad 2017, 2020). In particular, relatively rare long-distance dispersal events can contribute greatly to the demographic rescue of spatially structured populations (Bohrer et al. 2005; Trakhtenbrot et al. 2005; Huth et al. 2015). Thus, understanding dispersal and its effects on populations requires using proper tracking devices to capture a wide range of dispersal distances (Koenig et al. 1996; Nathan et al 2003).
In active dispersers with high cognitive and locomotion abilities, dispersal is a multicausal process that results from complex decision-making influenced by combinations of intrinsic traits of individuals and environmental factors. Such factors may operate differently at each of the three behavioural stages of dispersal (Matthysen 2012): emigration from the natal area, transfer, and immigration into a breeding area (Ronce 2007; Clobert et al. 2012a). For example, landscape structure is expected to strongly influence the transfer stage and, to a lower extent, emigration, but will have minimal influence on immigration. Conversely, local habitat quality and population density are more likely to influence emigration and immigration than transfer stages (Matthysen 2012).
The anthropogenic habitat loss and fragmentation (habitat size reduction, increase of hard edges, and habitat isolation) offers a framework to study how multiple and diverse factors influence different dispersal stages. Patch size reductions associated with loss and fragmentation of habitat can favour philopatry due to increased emigration cost (Travis and Dytham 1999). However, habitat isolation effects associated with fragmentation will depend on the mortality risks of remaining in the natal patches versus leaving them and transferring through the unfamiliar matrix (Heino and Hanski 2001; Yamaura et al. 2022). Indeed, a low permeability of the matrix can reduce the occurrence and success of long-distance dispersal events that are essential for connecting distant local populations in spatially structured populations (Trakhtenbrot et al. 2005). Regardless of fragmentation, intrinsic habitat degradation can promote emigration to escape adverse conditions in the natal patches such as scarcity of food resources (Dickinson and McGowan 2005; Baglione et al. 2006), or inhibit immigration into patches with reduced habitat quality (Osbourn et al. 2014). Yet, simultaneous evaluations of habitat fragmentation and degradation effects on dispersal by tracking animal movements during the long-neglected emigration and transfer stages are, to our knowledge, lacking.
Interactions among factors increase the complexity of the mechanisms underlying dispersal (Clobert et al. 2012b). For example, habitat degradation may promote dispersal propensity or early emigration to escape local adverse conditions in continuous habitats, but dispersal from degraded habitat patches may be constrained by edge effects associated with fragmentation. Moreover, intrinsic phenotypic traits and extrinsic environmental factors are expected to interact to shape dispersal because individuals with different characteristics are likely to face or experience environmental cues and pressures differently (Serrano et al. 2021). The scarcity of studies that address these and other interactions may explain, at least partly, why dispersal studies have frequently reported diverging or even opposing effects of the same causal agents (Matthysen 2012).
In this study, we examined the influence of multiple phenotypic traits and environmental factors, and their interactions, on the movements of juvenile middle spotted woodpeckers (Dendrocoptes medius) during emigration and transfer stages of dispersal. We addressed these issues by radiotracking dispersal movements of this model species in fragmented habitats (Robles and Ciudad 2017, 2020), which allowed us tracking a wide range of dispersal distances. In particular, we investigated the influence of local characteristics in the natal habitat patches (local habitat quality, population size) and landscape attributes (habitat patch size and isolation, edge softness, matrix composition) while controlling for intrinsic traits of individuals (sex, body size, body condition) in a single analytical framework. We assessed the variation in three key characteristics of natal dispersal: (i) the timing of emigration from the natal site, (ii) the duration of the transfer stage, and (iii) the transfer distance. We expected these three dispersal characteristics to be related to different sets of phenotypic and environmental factors. While local environmental conditions in the natal patches are expected to greatly influence the age at emigration, landscape attributes are expected to have strong influence on transfer duration and distance, and intrinsic morphological traits are also expected to be most influential on characteristics of the transfer stage. Finally, we assessed the influence of interactions among factors on each characteristic and stage of dispersal.
Methods
Study species and area
The middle spotted woodpecker is a territorial resident cavity-nesting bird associated with old rough-barked deciduous forests that provide foraging and nesting substrates in the Western Palearctic (Pasinelli 2003). The species is sensitive to habitat loss, fragmentation, and reduction in local habitat quality (e.g. Pettersson 1985; Robles et al. 2008; Fuller and Robles 2018). Both sexes share parental care and raise one brood per year starting after reaching sexual maturity in their second year of life (Pasinelli 2003).
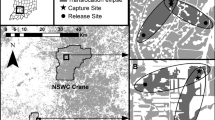
We studied dispersal of juveniles in a 770 km2 area in the Cantabrian Mountains (NW Spain, 42º N, 5º W; see Robles et al. 2007a; Robles and Ciudad 2017; Fig. 1), located at the southwestern edge of the species’ geographic range (Robles and Olea 2003; Kamp et al. 2019). In this area, middle spotted woodpeckers breed in old-growth deciduous oak forests dominated by Pyrenean oak Quercus pyrenaica, but not in other forested patches, such as young oak forests, riverine forests or pine plantations (Robles et al. 2007b). Anthropogenic loss and fragmentation of historically continuous old deciduous forests have reduced its breeding habitat dramatically (Robles et al. 2008; Robles and Ciudad 2012). Consequently, breeding habitat in recent decades covers only 4% of our study area (Fig. 1) throughout 104 old-oak forest patches that range from 0.1 to 874 ha (mean ± SE = 31.9 ± 8.8) in size (Robles and Ciudad 2012). While juveniles in the postfledging period are also mainly associated with old-growth Pyrenean oak forests, they use a variety of deciduous trees (large oaks, poplars and willows within oak and riverine forests) for foraging. Yet, like adults, juvenile woodpeckers strongly avoid open areas (pastures, scrubs, cereal crops) and pine plantations (Ciudad et al. 2009).
Tagging and radiotracking
In springs 2003–2010, we used leg-loop elastic harnesses to tag 79 nestlings one or a few days before fledging with transmitters that weighed 1.6–1.8 g (~ 3–3.5% of nestling mass; see Robles et al. 2007a; Ciudad et al. 2009). Because juvenile mortality of middle spotted woodpeckers is particularly high in the first three weeks after fledging during the dependence period prior to dispersal (Robles et al. 2007a), we restricted our study to the movements of 41 individuals born in 14 habitat patches that survived at least until independence. We radiotracked the juveniles once daily after fledging from June to early September of 2003 (n = 1 fledgling), 2004 (n = 7 fledglings from 4 broods), 2005 (n = 5 fledglings, 3 broods), 2009 (n = 19 fledglings, 9 broods) and 2010 (n = 9 fledglings, 6 broods). We located the birds by the homing-in method until the death of the bird, the end of the transmitter battery life or the disappearance of the radio-signal. Unlike the triangulation method, the homing-in method consists in confirming avian locations by direct visual and/or auditive contacts with the animal (Fuller et al. 2005). We located the birds with a GPS (Garmin eTrex Legend) with an error below 20 m (Ciudad et al. 2009).
Intrinsic individual factors
We estimated body size as the length (in mm) from the tip of the bill to the tip of the tail while laying the bird on its belly and holding its head and its body on a straightedge ruler. We calculated the ‘scaled mass index’ (SMI; Peig and Green 2009) as a measure of body condition as follows:
where Mi and \({L}_{i}\) are the body mass and the body length of individual i as estimated above; \({b}_{SMA}\) is the scaling exponent estimated by the Standardized Major Axis (SMA) regression of M on L; \({L}_{0}\) is the mean of L for the population; and SMI is the predicted body mass for individual i when the linear body measure is standardized to \({L}_{0}\). All the juveniles were weighed to the nearest 0.1 g (digital portable scale, TANITA 1479 V) and measured to the nearest 0.1 cm at 22–23 days old by the same observer. In addition, we obtained blood samples from each individual by puncturing the brachial vein to obtain DNA for molecular sexing (see Appendix S1 for the detailed molecular sexing procedure).
Extrinsic environmental factors
We used the density of large oaks (≥ 37 cm dbh [diameter 1.3 m above ground]) as a measure of local habitat quality (Robles and Ciudad 2012). Large oak density is inversely related to home range size (Pasinelli 2000) and positively associated with patch occupancy and colonization probabilities in the middle spotted woodpecker (Robles and Ciudad 2012, 2020). In small patches (6–68 ha), we calculated the density of large oaks by quantifying the number of large oaks in 0.04-ha circular plots located 100 m apart throughout the whole patch (Robles and Ciudad 2012). For larger patches (up to 874 ha), 0.04-ha plots were located 100 m apart in a circle with 500 m radius around the natal nests (~ 79 ha).
Similar to calculations of local habitat quality, we estimated local population density (territories/10 ha) by counting the number of territories in small habitat patches and in a circle with 500 m radius around the natal nests in larger patches. We repeatedly visited the study sites to map the territories (details in Robles and Ciudad 2012) and only included the area covered by potential breeding habitat (old oak forest) to calculate population density (i.e. we excluded the area covered by matrix within the circles sampled in large patches).
We calculated natal patch size and connectivity from georeferenced aerial photographs verified in the field (Ciudad et al. 2009; Robles and Ciudad 2012). To calculate the connectivity of habitat patches with similar land cover (i.e., “connectivity of landscape elements” sensu Moilanen and Hanski 2001, 2006; inverse of “habitat isolation”), we set pj equal to 1 for all habitat patches (i.e., old oak forest patches) in the following equation provided by Hanski (1994):
where Si is the connectivity of patch i to other patches j (including all habitat patches and a 10-km-wide zone around the north edge of the study area), α scales the effect of distance to dispersal (1/α is the average natal dispersal distance) and is estimated to be 0.192 on the basis of capture-resighting data in this study area (Robles and Ciudad 2012), dij is the distance from patch i to j, and Aj is the size of the patch j. Thus, Si considers distances to all potential habitat patches and their sizes, and is based on the dispersal ability of individuals (Moilanen and Hanski 2001).
To investigate the relationship between juvenile movements during the transfer stage and the matrix composition, we calculated the proportion of deciduous forest in the matrix within a 5.22-km-wide (median interpatch natal dispersal distance in this study area, Robles and Ciudad 2012) zone around the habitat patches. Unlike open areas and pine plantations, deciduous forests composed of young oak and riverside forests are likely to provide foraging and shelter opportunities to juvenile woodpeckers during inter-patch movements (Ciudad et al. 2009). In addition, we calculated the proportion of the perimeter in the natal patch that is surrounded by deciduous matrix to assess the relationship between age at dispersal and edge softness. We used ArcGIS 10.1 (ESRI) for spatial calculations.
Identifying dispersal stages from individual trajectories
We fitted a Hidden Markov Model (HMM) to each individual trajectory using the ‘moveHMM’ package (Michelot et al. 2016) in R (R Core Team 2021) to identify the time of first emigration from the natal home range, either as foray (excursions returning to the natal range) or as permanent emigration (no return), and the end of the first phase of the transfer stage (stop of directed movements farther away from the natal nest, starting exploration of the surroundings of a new area after permanent emigration). Specifically, we used the switches between consecutive movement modes to identify each dispersal stage. On the basis of our observations on the movement trajectories, we constrained HMMs to identifying three types of movement modes: (i) fast, long-distance movements that typically bring the juveniles away from the natal range; (ii) slow, short-distance movements usually associated with encamped behaviour before dispersal or after transfer; and (iii) intermediate movements in distance and velocity likely associated with prospecting behaviour either before or after permanent emigration.
To set up the initial values of the parameters (daily step lengths and turning angles) for each movement mode in the HMMs, we used K-means clustering analysis as an unsupervised machine learning algorithm that splits a given dataset into K groups or clusters (Jain 2010), with K being equal to three (i.e., the movement modes) in our case. We calculated the mean and standard deviations for the three groups of step lengths and used those as the initial values for a gamma distribution of step lengths in the HMMs (mode i: mean of 4.00 km, 1.76 SD; mode ii: 0.21 km, 0.16 SD; mode iii: 1.15 km, 0.42 SD). However, we did not set up values for the turning angles, which followed a uniform distribution, because K-means clustering did not clearly split the dataset into the three movement modes, probably because of the high intra-cluster variation (i.e., standard errors for the turning angles ranged from 2.1 to 2.3 radians). Consequently, we preferred not to fit a particular distribution for the turning angles in the HMMs.
Because HMMs sometimes failed to identify the cut-off points for the dispersal stages, particularly for the end of transfer (see Appendix S2), we used an expert opinion approach to complement and validate model results. Four experts (GP, HR, JF and MG) with previous experience on segmentation of dispersal movement trajectories performed independent (blind to each other) classifications of the dispersal stages for each individual by visual inspection of graphs of (i) movement trajectories, (ii) daily net distances from the natal nests, and (iii) daily step length (i.e., velocity) (Appendix S2).
We then compared the identification of the dispersal stages for each individual obtained from experts’ classifications with that from the HMMs. There was a high concordance among the experts’ classifications, as well as between the experts and the HMMs (see Appendix S2). Given the particularly high congruence among the experts, we gave prevalent value to the experts when their classifications differed from those of HMMs (see Appendix S2).
Statistical analysis of dispersal parameters
We used the age at first emigration from the natal range, regardless of whether the juveniles returned (i.e., forayed) or not (i.e., permanent emigration), as the age when dispersal started (Bowler and Benton 2005). After permanent emigration, we assessed the number of days during which birds remained at the first phase of the transfer stage and the net distances (in meters) to the natal nests at the end of the first phase of the transfer. In four cases, individuals performed a second transfer phase that was not considered in the analysis because movements can be triggered by different motivations in both phases of transfer. Because the locations of the future breeding territories were unknown at the end of the radiotracking period in the summer, we did not include any analysis of immigration defined as settlement into a breeding territory.
We used linear mixed-effects models (LMM) to assess the variation of all the continuous dependent variables (age at dispersal, duration of transfer, net distance at the end of transfer). The variables ‘duration of transfer’ and ‘net distance’ were log-transformed to improve residual outputs. Sex, body size, body condition (SMI), natal patch size, local population density and the density of large oaks around the natal site (i.e., local habitat quality) were fitted as fixed terms in all analyses. Moreover, edge softness was fitted as a fixed term on emigration age analysis. Likewise, matrix composition and the connectivity index of the natal patch to other patches were included as fixed terms on transfer duration and net distance analyses.
Apart from these individual and environmental factors of principal interest, we controlled for the Julian fledging date (with reference to May 1st of the corresponding year) in all analyses, as late fledglings may decide to disperse at earlier ages, stay shorter at transfer and end up closer to the natal nest than earlier fledglings to cope with a narrower time window for dispersal. In addition, the age at emigration was fitted as a fixed term to control for its influence on transfer duration and net distance during transfer, whereas transfer duration was fitted as a fixed term in distance analysis. Finally, brood identity nested within patch identity, and year identity, were fitted as random terms in all the analyses to control for the potential dependence associated with the occurrence of multiple observations within the same broods, patches and years.
We first ran full models with all explanatory variables without interactions for each dependent variable. This approach allowed us to assess the effect of a focal variable while controlling for the influence of other potentially influential variables, which is particularly useful in correlative ecological studies (Korner-Nievergelt et al. 2015). In addition, as the correlations between the continuous explanatory variables of interest were rather low (rs <|0.6|), we did not expect any numerical problems due to covariance (Dormann et al. 2013). We then plotted model residuals versus the explanatory variables to identify potential non-linear effects and interactions that were then added to the full model (Korner-Nievergelt et al. 2015). Continuous explanatory variables were standardised to a mean of zero and standard deviation of one to avoid numerical precision problems and make model coefficients comparable. We used the R package lme4 (Bates et al. 2015) to run the mixed models, and the MuMIn package (Bartón 2020) to calculate pseudo R-squared values of the models as a measure of the variation explained by the fixed terms (Nakagawa and Schielzeth 2013). Bayesian posterior distributions were simulated (5000 simulations) with the R package “arm” (Gelman and Su 2016) and used to calculate model prediction means and 95% credibility intervals (95% CrI).
Results
Age at emigration
Fledglings performed their first emigration out of the natal range at an average age of 42.5 days (SD = 6.4, range = 28–59 days, n = 41 individuals). Females, large individuals and birds in good body condition emigrated earlier in life than males, small birds and those in poor condition (Table 1, Fig. 2). Birds born in small patches and in high quality areas delayed dispersal compared to birds from large patches and low quality areas (Table 1, Fig. 2). Lastly, juveniles from patches with soft edges dispersed earlier in high density areas than in low density areas, whereas this pattern was not found for birds in patches with hard edges (significant interaction of natal population density and edge softness; Table 1, Fig. 3).
Age at first emigration in relation to body size (chord length from bill to tail), body condition (Scaled Mass Index, SMI), habitat quality of natal patch (density of large oaks) and natal patch size. Solid lines show mean values and grey shaded areas 95% CrI of model predictions for males (light grey) and females (dark grey) with all other model predictors set to their mean values. Dots depict raw data points for males (white dots) and females (black dots); n = 41 individuals
Age at emigration in relation to local population density in patches with hard edges (10th quantile, corresponding to a deciduous forest proportion of 0.292; solid line), intermediate edge softness (50th quantile, corresponding to 0.558; dashed line) and soft edges (90th quantile, corresponding to 1.000; pointed line). Lines show the mean values and grey shaded areas 95% CrI of model predictions for females with all other model predictors set to their mean values. Dots depict raw data points; n = 41 individuals
Duration of transfer
Once permanent emigration occurred, dispersers stayed on average 2.3 days at the first transfer stage (SD = 1.6, range = 1–7 days, n = 35 individuals). Males and birds in poor condition stayed longer at the transfer stage than females and individuals in good condition (Table 2, Fig. 4). Individuals that emigrated at older ages and those that were born in low populated patches tended to stay longer at transfer than those birds that emigrated at younger ages and individuals from high populated patches (Table 2, Fig. 4). Transfer duration was not significantly related to any other factor (Table 2).
Duration of transfer in relation to body condition (Scaled Mass Index, SMI) and local population density. Lines show mean values and shaded areas 95% CrI of model predictions for males (light grey) and females (dark grey) with all other model predictors set to their mean values. Dots are raw data points for males (white dots) and females (black dots); n = 35 individuals
Transfer distance
At the end of the transfer stage, dispersers were located on average 3071 m from the natal nests (SD = 1784, range = 224–6748 m, n = 35 individuals). Individuals from low density areas were likely to end up farther away from the natal nest than birds from high density areas (Table 3, Fig. 5). In addition, we did not find significant differences between sexes under low connectivity conditions (i.e., high isolation), but females moved farther than males under high connectivity conditions (significant interaction of sex and connectivity; Table 3, Fig. 5). Individuals from late broods and those birds that stayed short at transfer moved shorter distances than individuals from early broods and those that showed long transfer duration. Transfer distance was not significantly related to any other factor (Table 3).
Net distance from the natal nest at the end of transfer in relation to local population density and patch connectivity. Lines show mean values and shaded areas 95% CrI of model predictions for males (light grey) and females (dark grey) with all other model predictors set to their mean values. Dots are raw data points for males (white dots) and females (black dots); n = 35 individuals
Discussion
Our study revealed previously overlooked patterns underlying the inherently multi-causal natal dispersal process. Different dispersal movement features were associated with differing combinations and interactions of phenotypic traits, social factors, local habitat features, and landscape attributes. The age at emigration was strongly related to landscape fragmentation attributes (patch size, edge softness), local habitat quality and social conditions (population density) in the natal patches, whereas transfer duration (population density) and transfer distance (patch connectivity, population density) were associated with different sets of environmental features. Apart from environmental factors, different sets of phenotypic traits modulated the age at emigration (body size, body condition), transfer duration (body condition, sex) and transfer distance (sex). Finally, the influence of interactions on emigration age (edge softness by population density) and transfer distance (sex by connectivity) supports the hypothesis that interactions among factors play a major role to elucidate the underlying mechanisms of dispersal (Matthysen 2012; Serrano et al. 2021).
Phenotypic traits are expected to have stronger influence on transfer than on emigration parameters (Matthysen 2012). Our results, however, indicate that phenotypic traits can be strong predictors of parameters in both dispersal stages. After controlling for patch size and the density of large oaks (two proxies of habitat quality), large birds and those in good condition dispersed at earlier ages than small birds and those in poor condition. In addition, birds in good condition remained shorter at transfer than birds in poor condition. As our measure of body condition (the ‘scaled mass index’) is likely to be associated with the relative size of energetic reserves after controlling for structural body size (Peig and Green 2009), we suggest that positive body condition-dependent movement (early emigration and quick transfer) may be associated with a high capacity to overcome energetic constraints of dispersal (Stobutzki 1997). Not excluding this hypothesis, birds in good condition may cope better with predation risks during dispersal than those in poor condition (Bonte et al. 2012). In addition, because juveniles continue growing after fledging, early departure of large birds can be associated with a more advanced developmental stage compared to smaller birds at fledging time.
Because body size and body condition at fledging are correlated, at least partly, with environmental features (Richner 1992; Freeman et al. 2020), our results on body size- and body condition-dependent movements support the hypothesis that local environmental conditions early in life (i.e. in the natal nests) have carry-over effects on dispersal (Barbraud et al. 2003). Moreover, the covariation between dispersal parameters and phenotypic traits suggests the presence of dispersal syndromes, which are likely to be modified by habitat fragmentation through spatial structuring of local conditions (Cote et al. 2017). While individuals in good body condition can be seen as early and fast dispersers, individuals in worse body condition are comparatively late and slow dispersers. Fast travelling reduces the energetic costs of making large displacements (Klarevas-Irby et al. 2021) and may be suitable for individuals in good condition that are able to cope with the high dispersal costs in more fragmented landscapes (Benoit et al. 2020). Conversely, slow exploration in the neighbourhood of the natal grounds may be an adequate strategy for individuals in poor condition, which can be less proficient to overcome the costs of crossing unfamiliar environments such as hostile matrixes (Yoder et al. 2004; Bonte et al. 2012). Simulation models show that, unlike individuals with high energetic reserves (i.e., in good condition), those with low energetic reserves (i.e., in poor condition) can benefit from slowing down to forage in the matrix and thus overcome starvation risk when facing low predation risk, but optimal speed of dispersal may depend on the interaction between animal’s energetic reserves and predation risk in a complex way (Zollner and Lima 2005). In addition, slow exploration may help individuals to gather thorough information about the landscape that enable them to cope with the potential risks of crossing the matrix (Cornelius et al. 2017).
That juvenile females emigrated at earlier ages and remained longer at transfer than males fits into the well-known pattern of female-biased dispersal in birds (Greenwood and Harvey 1982; Clarke and Saether 1997). Further support for this hypothesis comes from the fact that females born in well-connected patches moved longer distances than males. However, females and males born in isolated patches moved similar distances, likely because habitat fragmentation, through an increase of patch isolation, precluded females from dispersing to more distant habitats. Consequently, habitat fragmentation masked the pattern of female-biased dispersal distances. Because dispersal is a sex-biased process in many animals (e.g,. Trochet et al. 2016) and habitat fragmentation is a widespread spatial process (Lindenmayer and Fischer 2006), we suggest that a lack of sex-biased dispersal distances as a result of habitat isolation may be a general but previously overlooked pattern that may alter eco-evolutionary dynamics in numerous organisms.
Natal dispersal movements were related to three spatial processes associated with landscape changes that are known to drive population declines in many organisms: increase of habitat patch isolation, reduction of patch size and increase in edge hardness of patches (Lens et al. 2002; Lindenmayer and Fischer 2006). The age at emigration was negatively related to the size of the natal patches, showing that fragmentation delayed dispersal. Because late dispersers may have less time to find high-quality areas for settlement than early dispersers (Stamps et al. 2005; Bonte et al. 2012), landscape fragmentation may reduce individual fitness by delaying dispersal. Moreover, in dense (i.e., more saturated) populations, juveniles born in patches with soft edges emigrated earlier compared to those in patches with hard edges. Juveniles may have delayed dispersal until they developed the conditions required to cope with the high risks of moving through the unfamiliar and hostile matrix beyond hard edges (Yoder et al. 2004; Bonte et al. 2012), but young birds were eager to leave saturated habitat patches earlier when they were surrounded by a less hostile matrix beyond soft edges. Unlike juveniles from small, saturated patches, young birds born in unsaturated populations (i.e., large and low-populated patches) may have enough space to disperse within the natal habitat patch, which will result in early emigration. Partly supporting this hypothesis, individuals born in large habitat patches were likely to disperse within the natal patch with no need to move through the matrix. Indeed, while only one of four juveniles (25%) left the largest patch (874 ha) and moved to an adjacent habitat fragment, most juveniles (86%) left smaller natal patches (< 150 ha) and crossed relatively long distances in the matrix before finding other habitat patches. Lastly, transfer distance was positively related to patch connectivity (inverse of isolation) in females (i.e., the long dispersing sex), suggesting that extreme isolation may inhibit dispersal mobility (see e.g., Cooper and Walters 2002).
Between-study variation in taxa and research approaches may explain why empirical assessments of dispersal in response to habitat fragmentation have provided contrasting results. Most studies that compared fragmented versus continuous landscapes failed to find evidence of fragmentation effects on dispersal propensity (e.g. in agamid lizards Amphibolurus nobbi, Driscoll and Hardy 2005; prairie voles Microtus ochrogaster, Smith and Batzli 2006; wolf spiders Pardosa monticola, Bonte et al. 2006; but see the case of skinks Egernia cunninghami, Stow et al. 2001), but reductions in patch size (increased fragmentation) in within-landscape studies have been associated with increased dispersal probability (Pakanen et al. 2017). Individuals in fragmented landscapes increased dispersal distances compared to those in continuous landscapes (e.g., in Eurasian nuthatches Sitta europaea, Matthysen et al. 1995; Florida scrub-jays Aphelocoma coerulenses, Breininger 1999; Coulon et al. 2010). Within a given landscape, however, increases in patch isolation may lead to reductions in distances moved by the most dispersing sex and to increases in distances by the most philopatric sex (this study). Moreover, the incorporation of matrix and patch edge conditions within given landscapes improves models on movement behaviour during emigration and transfer (e.g., Biz et al. 2017; this study), which will result in a better understanding of dispersal and its consequences on the persistence of spatially structured populations (Yamaura et al. 2022). Thus, even if both study approaches can be useful to understand the causes and mechanisms underlying dispersal patterns in response to landscape fragmentation, the within-landscape approach can reveal subtle behavioural responses that cannot be unravelled by the between-landscape approach. In combination with behavioural studies of movements, assessments of genetic structure and diversity within and between landscapes can help understanding the influence of landscape configuration on functional connectivity among populations through effective dispersal (e.g., Ritter et al. 2021; Pasinelli 2022).
Variation in local habitat quality adds substantial complexity to our understanding of dispersal patterns in fragmented landscapes. While the transfer components (duration, distance) of dispersal were not significantly related to local habitat quality, the age at emigration was positively correlated with local habitat quality in the natal area. Early dispersal may therefore contribute to escape low-quality habitats quickly. Supporting this hypothesis, experimental studies show that a scarcity of food resources in the natal areas may promote dispersal propensity (Dickinson and McGowan 2005) and early emigration (Fattebert et al. 2019). We did not find evidence for significant interactions between intrinsic habitat quality of patches and landscape features (patch size, edge softness, patch isolation), suggesting independent effects of anthropogenic habitat degradation (reduced habitat quality) and fragmentation on dispersal.
After controlling for habitat quality variables (i.e., patch size and local quality of habitat), we found negative density-dependence for timing of emigration (early age dispersal in high-populated patches) in patches surrounded by soft edges, which agrees with the hypothesis of increased intraspecific competition as a mechanism that promotes dispersal (Matthysen 2005; Ronce 2007). This hypothesis was not supported in patches surrounded by hard edges, likely because hard edges constrained emigration out of the natal patch. Thus, changes in edge hardness modulated the effect of population density on emigration timing in fragmented landscapes. Finally, dispersers increased transfer distances and duration when moving from low populated patches (i.e., negative density-dependence). Because low-populated areas can induce Allee effects (Allee 1951) that enhance the extinction risk of small populations by reducing individual fitness (Stephens and Sutherland 1999), increasing transfer distances may help dispersers to avoid detrimental Allee effects by leaving low-populated areas. This may be the case in our study system, where small local populations exhibit high extinction risks that are likely to be associated with Allee effects (Robles and Ciudad 2017). Like juvenile dispersers in this study, adult dispersers (i.e., floaters) also avoid low-populated patches in our system. As both juveniles after becoming adults in their second calendar year and floaters (i.e., at least in their third calendar year) contribute to renovate the breeding population by replacing the lost breeders (Robles and Ciudad 2017), escaping from low-populated areas will reduce the pool of individuals for population turnover, which ultimately can lead to further reductions of local populations that are associated with Allee effects.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Allee WC (1951) Cooperation among animals, with human implications. Henry Schuman, New York
Baglione V, Canestrari D, Marcos JM, Ekman J (2006) Experimentally increased food resources in the natal territory promotes offspring philopatry and helping in cooperatively breeding carrion crows. Proc R Soc Lond Ser B Biol Sci 273:1529–1535
Barbraud C, Johnson AR, Bertault G (2003) Phenotypic correlates of post-fledging dispersal in a population of greater flamingos: the importance of body condition. J Anim Ecol 72:246–257. https://doi.org/10.1046/j.1365-2656.2003.00695.x
Bartón K (2020) MuMIn: multi-model inference. R package version 1.43-17. http://cran.r-project.org/web/packages/MuMIn/index.html
Bates D, Maechler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48
Benoit L, Hewison AM, Coulon A, Debeffe L, Gremillet D, Ducros D et al (2020) Accelerating across the landscape: the energetic costs of natal dispersal in a large herbivore. J Anim Ecol 89:173–185
Biz M, Cornelius C, Metzger JP (2017) Matrix type affects risk perception and movement behavior of a Neotropical understory forest bird. Perspect Ecol Conserv 15:10–17
Bohrer GIL, Nathan RAN, Volis S (2005) Effects of long-distance dispersal for metapopulation survival and genetic structure at ecological time and spatial scales. J Ecol 93:1029–1040
Bonte D, Borre JV, Lens L, Maelfait J-P (2006) Geographical variation in wolf spider dispersal behaviour is related to landscape structure. Anim Behav 72:655–662
Bonte D, Van Dyck H, Bullock JM, Coulon A, Delgado M, Gibbs M et al (2012) Costs of dispersal. Biol Rev 87:290–312
Bowler DE, Benton TG (2005) Causes and consequences of animal dispersal strategies: relating individual behaviour to spatial dynamics. Biol Rev 80:205–225
Breininger DR (1999) Florida scrub-jay demography and dispersal in a fragmented landscape. Auk 116:520–527
Brown JH, Kodric-Brown A (1977) Turnover rates in insular biogeography: effect of immigration on extinction. Ecology 58:445–449
Ciudad C, Robles H, Matthysen E (2009) Postfledging habitat selection of juvenile middle spotted woodpeckers: a multi-scale approach. Ecography 32:676–682
Clarke AL, Saether B-E (1997) Sex biases in avian dispersal: a reappraisal. Oikos 79:429–438
Clobert J, Baguette M, Benton TG, Bullock JM (2012a) Dispersal ecology and evolution. Oxford University Press
Clobert J, Massot M, Le Galliard J-F (2012b) Multi-determinism in natal dispersal: the common lizard as a model system. In: Clobert J, Baguette M, Benton TG, Bullock JM (eds) Dispersal Ecology and Evolution. Oxford University Press, pp 3–18
Clobert J, Danchin E, Dhont AA, Nichols JD (2001) Dispersal. Oxford University Press, New York
Cooper CB, Walters JR (2002) Experimental evidence of disrupted dispersal causing decline of an Australian passerine in fragmented habitat. Conserv Biol 16:471–478
Cornelius C, Awade M, Cândia-Gallardo C, Sieving KE, Metzger JP (2017) Habitat fragmentation drives inter-population variation in dispersal behavior in a Neotropical rainforest bird. Perspect Ecol Conserv 15:3–9
Cote J, Bestion E, Jacob S, Travis J, Legrand D, Baguette M (2017) Evolution of dispersal strategies and dispersal syndromes in fragmented landscapes. Ecography 40:56–73
Coulon A, Fitzpatrick JW, Bowman R, Lovette IJ (2010) Effects of habitat fragmentation on effective dispersal of Florida scrub-jays. Conserv Biol 24:1080–1088
Dickinson JL, McGowan A (2005) Winter resource wealth drives delayed dispersal and family-group living in western bluebirds. Proc R Soc Lond Ser B: Biol Sci 272:2423–2428
Driscoll DA, Hardy CM (2005) Dispersal and phylogeography of the agamid lizard Amphibolurus nobbi in fragmented and continuous habitat. Mol Ecol 14:1613–1629
Dormann CF, Elith J, Bacher S, Buchmann C, Carl G, Carré G et al (2013) Collinearity: a review of methods to deal with it and a simulation study evaluating their performance. Ecography 36:27–46
Fattebert J, Perrig M, Naef-Daenzer B, Grüebler MU (2019) Experimentally disentangling intrinsic and extrinsic drivers of natal dispersal in a nocturnal raptor. Proc R Soc Lond Ser B: Biol Sci 286(1910):20191537
Freeman NE, Norris DR, Sutton AO, Newman AEM (2020) Raising young with limited resources: supplementation improves body condition and advances fledging of Canada Jays. Ecology 101(1):e02909. https://doi.org/10.1002/ecy.2909
Fuller MR, Millspaugh JJ, Church KE, Kenward RE (2005) Wildlife radiotelemetry. In: Braun CE (ed) Techniques for wildlife investigations and management, 6th edn. The Wildlife Society Press, Bethesda, pp 377–417
Fuller R, Robles H (2018) Conservation strategies and habitat management for European forest birds. In: Mikusiński G, Roberge J-M, Fuller RJ (eds) Ecology and conservation of forest birds. Cambridge University Press, Cambridge, pp 455–507
Gelman A, Su YS (2016) Data analysis using regression and multilevel/hierarchical models. Package Version 1:9–3
Greenwood PJ (1980) Mating systems, philopatry and dispersal in birds and mammals. Anim Behav 7:165–167
Greenwood PJ, Harvey PH (1982) The natal and breeding dispersal of birds. Annu Rev Ecol Evol Syst 13:1–21
Hanski I (1994) A practical model of metapopulation dynamics. J Anim Ecol 63:151–162
Hanski I, Gilpin ME (1997) Metapopulation biology: ecology, genetics and evolution. Academic Press, San Diego
Heino M, Hanski I (2001) Evolution of migration rate in a spatially realistic metapopulation model. Am Nat 157:495–511
Heino M, Kaitala V, Ranta E, Lindström J (1997) Synchronous dynamics and rates of extinctions in spatially structured populations. Proc R Soc Lond Ser B Biol Sci 264:481–486
Huth G, Haegeman B, Pitard E, Munoz F (2015) Long-distance rescue and slow extinction dynamics govern multiscale metapopulations. Am Nat 186:460–469
Jain AK (2010) Data clustering: 50 years beyond K-means. Pattern Recognit Lett 31(8):651–666
Kamp L, Pasinelli G, Milanesi P, Drovetski SV, Kosiński Z, Kossenko S, Robles H, Schweizer M (2019) Significant Asia-Europe divergence in the middle spotted woodpecker (Aves: Picidae: Dendrocoptes medius). Zoo Scr 48:17–32
Klarevas-Irby JA, Wikelski M, Farine DR (2021) Efficient movement strategies mitigate the energetic cost of dispersal. Ecol Lett 24:1432–1442
Koenig WD, Van Vuren D, Hooge PN (1996) Detectability, philopatry, and the distribution of dispersal distances in vertebrates. Trends Ecol Evol 11:514–517
Korner-Nievergelt F, Roth T, von Felten S, Guélat J, Almasi B, Korner-Nievergelt P (2015) Bayesian data analysis in ecology using linear models with R, BUGS, and Stan. Academic Press, Amsterdam
Lens L, Van Dongen S, Norris K, Githiru M, Matthysen E (2002) Avian persistence in fragmented rainforest. Science 298:1236–1238
Lindenmayer DB, Fischer J (2006) Habitat fragmentation and Landscape change. An ecological and conservation synthesis. Island Press, Washington
Matthysen E (2005) Density-dependent dispersal in birds and mammals. Ecography 28:403–416
Matthysen E (2012) Multicausality of dispersal: a review. In: Clobert J, Baguette M, Benton TG, Bullock JM (eds) Dispersal ecology and evolution. Oxford University Press, Oxford, pp 3–18
Matthysen E, Adriaensen F, Dhont AA (1995) Dispersal distances of nuthatches, Sitta europaea, in a highly fragmented forest habitat. Oikos 72:375–381
Michelot T, Langrock R, Patterson TA (2016) moveHMM: an R package for the statistical modelling of animal movement data using hidden Markov models. Methods Ecol Evol 7(11):1308–1315
Moilanen A, Hanski I (2001) On the use of connectivity measures in spatial ecology. Oikos 95:147–151
Moilanen A, Hanski I (2006) Connectivity and metapopulation dynamics in highly fragmented landscapes. In: Crooks KR, Sanjayan M (eds) Connectivity conservation. Cambridge University Press, Cambridge, pp 44–71
Nakagawa S, Schielzeth H (2013) A general and simple method for obtaining R2 from generalized linear mixed-effects models. Methods Ecol Evol 4:133–142
Nathan R, Perry G, Cronin JT, Strand AE, Cain ML (2003) Methods for estimating long-distance dispersal. Oikos 103:261–273
Osbourn MS, Connette GM, Semlitsch RD (2014) Effects of fine-scale forest habitat quality on movement and settling decisions in juvenile pond-breeding salamanders. Ecol Appl 24(7):1719–1729
Pakanen V-M, Koivula K, Flodin L-Å, Grissot A, Hagstedt R, Larsson M et al (2017) Between-patch natal dispersal declines with increasing natal patch size and distance to other patches in the endangered Southern Dunlin Calidris alpina schinzii. Ibis 159:611–622
Pasinelli G (2000) Oaks Quercus sp. and only oaks? Relations between habitat structure and home range size of the middle spotted woodpecker Dendrocopos medius. Biol Conserv 93:227–235
Pasinelli G (2003) Dendrocopos medius middle spotted woodpecker. BWP Update 5:49–99
Pasinelli G (2022) Genetic diversity and spatial genetic structure support the specialist-generalist variation hypothesis in two sympatric woodpecker species. Conserv Genet 23:821–837
Peig J, Green AJ (2009) New perspectives for estimating body condition from mass/length data: the scaled mass index as an alternative method. Oikos 118(12):1883–1891
Pettersson B (1985) Relative importance of habitat area, isolation and quality for the occurrence of Middle Spotted Woodpecker Dendrocopos medius (L.) in Sweden. Holarct Ecol 8:53–58
R Core Team (2021) R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria.
Richner H (1992) The effect of extra food on fitness in breeding carrion crows. Ecology 73:330–335
Ritter CD, Ribas CC, Menger J, Borges SH, Bacon CD, Metzger JP, Bates J, Cornelius C (2021) Landscape configuration of an Amazonian island-like ecosystem drives population structure and genetic diversity of a habitat-specialist bird. Landscape Ecol 36:2565–2582
Robles H, Ciudad C (2012) Influence of habitat quality, population size, patch size, and connectivity on patch-occupancy dynamics of the middle spotted woodpecker. Conserv Biol 26:284–293
Robles H, Ciudad C (2017) Floaters may buffer the extinction risk of small populations: an empirical assessment. Proc R Soc Lond Ser B: Biol Sci 284:20170074
Robles H, Ciudad C (2020) Assessing the buffer effect of floaters by reinforcing local colonisation in spatially structured populations. Anim Conserv 23:484–490
Robles H, Olea PP (2003) Distribución y abundancia del Pico Mediano (Dendrocopos medius) en una población meridional de la Cordillera Cantábrica. Ardeola 50:275–280
Robles H, Ciudad C, Vera R, Baglione V (2007a) No effect of habitat fragmentation on post-fledging, first-year and adult survival in the middle spotted woodpecker. Ecography 30:685–694
Robles H, Ciudad C, Vera R, Olea PP, Matthysen E (2008) Demographic responses of middle spotted woodpeckers (Dendrocopos medius) to habitat fragmentation. Auk 125:131–139
Robles H, Ciudad C, Vera R, Olea PP, Purroy FJ, Matthysen E (2007b) Sylvopastoral management and conservation of the middle spotted woodpecker at the south-western edge of its distribution range. Forest Ecol Manag 242:343–352
Ronce O (2007) How does it feel to be like a rolling stone? Ten questions about dispersal evolution. Ann Rev Ecol Evol Syst 38:231–253
Serrano D, Cortés-Avizanda A, Zuberogoitia I, Blanco G, Benítez JR, Ponchon C et al (2021) Phenotypic and environmental correlates of natal dispersal in a long-lived territorial vulture. Sci Rep 11:5424
Smith JE, Batzli GO (2006) Dispersal and mortality of prairie voles (Microtus ochrogaster) in fragmented landscapes: a field experiment. Oikos 112:209–217
Stamps JA, Krishnan VV, Reid ML (2005) Search costs and habitat selection by dispersers. Ecology 86:510–518
Stephens PA, Sutherland WJ (1999) Consequences of the Allee effect for behaviour, ecology and conservation. Trends Ecol Evol 14:401–405
Stobutzki IC (1997) Energetic cost of sustained swimming in the late pelagic stages of reef fishes. Mar Ecol Progr Ser 152:249–259
Stow AJ, Sunnucks P, Gardner MG (2001) The impact of habitat fragmentation on dispersal of Cunningham’s skink (Egernia cunninghami): evidence from allelic and genotypic analyses of microsatellites. Mol Ecol 10:867–878
Trakhtenbrot A, Nathan R, Perry G, Richardson DM (2005) The importance of long-distance dispersal in biodiversity conservation. Divers Distrib 11:173–181
Travis JMJ, Dytham C (1999) Habitat persistence, habitat availability and the evolution of dispersal. Proc R Soc Lond Ser B: Biol Sci 266:723–728
Trochet A, Courtois EA, Stevens VM, Baguette M, Chaine A, Schmeller DS et al (2016) Evolution of sex-biased dispersal. Q Rev Biol 91(3):297–320
Watkinson AR, Sutherland WJ (1995) Sources, sinks and pseudo-sinks. J Anim Ecol 64:126–130
Yamaura Y, Fletcher RJ, Lade SJ, Higa M, Lindenmayer D (2022) From nature reserve to mosaic management: Improving matrix survival, not permeability, benefits regional populations under habitat loss and fragmentation. J Appl Ecol 59:1472–1483
Yoder JM, Marschall EA, Swanson DA (2004) The cost of dispersal: predation as a function of movement and site familiarity in ruffed grouse. Behav Ecol 15:469–476
Zollner PA, Lima SL (2005) Behavioral tradeoffs when dispersing across a patchy landscape. Oikos 108:219–230
Acknowledgements
We are grateful to the numerous volunteers who assisted with data collection, especially to Chano Robles, Gema Solana, Rubén Vera, Gianluca Roncalli, Lieven Devreese and Elfi Verschoren. Patrick Scherler provided useful comments during data analysis. Two anonymous referees provided useful comments on an earlier version of the manuscript. Bird banding and radiotracking was conducted under permits from the Junta de Castilla y León and Spanish Ministry of Environment.
Funding
Open access funding provided by Swiss Ornithological Institute. Several grants and institutions covered our expenses: FPU grant of the Spanish Ministry of Education to CC; grants by Junta de Castilla y León, University of León, Xunta de Galicia (Ángeles Alvariño and Plan I2C grants), and Schifferli scholarship from the Swiss Ornithological Institute to HR. The Spanish Ministry of Education and Science (REN 2002-03587/GLO) and Xunta de Galicia (INCITE08PXIB103259PR, ED431C 2018/57) partly financed this research.
Author information
Authors and Affiliations
Contributions
HR conceived the ideas and designed the methodology; HR and CC collected the data; HR and MG led manuscript conceptualization; HR led data analysis and the writing of the manuscript; CC, ZP, JF, GP, MT and MG assisted with analysis and interpretation of the data; MV led the molecular sexing analysis; all authors contributed critically to the drafts and gave final approval for publication.
Corresponding author
Ethics declarations
Conflict of interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Robles, H., Ciudad, C., Porro, Z. et al. Phenotypic and environmental correlates of natal dispersal movements in fragmented landscapes. Landsc Ecol 37, 2819–2833 (2022). https://doi.org/10.1007/s10980-022-01509-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-022-01509-6