Abstract
Context
A core theme in ecohydrology is understanding how hydrology affects spatial variation in the composition of species assemblages (i.e., beta diversity). However, most empirical evidence is from research in upland rivers spanning small spatial extents. Relatively little is known of the consequences of hydrological variation for beta diversity across multiple spatial scales in lowland rivers.
Objectives
We sought to examine how spatial variation in hydrology and fish beta diversity within and among rivers changed over time in response to intensification and cessation of hydrological drought.
Methods
We used monitoring data of fish assemblages, coupled with hydrological and biophysical data, to test how spatial variation in hydrology and multiple components of fish beta diversity in lowland rivers of the Murray—Darling Basin (Australia) varied across spatial scales during contrasting hydrological phases.
Results
Spatial variation in hydrology among rivers declined with increasing duration of drought before increasing during a return to above-average flows. Spatial variation in hydrology within rivers did not show consistent changes between hydrological phases. Beta diversity among and within rivers showed variable, river-specific changes among hydrological phases for both incidence- and abundance-based components of assemblage composition.
Conclusions
Inconsistent hydrology—beta diversity patterns found here suggest that mechanisms and outcomes of drought and flooding impacts to beta diversity are context-dependent and not broadly generalisable. Our findings indicate that hydrological fluctuations occurring in the Murray—Darling Basin in the period analysed here did not cause significant or consistent homogenisation or differentiation of freshwater fish assemblages.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ecological research is increasingly expanding to understanding and predicting the factors responsible for variation in species assemblages across multiple spatial scales (McGill et al. 2015; Pound et al. 2019; Chase et al. 2020). A fundamental theme in this expansion is spatial variation in the composition of assemblages or ‘beta diversity’ (Anderson et al. 2006; Socolar et al. 2016; Bush et al. 2016; Soininen et al. 2018). Beta diversity quantifies the similarity or difference in communities across scales and can be applied to different facets of biodiversity: taxonomic (emphasising species identity), functional (ecological traits), or phylogenetic (evolutionary history and potential) (Rocha et al. 2018; Carvalho et al. 2020). Our understanding of the factors affecting beta diversity across both terrestrial and aquatic ecosystems is largely based on fixed or ‘static’ differences in ecological variables across landscapes (e.g., Heino et al. 2015 for review). We know far less about how beta diversity varies over time time in response to temporally variable natural and anthropogenic factors (Cañedo‐Argüelles et al. 2020; Crabot et al. 2020; Li et al. 2020; Lindholm et al. 2020).
Hydrological variability in space and time is a driver of beta diversity of freshwater, riparian, and floodplain biota via multiple ecohydrological mechanisms (Bogan et al. 2013; Rolls et al. 2018; Crabot et al. 2020). Variation in hydrological connectivity influences beta diversity by determining organism dispersal among patches of habitat, with floods frequently reducing beta diversity by increasing connectivity, and low water levels increasingly fragmenting patches of habitat leading to increased beta diversity as local assemblages diverge (Larned et al. 2010; de Macedo-Soares et al. 2010). Additionally, spatial variation in hydrology drives beta diversity because differences in hydrological regimes favour selection of contrasting species assemblages across space (Lytle and Poff 2004; Leigh and Sheldon 2009; Bogan et al. 2013). When spatial ‘hydro-diversity’ declines, such as during broad-scale hydrological drought, increasingly harsh environmental conditions trigger community disassembly (e.g., Chase 2007; O’Neill, 2016) by eliminating intolerant species from individual locations up to the extent of entire regions. Changes in the spatial variation in riverine hydrology over time are predicted to reduce or increase beta diversity (‘biotic homogenisation’ and ‘biotic differentiation’, respectively; Olden and Rooney 2006), possibly by changes in spatial ‘hydro-diversity’ and loss and recovery of species during hydrological fluctuations. Most empirical evidence of hydrology-beta diversity linkages is from studies spanning either headwater streams or single river systems (see Rolls et al. 2018). In comparison, we are unaware of empirical evidence of the effect of hydrology on beta diversity in a multi-scaled context across large, lowland river systems.
Climate fluctuations cause temporal variation in hydrology and provide opportunities to examine how freshwater beta diversity is driven by hydrology across multiple spatial scales. Water-resource development has been the predominantly perceived cause of altered spatial variation in hydrology; hydrological regimes can either increase or decrease in similarity among and within rivers because of dams and water diversions (Thoms and Parsons 2003; Poff et al. 2007; McManamay et al. 2012; Mackay et al. 2014; Rolls et al. 2021). However, hydrological fluctuations (drought and flooding) imposed by climate variability may also lead to periods of spatial hydrological homogenisation or differentiation, which in turn drive changes in riverine beta diversity through spatial variation in assembly-disassembly processes (e.g., Thomaz et al. 2007; Leigh and Sheldon, 2009; Larned et al., 2010). Assessing how climate fluctuations affect beta diversity of riverine assemblages would help to understand and predict hydrological scenarios that lead to biotic homogenisation or differentiation across spatial scales. Such evidence is necessary because change in beta diversity is a fundamental component of global biodiversity change (McGill et al. 2015), especially for freshwater fish (Villéger et al. 2011). Yet most empirical research on change in beta diversity in freshwaters has tested effects of non-native species invasions (e.g., Leprieur et al. 2008; Taylor 2010; Toussaint et al. 2014; Sommerwerk et al. 2017), with only six of 116 studies published to May 2020 testing the effects of temporal hydrological variability (Rolls, unpublished data).
One approach to test the effect of hydrology on beta diversity of river biota is to examine how spatial variation in hydrology differs among distinct hydrological phases, and the associated patterns of beta diversity across multiple spatial scales. Australia’s Murray – Darling Basin spans multiple climatic zones contributing to spatial variability in hydrology among its major rivers. Inter-annual variation in rainfall and temperature in the basin is substantial, with prolonged periods of below-average annual rainfall and above-average annual temperature punctuated with wetter periods leading to distinct deviations in river discharge as low flows and floods, respectively. The fish fauna of the Murray – Darling Basin comprises species that have contrasting linkages between hydrology and population dynamics (e.g., Baumgartner et al. 2014). Studies of beta diversity of fish assemblages in the basin have revealed (i) substantial spatial variation in composition associated with variation in hydrology among rivers (e.g., Gehrke et al. 1995), (ii) that assemblage composition in individual rivers shifts between hydrological events such as floods (e.g., Rolls and Wilson 2010; Rayner et al. 2015; Cruz et al. 2020), and (iii) that spatial variation in assemblage composition within rivers differs in association with low- and high-flow events (e.g., Balcombe et al. 2006, 2011). Elsewhere (e.g., in the USA), declining surface water availability was associated with both differentiation and homogenisation of fish assemblages at different spatial scales (Taylor et al. 2019; Rogosch and Olden 2019). The scale-specific effects of temporal variation in hydrology on beta diversity have not yet been explored across large rivers such as those of the Murray – Darling Basin.
The objective of this study was to examine how beta diversity of lowland fish assemblages varied among multiple spatial scales over nine years of contrasting hydrological phases driven by climate fluctuations (Table 1). We first examined how spatial variation in hydrology differed among and within rivers of the Murray—Darling Basin during multiple phases of hydrological drought and flooding. Second, we tested how beta diversity of fish assemblages varied among hydrological phases to determine whether drought or post-drought periods were associated with apparent homogenisation or differentiation. We examined beta diversity in the context of replacement and richness difference components of total incidence-based beta diversity (Podani and Schmera 2011; Legendre 2014) in conjunction with temporal variation in abundance-based compositional heterogeneity. Examining these different components of beta diversity is informative because evidence is scant regarding whether temporal changes in beta diversity in response to hydrological variation manifest as changes in total beta diversity or replacement, richness-difference, or abundance components (Ruhí et al. 2017; Taylor et al., 2019; Crabot et al. 2020), particularly for fish assemblages. Finally, we tested how hydrological variability affected the relative contributions of antecedent hydrology, distances among pairs of sampling sites, and local environmental variables to each aspect of beta diversity to determine whether assemblage-environment relationships were variable or consistent across contrasting hydrological conditions.
Methods
Study area
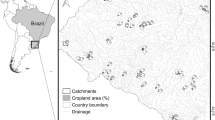
The Murray – Darling Basin spans 1,061,000 km2 of south-eastern Australia and is drained by 23 major rivers (Fig. 1). Most of the basin is low gradient, yet portions along its eastern boundary are undulating and steeper terrain. The climate varies across the basin, transitioning from hot to cool semi-arid (Köppen climate classification: Bsh, Bsk) from the north to the south in the western half, and from humid subtropical (Cfa) to oceanic (Cfb) in the eastern half, with small patches of warm-summer Mediterranean (Csb) along the southern perimeter. Consequently, annual rainfall increases from west to east, with the seasonality of rainfall switching from summer (November-March) dominated in the north to winter (May–October) dominated in the south. The Murray – Darling Basin is Australia’s major agricultural region, producing 39% of the nation’s total agricultural production (ABS 2008).
Location of sites (circles) where fish assemblages were sampled from eight major river systems of the southern and northern Murray-Darling Basin during November 2004-May 2013. Filled circles are sites that were paired with nearby hydrological gauging stations (see text for details) and used for modelling spatial, environmental, and hydrological drivers of beta diversity across seven of the selected study rivers. Unfilled circles indicate sites that were not close enough to be paired to hydrological gauging stations (and therefore excluded from beta diversity modelling), yet were included in broader analyses of temporal change in beta diversity within and among the eight study rivers
Natural hydrological variability within rivers of the Murray—Darling Basin is substantial (Kennard et al. 2010), with discharge seasonality switching from predominantly late summer in the northern portion (i.e., rivers draining into the Darling River) to late winter and early spring in the south (rivers draining into the Murray River) (MDBC 2008). Natural hydrological regimes have been altered substantially by the combined effects of large dams (generally located in upland and headwater regions) and both water extraction and irrigation diversion (particularly in lowland regions). Anthropogenic impacts on hydrological regimes have occurred as decreased magnitude and frequency of flooding, altered discharge seasonality (particularly in the southern portion), and increased baseflow magnitude, overall leading to ‘temporal flow homogenisation’ at intra-annual and inter-annual time scales (Maheshwari et al. 1995; McMahon and Finlayson 2003; Humphries et al. 2008). While a detailed analysis of the effects of anthropogenic hydrological alteration on the magnitude of spatial variation in hydrological regimes within and among rivers of the Murray—Darling Basin has not been done, water-resource development has reduced longitudinal hydrological variability in some rivers in the basin’s north (Thoms and Parsons 2003).
During 2002-early 2010, the Murray—Darling Basin had a protracted period of below-average rainfall, leading to increasing hydrological drought (‘Millennium Drought’), followed by a La-Niña event of substantial rainfall and flooding during late 2010–2012 (Fig. 2). We used a nine-year period spanning this prolonged drought and return to wetter conditions to examine how beta diversity of riverine fish assemblages varied over time. We distinguished hydrological phases using temporal analysis of the annual standardised discharge averaged across 198 stream flow records from 1960 to 2018. Hydrological phases were distinguished via trends in standardised discharge as ‘initial drought’ (2004–2006), ‘prolonged drought’ (2007–2009), and ‘post-drought’ (2010–2012) (Fig. 2).
Mean annual rainfall (a), air temperature (b), and standardised discharge from 1960 to 2018 from 198 streamflow gauging stations (c) spread throughout the central and northern Murray-Darling Basin. Mean annual discharge is presented as a proportion of the long-term mean annual discharge recorded between 1960 and 2018 for sites with complete gauging records. Hydrological phases were defined as ‘initial drought’ (2004–2006), ‘prolonged drought’ (2007–2009), and ‘post-drought’ (2010–2012), represented by the yellow, orange, and green bars respectively. The horizontal lines in each plot indicate mean values for each variable
Data sources
Fish: Fish were sampled in each of the 23 major river systems at three-year cycles during 2004–2013 as part of a “river health” monitoring program (Davies et al. 2010; MDBA 2012). Sites were selected with a stratified random design, whereby rivers were stratified into elevation zones (i.e., lowland, slope, upland, and montane), and the locations of sampling sites within each zone were randomly selected with GIS. The number of sites in each zone differed among rivers because of variation in river network length. At each site, fish were sampled with a standardised protocol of boat and/or backpack electrofishing combined with unbaited shrimp traps. If randomly selected sites were unable to sampled effectively at the time of sampling (e.g., reach was dry, access was unable to be gained), replacement sites were sampled (see Chessman 2018). Of the 23 river systems sampled, we selected the Border Rivers, Condamine, Darling, Gwydir, Lachlan, Macquarie, Murrumbidgee, and Namoi rivers as they span the major climate zones of the Basin and ≥ eight sites were sampled within the slope and lowland zones of each river during each hydrological phase. Of these eight selected rivers, the Lachlan and Murrumbidgee are part of the ‘southern basin’ and the Border Rivers, Condamine, Darling, Gwydir, Macquarie, and Namoi rivers represent the ‘northern basin’ (Fig. 1).
Hydrology: We sourced daily discharge data (ML day−1) from 1990 to 2013 from government water agencies (WaterNSW and the Queensland Department of Natural Resources, Mines and Energy) for all streamflow gauging stations that were located within 20 km of fish monitoring sites. Gaps in records (mean days across gauging records = 1.34%, maximum = 7.31% of all days during 1990–2014) were infilled by multiple regression from the nearest gauge record within each river using the River Analysis Package (Marsh 2004), providing the R2 value was ≥ 0.80 (stream gauge data were discarded if there were gaps that could not be filled confidently with an R2 < 0.80).
Spatial and local environmental variables: Spatial variation in riverine fish assemblages is influenced by a variety of spatial, environmental, and biotic factors (e.g., Jackson et al. 2001). We matched study sites to segments of the Australian national stream (GIS) layer using ArcMap, to extract values for environmental variables as the Australian Stream Environmental Attributes Database (Stein and Nevill, 2011). We selected a subset of these environmental variables for each sampling site to estimate the physical, climatic, and vegetation cover (reach elevation, stream order, proportion of natural forest within the stream segment, average annual mean air temperature, average coldest-month minimum temperature, and average hottest-month maximum temperature calculated during 1970–2000).
Data processing and variable calculation
Fish: We extracted all fish data produced from sampling in these eight river systems during 2004–2013 from the Murray – Darling Basin Authority Fish and Macroinvertebrate database (www.data.gov.au) to produce a sample × species (count) matrix. Two different sets of analyses were done to examine fish beta diversity patterns and beta diversity—environmental relationships among hydrological phases. The first made use of all fish data from the eight study rivers recorded during the study period, and the second used a ‘reduced’ dataset where fish assemblage data were used only if fish monitoring sites were matched with hydrological gauging station data.
Hydrology
For each streamflow record, we calculated values of eight hydrological metrics for the three-year period (Online Appendix S1) antecedent to fish sampling. We chose this period to match the time interval between sampling events, and because we were specifically interested in how the magnitude of spatial variation in hydrology within and among rivers changed over time. Hydrological metrics reflected the mean duration, frequency, and timing of overbank flows (defined as the two-year annual return interval from the annual flood series) and low-flow events (less than the long-term 90th percentile flow), and the frequency and duration of in-channel flow pulses (defined as events > 0.5 the long-term mean daily flow). Collinear variables were identified if they had Variance Inflation Factors (VIFs) greater than five (5) using the ‘vifcor’ function in the USDM package. A single variable, ‘duration of in-channel flow pulses’, was highly correlated to other variables and was removed prior to all subsequent analyses.
Spatial variables
We used the origin–destination function in the network analysist tool of ArcMap 10.6 to calculate the network distance between pairs of sites based on the river network to create a pairwise distance matrix of watercourse distance between all pairs of study sites (reflecting the distances between pairs of sites relevant for dispersal by strictly aquatic organisms; Heino et al. 2017). From this distance matrix, spatial variables were created using distance-based Moran’s Eigenvector Maps (dbMEM) (Dray et al. 2006), retaining only positive and significantly spatially correlated eigenvectors. No further manipulation of reach-scale environmental variables was needed prior to analyses.
Data analyses
Temporal changes in spatial variation in hydrology within and among rivers
Prior to analyses, we standardised each hydrological variable (mean = 0, SD = 1) to convert all variables to the same scale and prevent any variable having an overwhelming influence on patterns in the dataset by having a broader range of values than another. Dissimilarity in hydrological metrics for pairs of samples was quantified by Euclidean distance. We used a two-factor Permutational Analysis of Variance (PERMANOVA; Anderson 2001) with the ‘adonis’ function in vegan to test how spatial variation in hydrology among rivers (‘River’; fixed) varied among hydrological phases (‘Phase’; fixed). PERMANOVA tests for ‘location’ (i.e., centroids) differences among groups of samples. Therefore, we calculated the mean distance between group centroids for groups of sites from each river during each three-year hydrological phase (n = 24; eight rivers x three periods) using the ‘meandist’ function in the vegan package. Centroids of each group were displayed with Principal Coordinates Analysis (PCoA). Using the same Euclidean distance matrix, we then tested how within-river variation in hydrology varied among hydrological phases using tests of homogeneity of dispersions (PERMDISP; Anderson 2006). Patterns of within-river variation in hydrology were displayed in box plots (x-axis: time, y-axis: mean pairwise distance among group centroids). PERMANOVA and PERMDISP analyses were done using the ‘adonis’ and ‘betadisper’ functions in the vegan package (Oksanen et al. 2019), and PCoA ordinations were produced with the ape package (Paradis et al. 2019) in R 4.0.0 (R Core Development Team 2020).
Temporal changes in beta diversity of fish assemblages
We produced four dissimilarity matrices from the site × species matrix, each quantifying different aspects of incidence and abundance composition to determine the underlying characteristics of beta diversity over time (sensu Anderson et al. 2011). Sørensen’s distance (1) was used to calculate a ‘broad-sense’ dissimilarity in incidence-based composition among samples, which was then decomposed into the replacement (2) and richness-difference (3) dissimilarities using the ‘beta’ function in the BAT package (Cardoso et al. 2015). A fourth dissimilarity matrix used was abundance-standardised assemblage dissimilarity (percentage-difference or ‘Bray–Curtis’ distance; Legendre and De Cáceres 2013). We retained all species not native to the Murray – Darling Basin or possibly translocated among study rivers, because non-native species may be important manifestations of changes in beta diversity across scales (Olden and Poff 2003).
For each dissimilarity matrix, we used a combination of Permutational Multivariate Analysis of Variance (PERMANOVA; Anderson 2001) and tests of homogeneity of multivariate dispersions (PERMDISP; Anderson 2006) to determine how spatial variation in fish assemblage composition varied over time among and within river systems. This combination of statistical tests helps to determine the specific scale(s) of variation in beta diversity over time (see Anderson et al. 2011). We displayed patterns in beta diversity with both PCoA ordinations and graphing distances from group centroids for each group over time. To assist in interpreting changes in beta diversity among hydrological phases (both within and among rivers), we (i) plotted values for recorded site-level species richness (alpha diversity) as grouped boxplots, (ii) tested differences in alpha diversity using PERMANOVA based on a Euclidean distance, and (iii) presented summary data of gamma diversity (γ), mean alpha diversity (α), and Whittaker’s beta diversity (as γ/ α) (see Supplementary Material S3).
Assessing the association of antecedent hydrology and spatial and environmental variables with beta diversity across hydrological periods
We used variation partitioning in constrained ordination (Borcard et al. 1992; Peres-Neto et al. 2006) to assess the relative importance of antecedent hydrology and spatial and local environmental variables in explaining variation in each of the four distance matrices of fish assemblages during each hydrological phase. Predictor variables were grouped into ‘spatial’, ‘local environmental’, and ‘antecedent hydrology’ sets. For each set of predictors, we used forward selection (‘ordiR2step’ in vegan) to determine which specific variables to retain in subsequent analyses (Blanchet et al. 2008). Variation partitioning identifies the unique (‘pure’) and combined contribution of predictor variables in explaining variation in assemblage composition (Borcard et al. 1992; Anderson and Gribble 1998). Variation partitioning and the significance of each pure fraction were assessed by distance-based redundancy analysis (dbRDA; Legendre and Anderson 1999) with the ‘varpart’ and ‘capscale’ functions in vegan (Oksanen et al. 2019). The statistical significance of each pure fraction was tested with the ‘anova’ function.
Results
Spatial variation in hydrology across hydrological periods
Differences in hydrology among rivers were inconsistent among hydrological phases (significant River x Phase interaction; Table 2). Overall, spatial variation in hydrology among rivers was significantly lower during the prolonged-drought phase than during both initial-drought and post-drought phases (PERMDISP test of centroids P = 0.048), as evidenced by clustering of river centroids in the Principal Coordinates Analysis (Fig. 3a). Within-river hydrological variability was similar among the three hydrological phases for all rivers (Fig. 3b) except for the Condamine River, where spatial variation in hydrology was lowest during initial drought, increased during prolonged drought, and was highest after the drought.
Spatial variation in hydrology (a) among rivers (as centroids of hydrological gauging sites presented with Principal Coordinates Analysis ordination) and (b) within each river across contrasting hydrological phases. In ordination space (a) magnitude of separation (dissimilarity) of river centroids for each hydrological phase reflects spatial variation in hydrology among rivers. In (b), boxplots display the median (horizontal line), 25th and 75th percentile values and, the whiskers (vertical lines) display 1.5 × the upper and lower interquartile range; increasing distance from group median indicates increasing longitudinal variation in hydrology
Scale-dependent change in beta diversity of fish assemblages associated with hydrological phases
Beta diversity among rivers was inconsistent across hydrological phases for each aspect of assemblage composition except the richness-difference component (significant interaction term for River × Hydrological phase; Table 3). Ordination of total incidence-based beta diversity (Sørensen) revealed that fish assemblages in the northern Murray—Darling Basin (Border Rivers, Condamine, Darling, Gwydir, Macquarie and Namoi rivers) were most dissimilar during initial drought, increasingly similar during prolonged drought, and most similar during the post-drought phase (Fig. 4). In contrast, mean assemblage composition of both southern (Lachlan and Murrumbidgee) rivers changed minimally between hydrological phases. Decomposition of overall incidence-based beta diversity into replacement and richness-difference components indicated that lower beta diversity during the post-drought phase was driven by replacement rather than richness differences (Fig. 4). This was further supported by the fact that the richness difference component of beta diversity differed consistently among rivers in all three hydrological phases (non-significant River x Phase interaction; Table 3). For abundance-based assemblage composition, both rivers in the southern basin showed little change in mean composition across hydrological phases, whereas average composition of the remaining rivers varied substantially among hydrological phases (Fig. 4). For each dissimilarity matrix, significant differences among rivers explained the largest proportion of variation (14.7–22.7%), and while the interaction of River × Hydrological phase was usually significant, it contributed only a moderate amount of explained variation (Table 3).
Ordinations by Principal Coordinates Analysis (PCoA) illustrating differences in average fish assemblage composition among rivers and between hydrological phases based on Sørensen dissimilarity (and replacement and richness difference components) for incidence data and percentage-difference (Bray–Curtis) for abundance data. The percentages of total variance explained by each of the first two PCoA axes are indicated in brackets
Overall tests of homogeneity of dispersions (PERMDISP) found that beta diversity within rivers differed significantly among combinations of rivers and hydrological phases for each aspect of assemblage composition (Table 4). For overall incidence-based beta diversity (Sørensen dissimilarity), post-hoc pairwise tests determined that within-river beta diversity in the Gwydir River was significantly lower (P = 0.006) during prolonged drought than during initial drought. In both the Condamine and Darling rivers, within-river beta diversity was significantly lower during the post-drought period (Fig. 5; post-hoc pairwise tests: Condamine P = 0.0008, Darling P = 0.024). Decomposing overall beta diversity into replacement and richness-difference components showed that these changes in within-river beta diversity among hydrological phases were driven primarily by replacement rather richness differences among sites (Fig. 5). Alpha diversity among hydrological phases showed inconsistent patterns among rivers, with the Lachlan, Macquarie, Murrumbidgee, and Namoi rivers showing no significant change in alpha diversity, the Border Rivers and Darling rivers having higher richness during the post-drought phase (compared to both drought-phases), the Condamine River showing lower richness post-drought, and the Gwydir River having lower richness during the initial drought phase (Supplementary Material S4, S5). Spatial variation in abundance-based composition within rivers generally did not differ significantly among hydrological phases, the only exception being the Murrumbidgee River where spatial variation in fish assemblages was significantly lower during the post-drought period (c.f. both the initial and prolonged-drought periods) (Fig. 5; PERMDISP post-hoc pairwise tests P ≤ 0.001).
Grouped box plots of distances from group (river × hydrological phase) medians of fish assemblages for eight large rivers of the Murray – Darling Basin among climate phases based on Sørensen dissimilarity (and replacement and richness components) for incidence data and percentage-difference (Bray–Curtis) for abundance data. Each boxplot displays the median (horizontal line), 25th and 75th percentile values and the whiskers (vertical lines) display 1.5 × the upper and lower interquartile range for each combination of river and hydrological phase; increasing distance from group median indicates increasing within-river beta diversity
Assemblage-environment relationships during contrasting hydrological phases
Variation partitioning showed that the contribution of antecedent hydrology and spatial and environmental variables to beta diversity varied among hydrological phases. The relative contribution and significance of each predictor variable group also varied among beta-diversity components (Table 5). Antecedent hydrology explained a small but significant component of fish beta diversity in the initial-drought and post-drought phases, yet it explained either none or very little of beta diversity during prolonged drought. Across all three hydrological phases, hydrology was significantly associated with overall incidence-based beta diversity, yet it had no significant association with the richness-difference component of beta diversity (Table 5). Local environmental variables were significantly associated with most or all aspects of beta diversity across all hydrological phases. In contrast to environmental factors, spatial variables were not significantly associated with any aspect of incidence-based beta diversity during initial drought, yet were significantly associated with both incidence-based and abundance-based beta diversity during the prolonged-drought and post-drought phases.
Discussion
Beta diversity of fish assemblages in lowland rivers of the Murray – Darling Basin varied inconsistently with contrasting hydrological phases depending on the scale of analysis. Both incidence-based and abundance-based beta diversity among rivers were higher during both initial and prolonged drought phases and lower following drought in the northern Murray – Darling Basin. However, differences in assemblage composition between the two southern rivers remained similar through time. Changes in within-river beta diversity were variable and unique to each river, and depended on whether analyses were based on incidence or abundance composition. These findings suggest that changes in spatial beta diversity associated with temporal variation in hydrology, manifesting as biotic homogenisation or biotic differentiation, are not broadly generalisable to fish assemblages of lowland rivers.
Change in spatial hydrological variability with climate fluctuations
Australia’s Millennium drought was the most severe in terms of the magnitude and duration of reduced precipitation to have occurred during the period 1783–2009 (Gergis et al. 2012), contributing to substantial declines in river discharge in the Murray – Darling Basin (Leblanc et al. 2012). River-scale hydrology differed significantly between the post-drought phase and both initial-drought and prolonged-drought phases. Spatial variation in hydrology among rivers was lowest during prolonged drought compared to both initial drought and post-drought phases, indicating that increased duration of broad-spanning drought leads to loss of spatial hydro-diversity. Such reductions in hydro-diversity are expected because increasing duration of broad-scale drought increases the prevalence and simultaneous occurrence of low or zero-flow conditions (Leblanc et al. 2009; Van Loon 2015). We are unaware of other research examining changes in spatial variation in river hydrology across spatial scales linked with climate fluctuations using a similar analytical process to ours. Yet research from other climatically heterogeneous regions (e.g., the Iberian Peninsula) reveals that moderate and severe drought spans increasing proportions of the landscape (Lorenzo-Lacruz et al. 2013), suggesting that homogenisation of hydrological regimes with drought occurs widely.
Within-river (i.e. longitudinal) hydrological variability was similar among the three hydrological phases for all but one of the rivers where hydrological data were analysed. The exception was the Condamine River, where longitudinal hydrological variability was lowest during initial drought, increased during prolonged drought and was highest during the post-drought phase. Under modified (regulated) conditions, the hydrological regime of the Condamine River has substantial longitudinal variation (Thoms and Parsons 2003), and our analysis indicates that the magnitude of longitudinal hydrological variability varies with hydrological drought. Increasing longitudinal variability between initial-drought and prolonged-drought phases may have been caused by increased irrigation pressure from agriculture as the drought progressed, leading to increased extraction of connected surface and subsurface water, particularly in lowland reaches (e.g. Wen et al. 2011). Further increases in longitudinal hydrological variability following drought may have been due to either geographical variation in runoff that is intercepted by off-river storages that account for most of the stored water in the Condamine River system, or possible losses of surface water via groundwater recharge that are common in alluvial lowland rivers (sensu Larned et al. 2008). These mechanisms are also likely to operate in the remaining study rivers across the northern Murray – Darling Basin, yet the lack of a consistent effect may be due to differences in extraction demands for water masking effects of climate fluctuations.
Temporal changes in beta diversity associated with hydrological phases across spatial scales
Significant changes in among-river beta diversity were associated with contrasting hydrological phases. As Australia’s Millennium Drought progressed, seven of 39 fish species present in the Murray—Darling Basin had significant changes in their occurrence (decreased occurrence: two species, increased occurrence: five species) (Chessman 2013). Significant changes in abundance were also detected for five of these seven species (Chessman 2013). These changes in occurrence and abundance suggest that effects of hydrological variation would be reflected as changes in fish assemblage composition as either incidence-based or abundance-based beta diversity. In our study, beta diversity was lowest among the five northern study rivers during the post-drought phase and higher during both initial-drought and prolonged-drought phases. One hypothesis for this pattern is the role of spatial variation in community assembly-disassembly patterns during contrasting hydrological phases (e.g. inundation, discharge recession) (O’Neill 2016). During hydrological drought, loss of flowing water and habitat contraction likely vary substantially among rivers, meaning that assemblages occupying multiple rivers are at contrasting states of disassembly and therefore have increasing beta diversity. Flooding, in contrast, potentially leads to more pronounced consistency in environmental conditions among rivers (and reaches within rivers), likely contributing to lower beta diversity as multiple assemblages are in more synchronised phases of assembly.
Overall, change in beta diversity associated with hydrological phases varied among different aspects of assemblage composition. Specifically, abundance-based composition showed a pronounced shift in ordination space between drought phases and the post-drought phase. In contrast, incidence-based composition showed minimal changes in overall ordination space among hydrological phases but showed a trend of declining beta diversity among rivers from initial-drought to post-drought. These findings highlight that hydrological variation likely affects different aspects of beta diversity in different ways and combined use of incidence and abundance-based composition is necessary to reveal distinct aspects of multi-species diversity patterns across landscapes (Taylor et al. 2019).
Within-river beta diversity did not change significantly among hydrological phases for most rivers. The lack of a consistent change in within-river beta diversity may have occurred because change in longitudinal hydrological variation was minimal, and therefore often insufficient to affect the distribution and abundance of fish species. Yet, changes in within-river beta diversity among hydrological phases did occur for individual rivers. For example, fish assemblages in the Gwydir River became increasingly homogeneous during prolonged drought and this homogenisation was predominantly driven by changes (i.e., reduction) in species replacement among samples. This decline of beta diversity due to reduced replacement during prolonged drought suggests that some species are ‘filtered out’ of the regional species pool (sensu Chase 2007). Two species (Australian smelt, freshwater catfish) showed marked declines in occurrence, and one species (bony bream) had marked increases in occurrence in the Gwydir River during prolonged drought compared to both initial-drought and post-drought phases (Online Appendix S2). Of these three species, bony bream is predicted to have low vulnerability to drought (Chessman 2013), and Australian smelt has either moderate vulnerability to drought (Chessman 2013) or intermediate resistance but high resilience to drought (Crook et al. 2010). Overall, the progressive filtering of species during increasingly harsh environmental conditions plays a major role in broader changes in beta diversity during hydrological fluctuations. In contrast to the findings from the Gwydir River, within-river beta diversity was significantly lower in the Condamine and Darling rivers during the post-drought phase than during both initial- and prolonged-drought phases. Lower beta diversity in these rivers during the post-drought phase may have been due to the low storage capacity in reservoirs in these two rivers compared to the other study rivers, possibly contributing to an inability to sustain base flows (and hence hydrological connectivity) during drought (Mallen-Cooper and Zampatti 2020). Increased hydrological connectivity enhances organism dispersal among patches during flooding episodes leading to reduced beta diversity (e.g. de Macedo-Soares et al. 2010; Cruz et al. 2020), and this process possibly explains the decline in within-river beta diversity during the post drought phase in the Condamine and Darling Rivers (Larned et al. 2010).
Previous analyses of effects of hydrological variability on fish beta diversity in the Murray – Darling Basin and elsewhere are based on assessments at the scale of individual rivers and frequently report reduced beta diversity associated with increased hydrological connectivity during flooding or loss of species during drought phases (Balcombe et al. 2006, 2011; Rolls and Wilson 2010; Rayner et al. 2015; Miyazono et al. 2015; Cruz et al. 2020). By examining beta diversity – hydrology associations among multiple lowland river systems, our work here indicates that outcomes of hydrological variability (drought, flooding) are variable across river basins. These inconsistent hydrology—beta diversity associations suggest that mechanisms and outcomes of drought and flooding for beta diversity are context-specific and not broadly generalisable. Such conclusions are further supported by the fact that the contribution of antecedent hydrology to assemblage variation relative to the contributions of spatial and environmental variables was highly variable among hydrological phases.
Limitations and caveats applicable to assessing hydrological consequences for beta diversity
The absence of consistent significant change in beta diversity of fish assemblages associated with contrasting hydrological phases in the Murray – Darling Basin can possibly be explained by at least four general hypotheses worthy of consideration in future hydrology – beta diversity research. First, the hydrology of all the rivers in this study has long been altered to some degree by water-resource development prior to fish sampling (MDBC 2008). Such hydrological alterations may have reduced the abundance of, or eliminated, flow-dependent species (sensu Baumgartner et al. 2014) from the river systems studied here, with the remaining species being resistant to hydrological variability and therefore unresponsive to drought, in turn contributing to stable beta diversity. A second hypothesis (an alternative to the first) is that while hydrological fluctuations during the study period were substantial, the biota of the Murray – Darling Basin is likely adapted to even more major hydrological variability over evolutionary time scales that has occurred in the basin over millennia (Ho et al. 2015), and hence show limited response to drought events at the magnitude of the one examined here. Third, if randomly selected study sites could not be sampled in this monitoring program (e.g., inadequate depth for sampling or completely dry), they were replaced with sites in the same zone. Replacement of study sites during drought phases possibly dampened observed changes in the occurrence and abundance of fish species (and therefore beta diversity) by concentrating sampling on drought refuges (Elliot 2000; Magoulick and Kobza 2003; Rayner et al. 2009). The fourth hypothesis relates to the timing over which changes in beta diversity are apparent in response to hydrological events such as drought and flooding. Changes in beta diversity may have occurred primarily during the early stages of the Millennium Drought before SRA fish sampling began, or else beta diversity responses may be delayed and become evident over longer time periods. Although some sampling of fish assemblages was undertaken across the Murray – Darling Basin during the 1990s when rainfall, temperature, and discharge were near to average (Gehrke and Harris 2000), the resulting data are insufficient for comparison with those used in the present study. In contrast to responses during the initial phases of drought, changes in beta diversity driven by hydrological events (particularly flooding, yet also drought) may not be immediately evident but rather may be delayed. For example, beta diversity changes associated with post-drought hydrology in this study may be underestimated by monitoring being restricted to the first two–three years after the end of the drought. Understanding the complete linkages between hydrology and beta diversity therefore requires sufficiently long time-series hydrology and fish-assemblage data to explore how beta diversity at a specific point in time is associated with hydrological events occurring over various antecedent periods.
Conclusions and recommendations
Spatial variation in hydrological regimes is potentially altered over time with climate fluctuations, with possible outcomes for biodiversity across landscape scales. Our study found that changes in (i) the spatial variation in hydrology associated with climate fluctuations and (ii) corresponding spatial beta diversity were river specific (Table 1). Overall, these findings indicate that hydrological fluctuations occurring in the Murray – Darling Basin in the period analysed here did not cause consistent homogenisation or differentiation of freshwater fish assemblages. The increasing availability of broad-scale species-assemblage data combined with hydrological data spanning increasingly large extents in Australia and elsewhere provides ongoing opportunities to determine the ecohydrological mechanisms, hydrological scenarios, and scales where change in freshwater beta diversity is expected to occur.
Data availability
All data used in this study are freely available from data custodians and publicly available databases referred to in the text.
Code availability
Not applicable.
References
ABS (2008) Water and the Murray – Darling Basin. A Statistical Profile 2000–01 to 2005–06. Canberra, Australia
Anderson MJ (2001) A new method for non-parametric multivariate analysis of variance. Austral Ecol 26:32–46
Anderson MJ (2006) Distance-based tests for homogeneity of multivariate dispersions. Biometrics 62:245–253
Anderson MJ et al (2011) Navigating the multiple meanings of β diversity: a roadmap for the practicing ecologist. Ecol Lett 14:19–28
Anderson MJ, Gribble NA (1998) Partitioning the variation among spatial, temporal and environmental components in a multivariate data set. Austral J Ecol 23:158–167
Balcombe SR, Arthington AH, Foster ND, Thoms MC, Wilson GA, Bunn SE (2006) Fish assemblages of an Australian dryland river: abundance, assemblage structure and recruitment patterns in the Warrego River, Murray – Darling Basin. Mar Freshwater Res 57:619–633
Balcombe SR, Arthington AH, Thoms MC, Wilson GG (2011) Fish assemblage patterns across a gradient of flow regulation in an Australian dryland river system. River Res Appl 27:168–183
Baumgartner LJ, Conallin J, Wooden I, Campbell B, Gee R, Robinson WA, Mallen-Cooper M (2014) Using flow guilds of freshwater fish in an adaptive management framework to simplify environmental flow delivery for semi-arid riverine systems. Fish Fish 15:410–427
Blanchet FG, Legendre P, Borcard D (2008) Forward Selection of Explanatory Variables. Ecology 89:2623–2632
Bogan MT, Boersma KS, Lytle DA (2013) Flow intermittency alters longitudinal patterns of invertebrate diversity and assemblage composition in an arid-land stream network. Freshw Biol 58:1016–1028
Borcard D, Legendre P, Drapeau P (1992) Partialling out the spatial component of ecological variation. Ecology 73:1045–1055
Bush A, Harwood T, Hoskins AJ, Mokany K et al (2016) Current uses of beta-diversity in biodiversity conservation: a response to Socolar. Trends Ecol Evolution 31:337–338
Cañedo-Argüelles M et al (2020) As time goes by: 20 years of changes in the aquatic macroinvertebrate metacommunity of Mediterranean river networks. J Biogeograp 47:1861–1874
Cardoso P, Rigal F, Carvalho JC (2015) BAT – Biodiversity Assessment Tools, an R package for the measurement and estimation of alpha and beta taxon, phylogenetic and functional diversity. Methods Ecol Evol 6:232–236
Carvalho JC, Malumbres-Olarte J, Arnedo MA, Crespo LC, Domenech M, Cardoso P (2020) Taxonomic divergence and functional convergence in Iberian spider forest communities: Insights from beta diversity partitioning. J Biogeogr 47:288–300
Chase JM (2007) Drought mediates the importance of stochastic community assembly. Proceedings of the Natural Academy of Sciences 104:17430–17434
Chase JM, Jeliazkov A, Ladouceur E, Viana DS (2020) Biodiversity conservation through the lens of metacommunity ecology. Ann N Y Acad Sci 1469:86–104
Chessman BC (2013) Identifying species at risk from climate change: Traits predict the drought vulnerability of freshwater fishes. Biol Cons 160:40–49
Chessman BC (2018) Dissolved-oxygen, current and temperature preferences of stream invertebrates estimated from field distributions: application to assemblage responses to drought. Hydrobiologia 809:141–153
Crabot J, Heino J, Launay B, Datry T (2020) Drying determines the temporal dynamics of stream invertebrate structural and functional beta diversity. Ecography 43:620–635
Crook DA, Reich P, Bond NR, McMaster D, Koehn JD, Lake PS (2010) Using biological information to support proactive strategies for managing freshwater fish during drought. Mar Freshw Res 61:379–387
Cruz DO, Kingsford RT, Suthers IM, Rayner TS, Smith JA, Arthington AH (2020) Connectivity but not recruitment: Response of the fish community to a large-scale flood on a heavily regulated floodplain. Ecohydrology 13:e2194
Davies PE, Harris JH, Hillman TJ, Walker KF (2010) The Sustainable Rivers Audit: assessing river ecosystem health in the Murray-Darling Basin. Australia Marine and Freshwater Research 61:764–777
de Macedo-Soares PHM, Petry AC, Farjalla VF, Caramaschi EP (2010) Hydrological connectivity in coastal inland systems: lessons from a Neotropical fish metacommunity. Ecol Freshw Fish 19:7–18
Dray S, Legendre P, Peres-Neto PR (2006) Spatial modelling: a comprehensive framework for principal coordinate analysis of neighbour matrices (PCNM). Ecol Model 196:483–493
Elliott JM (2000) Pools as refugia for brown trout during two summer droughts: trout responses to thermal and oxygen stress. J Fish Biol 56:938–948
Gehrke PC, Brown P, Schiller CB, Moffatt DB, Bruce AM (1995) River regulation and fish communities in the Murray – Darling river system, Australia. Regul Rivers: Res Manage 11:363–375
Gehrke PC, Harris JH (2000) Large-scale patterns in species richness and composition of temperate riverine fish communities, south-eastern Australia. Mar and Freshwater Res 51:165–182
Gergis J et al (2012) On the long-term context of the 1997–2009 ‘Big Dry’ in South-Eastern Australia: insights from a 206-year multi-proxy rainfall reconstruction. Clim Change 111:923–944
Heino J, Melo AS, Siqueira T, Soininen J, Valanko S, Bini LM (2015) Metacommunity organisation, spatial extent and dispersal in aquatic systems: patterns, processes and prospects. Freshwater Biol 60:845–869
Heino J, Alahuhta J, Ala-Hulkko T et al (2017) Integrating dispersal proxies in ecological and environmental research in the freshwater realm. Environ Rev 25(3):334–349
Ho M, Kiem AS, Verdon-Kidd DC (2015) A paleoclimate rainfall reconstruction in the Murray-Darling Basin (MDB), Australia: 2 Assessing hydroclimatic risk using paleoclimate records of wet and dry epochs. Water Resour Res 51(10):8380–8396
Humphries P, Brown P, Douglas J, Pickworth A, Strongman R, Hall K, Serafini L (2008) Flow-related patterns in abundance and composition of the fish fauna of a degraded Australian lowland river. Freshw Biol 53:789–813
Jackson DA, Peres-Neto PR, Olden JD (2001) What controls who is where in freshwater fish communities- the roles of biotic, abiotic, and spatial factors. Can J Fish Aquat Sci 58:157–170
Kennard MJ, Pusey BJ, Olden JD, Mackay SJ, Stein JL, Marsh N (2010) Classification of natural flow regimes in Australia to support environmental flow management. Freshw Biol 55:171–193
Larned ST, Datry T, Arscott DB, Tockner K (2010) Emerging concepts in temporary-river ecology. Freshwater Biol 55:717–738
Larned ST et al (2008) The Selwyn River of New Zealand: a benchmark system for alluvial plain rivers. River Res Appl 24:1–21
Leblanc MJ, Tregoning P, Ramillien G, Tweed SO, Fakes A (2009) Basin-scale, integrated observations of the early 21st century multiyear drought in southeast Australia. Water Resour Res. https://doi.org/10.1029/2008WR007333
Leblanc M, Tweed S, Van Dijk A, Timbal B (2012) A review of historic and future hydrological changes in the Murray-Darling Basin. Global Planet Change 80–81:226–246
Legendre P (2014) Interpreting the replacement and richness difference components of beta diversity. Glob Ecol Biogeogr 23:1324–1334
Legendre P, Anderson MJ (1999) Distance-based redundancy analysis: testing multispecies responses in multifactorial ecological experiments. Ecol Monogr 69:1–24
Legendre P, De Cáceres M (2013) Beta diversity as the variance of community data: dissimilarity coefficients and partitioning. Ecol Lett 16:951–963
Leigh C, Sheldon F (2009) Hydrological connectivity drives patterns of macroinvertebrate biodiversity in floodplain rivers of the Australian wet/ dry tropics. Freshw Biol 54:549–571
Leprieur F, Beauchard O, Hugueny B, Grenouillet G, Brosse S (2008) Null model of biotic homogenization: a test with the European freshwater fish fauna. Divers Distrib 14:291–300
Li Z, Xing Y, Liu Z, Chen X, Jiang X, Xie Z, Heino J (2020) Seasonal changes in metacommunity assembly mechanisms of benthic macroinvertebrates in a subtropical river basin. Sci Total Environ 729:139046
Lindholm M, Alahuhta J, Heino J, Toivonen H (2020) No biotic homogenisation across decades but consistent effects of landscape position and pH on macrophyte communities in boreal lakes. Ecography 43:294–305
Lorenzo-Lacruz J, Morán-Tejeda E, Vicente-Serrano SM, López-Moreno JI (2013) Streamflow droughts in the Iberian Peninsula between 1945 and 2005: spatial and temporal patterns. Hydrol Earth Syst Sci 17:119–134
Lytle DA, Poff NL (2004) Adaptation to natural flow regimes. Trends Ecol Evol 19:94–100
Mackay SJ, Arthington AH, James CS (2014) Classification and comparison of natural and altered flow regimes to support an Australian trial of the Ecological Limits of Hydrologic Alteration framework. Ecohydrology 7:1485–1507
Magoulick DD, Kobza RM (2003) The role of refugia for fishes during drought: A review and synthesis. Freshw Biol 48:1186–1198
Maheshwari BL, Walker KF, McMahon TA (1995) Effects of regulation on the flow regime of the River Murray Australia. Regul Rivers: Resmanag 10:15–38
Mallen-Cooper M, Zampatti BP (2020) Restoring the ecological integrity of a dryland river: Why low flows in the Barwon-Darling River must flow. Ecol Manag Restor 21:218–228
Marsh N (2004) River Analysis Package. Monash University, Melbourne, Cooperative Research Centre for Catchment Hydrology
McGill BJ, Dornelas M, Gotelli NJ, Magurran AE (2015) Fifteen forms of biodiversity trend in the Anthropocene. Trends Ecol Evol 30:104–113
McMahon TA, Finlayson BL (2003) Droughts and anti-droughts: the low flow hydrology of Australian rivers. Freshw Biol 48:1147–1160
McManamay RA, Orth DJ, Dolloff CA (2012) Revisiting the homogenization of dammed rivers in the southeastern US. J Hydrol 424–425:217–237. https://doi.org/10.1016/j.jhydrol.2012.01.003
MDBA (2012) Sustainable Rivers Audit 2: The ecological health of rivers in the Murray–Darling Basin at the end of the Millennium Drought (2008–2010) Canberra Australia
MDBC (2008) Murray – Darling Basin Rivers: Ecosystem Health Check, 2004–2007. A summary report based on the Independent Sustainable Rivers Audit Group's SRA Report 1: A Report on the Ecological Health of Rivers in the Murray – Darling Basin, 2004–2007. Murray – Darling Basin Commission, Canberra Australia
Miyazono S, Patino R, Taylor CM (2015) Desertification, salinization, and biotic homogenization in a dryland river ecosystem. Sci Total Environ 511:444–453
Oksanen J et al. (2019) vegan: Community Ecology Package. R package version 2.5–6.
Olden JD, Poff NL (2003) Toward a mechanistic understanding and prediction of biotic homogenization. Am Nat 162:442–460
Olden JD, Rooney TP (2006) On defining and quantifying biotic homogenization. Glob Ecol Biogeogr 15:113–120
O'Neill BJ (2016) Community disassembly in ephemeral ecosystems. Ecology 97:3285–3292
Paradis E, al. e (2019) Package 'Ape': Analyses of Phylogenetics and Evolution
Peres-Neto PR, Legendre P, Dray S, Borcard D (2006) Variation partitioning of species data matrices: estimation and comparison of fractions. Ecology 87:2614–2625
Podani J, Schmera D (2011) A new conceptual and methodological framework for exploring and explaining pattern in presence – absence data. Oikos 120:1625–1638
Poff NL, Olden JD, Merritt DM, Pepin DM (2007) Homogenization of regional river dynamics by dams and global biodiversity implications. Proc Natl Acad Sci USA 104:5732–5737
Pound KL, Lawrence GB, Passy SI (2019) Beta diversity response to stress severity and heterogeneity in sensitive versus tolerant stream diatoms. Divers Distrib 25:374–384
Rayner TS, Jenkins KM, Kingsford RT (2009) Small environmental flows, drought and the role of refugia for freshwater fish in the Macquarie Marshes, arid Australia. Ecohydrology 2:440–453
Rayner TS, Kingsford RT, Suthers IM, Cruz DO (2015) Regulated recruitment: native and alien fish responses to widespread floodplain inundation in the Macquarie Marshes, arid Australia. Ecohydrology 8:148–159
Rocha MP et al (2018) Local environment and space drive multiple facets of stream macroinvertebrate beta diversity. J Biogeogr 45:2744–2754
Rogosch JS, Olden JD (2019) Dynamic contributions of intermittent and perennial streams to fish beta diversity in dryland rivers. J Biogeogr 46:2311–2322
Rolls RJ, Chessman BC, Heino J et al (2021) Consequences of hydrological alteration for beta diversity of fish assemblages at multiple spatial scales. Sci Total Environ 798:149170
Rolls RJ, Heino J, Ryder DS, Chessman BC, Growns IO, Thompson RM, Gido KB (2018) Scaling biodiversity responses to hydrological regimes. Biol Rev 93:971–995
Rolls RJ, Wilson GG (2010) Spatial and temporal patterns in fish assemblages following an artificially extended floodplain inundation event, northern Murray – Darling Basin. Australia Environmental Management 45:822–833
Ruhí A, Datry T, Sabo JL (2017) Interpreting beta-diversity components over time to conserve metacommunities in highly dynamic ecosystems. Conserv Biol 31:1459–1468
Socolar JB, Gilroy JJ, Kunin WE, Edwards DP (2016) How should beta-diversity inform biodiversity conservation? Trends Ecol Evol 31:67–80
Soininen J, Heino J, Wang J (2018) A meta-analysis of nestedness and turnover components of beta diversity across organisms and ecosystems. Glob Ecol Biogeogr 27:96–109
Sommerwerk N, Wolter C, Freyhof J, Tockner K (2017) Components and drivers of change in European freshwater fish faunas. J Biogeogr 44:1781–1790
Stein J, Nevill J (2011) Counting Australia’s protected rivers. Ecol Manag Restor 12:200–206
Taylor CM, Miyazono S, Cheek CA, Edwards RJ, Patino R (2019) The spatial scale of homogenisation and differentiation in Chihuahuan Desert fish assemblages. Freshw Biol 64:222–232
Taylor EB (2010) Changes in taxonomy and species distributions and their influence on estimates of faunal homogenization and differentiation in freshwater fishes. Divers Distrib 16:676–689
Team RCD (2020) R: A Language and Environment for Statistical Computing, 4.0.0 edn. R Foundation for Statistical Computing, Vienna Austria
Thomaz S, Bini L, Bozelli R (2007) Floods increase similarity among aquatic habitats in river-floodplain systems. Hydrobiologia 579:1–13
Thoms MC, Parsons M (2003) Identifying spatial and temporal patterns in the hydrological character of the Condamine-Balonne river. Australia, Using Multivariate Statistics River Research and Applications 19:443–457
Toussaint A, Beauchard O, Oberdorff T, Brosse S, Villeger S (2014) Historical assemblage distinctiveness and the introduction of widespread non-native species explain worldwide changes in freshwater fish taxonomic dissimilarity. Glob Ecol Biogeogr 23:574–584
Van Loon AF (2015) Hydrological drought explained. Wires Water 2:359–392
Villéger S, Blanchet S, Beauchard O, Oberdorff T, Brosse S (2011) Homogenization patterns of the world’s freshwater fish faunas. Proc Natl Acad Sci USA 108:18003–18008
Wen L, Rogers K, Ling J, Saintilan N (2011) The impacts of river regulation and water diversion on the hydrological drought characteristics in the Lower Murrumbidgee River Australia. J Hydrol 405:382–391
Acknowledgements
This work was supported by the New South Wales Environmental Trust (reference: 2018/RD/0051). We thank the Murray – Darling Basin Authority (particularly Ben Seddon) for making the fish datasets publicly available, and WaterNSW (particularly Ashley Webb) and the Queensland Department of Natural Resources and Mines (particularly Ralph deVoil) for providing hydrology data. Guillaume Blanchet provided useful advice regarding statistical analyses. Two reviewers provided useful comments to improve the manuscript.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions. This work was supported by the New South Wales Environmental Trust (Reference: 2018/RD/0051).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rolls, R.J., Chessman, B.C., Heino, J. et al. Change in beta diversity of riverine fish during and after supra-seasonal drought. Landsc Ecol 37, 1633–1651 (2022). https://doi.org/10.1007/s10980-022-01424-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-022-01424-w