Abstract
Context
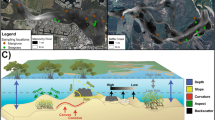
Landscape modification alters the condition of ecosystems and the structure of terrain, with widespread impacts on biodiversity and ecosystem functioning. Seafloor dredging impacts a diversity of flora and fauna in many coastal landscapes, and these processes also transform three-dimensional terrain features. The potential ecological significance of these terrain changes in urban seascapes has, however, not been investigated.
Objectives
We examined the effects of terrain variation on fish assemblages in 29 estuaries in eastern Australia, and tested whether dredging changes how fish associate with terrain features.
Methods
We surveyed fish assemblages with baited remote underwater video stations and quantified terrain variation with nine complementary metrics (e.g. depth, aspect, curvature, slope, roughness), extracted from bathymetry maps created with multi-beam sonar.
Results
Fish diversity and abundance were strongly linked to seafloor terrain in both natural and dredged estuaries, and were highest in shallow waters and near features with high curvature. Dredging, however, significantly altered the terrain of dredged estuaries and transformed the significance of terrain features for fish assemblages. Abundance and diversity switched from being correlated with lower roughness and steeper slopes in natural estuaries to being linked to features with higher roughness and gentler slopes in dredged estuaries.
Conclusions
Contrasting fish-terrain relationships highlight previously unrecognised ecological impacts of dredging, but indicate that plasticity in terrain use might be characteristic of assemblages in urban landscapes. Incorporating terrain features into spatial conservation planning might help to improve management outcomes, but we suggest that different approaches would be needed in natural and modified landscapes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Urbanisation is the leading form of landscape modification globally, with most ecosystems having been transformed to support the needs of an ever-growing human population (Seto et al. 2012; Song et al. 2018). This intense form of landscape modification is typically associated with habitat loss and degradation, pollution, the introduction of pest species, and in some extreme cases, changes in regional climate (Kowarik 2011; Pickett et al. 2011). These negative consequences of urbanisation have significant and widespread impacts on the composition of floral and faunal communities, and can lead to declines in biodiversity, productivity, the rate and distribution of ecological functions and ecosystem services (Johnson and Munshi-South 2017; Berger-Tal and Saltz 2019). In urban environments, ecosystems are replaced with impervious surfaces (e.g. concrete, asphalt, rock-walls) and this typically reduces the number, area, diversity and quality of habitats that are available to organisms in both terrestrial and aquatic landscapes (Bishop et al. 2017; Lepczyk et al. 2017). Intense earthworks and dredging activities are also characteristics of urban environments that are associated with the deepening of shipping channels and the construction of ports and canals, and these practices change the three-dimensional structure, topography and terrain complexity of modified landscapes (Fischer and Lindenmayer 2007; Grimm et al. 2008; Carse and Lewis 2020). There has been considerable research into the impacts of habitat loss, landscape fragmentation and species translocation in urban environments, both on land and in the sea (Pickett et al. 2011; McCauley et al. 2015), but the potential ecological consequences of intense terrain modification are rarely studied in coastal waters (Borland et al. 2021).
Coastal landscapes are focal points for human cities, and this concentrated urbanisation often leads to profound changes in the quality, condition and structure of marine ecosystems, and alterations to the distribution and diversity of animal assemblages (Dafforn et al. 2015; Mayer-Pinto et al. 2018). These impacts occur when ecosystems (e.g. mangrove forests, seagrass meadows, dune vegetation, coastal forests) are modified by artificial structures (e.g. buildings, rock-walls, jetties, pontoons), when nutrients, sediments and pollutants are transported from modified lands into the sea, and where the seafloor is dredged to improve the navigability of coastal waters for shipping (Strain et al. 2018; Todd et al. 2019). Well known examples that illustrate the diversity of impacts from coastal habitat loss and transformation include: reductions in the density and diversity of vertebrate and invertebrate populations, and changes to rates of ecological functions performed by these organisms (Noriega et al. 2012; Huijbers et al. 2015); changes to the composition, functional traits and ecological roles of fish species (Brook et al. 2018; Henderson et al. 2020; Osborne et al. 2021); and changes in the distribution of a diversity of coastal invertebrates, fish, bird and mammal populations in response to declining water quality from dredging operations (Pérez-Ruzafa et al. 2006; Erftemeijer and Lewis 2006; Drabble 2012; Arizaga et al. 2017; Marley et al. 2017). In addition to these widely reported, and direct, ecological impacts of coastal urbanisation on species, assemblages and ecosystems, there are also a myriad of indirect consequences for many species which result from significant changes to the physical features of the seafloor (Waltham and Connolly 2011; Munsch et al. 2017). The terrain of estuaries and coastal waters has been modified widely via the combined effects of dredging, trawling, nourishing, armouring and mining operations, and these structural changes likely have consequences for both coastal biodiversity and ecosystem functioning (Rochette et al. 2010; Teichert et al. 2016; Bolam et al. 2021).
The spatial distribution, abundance and diversity of many populations and assemblages is shaped by variation in the three-dimensional complexity of the landscapes and seascapes in which they live (Badgley et al. 2017; Lepczyk et al. 2021). Terrain features that are characterised by high-relief, great complexity or large morphological variation (e.g. mountains, canyons, pinnacles) are typically hot-spots for biodiversity, particularly when they occur in landscapes that are relatively flat or featureless (Pittman and Brown 2011; Davies and Asner 2014). Topographically complex features of landscapes and seascapes support a diversity of fauna, because they provide refuge from physical stress (e.g. temperature, rainfall, currents) and predators, harbour an abundance of food, and are used as important sites for reproduction (e.g. nesting, spawning) by migratory species both on the land and in the sea (Laundré et al. 2010; Dobrowski 2011; Pittman and Brown 2011). Positive effects of greater relief, complexity and morphological variation in terrain have been reported for most animal groups (e.g. birds, mammals, reptiles, insects) in terrestrial landscapes (Müller and Brandl 2009; Flaspohler et al. 2010; McLean et al. 2016; Einoder et al. 2018), and for invertebrates, bony fishes, elasmobranchs and mammals in a variety of marine seascapes (e.g. coral and rocky reefs, continental shelves, the deep sea) (Bouchet et al. 2015; Pygas et al. 2020; Borland et al. 2021). The ecological significance of terrain variation has, however, never been examined in shallow coastal waters and estuaries. The absence of seafloor terrain research in coastal seascapes is a significant gap in urban ecology, and is surprising, given that they are subjected to frequent modification from dredging and trawling activities and support a range of both natural and artificial terrain features that are characterised by high-relief and complexity (Brook et al. 2018; Henderson et al. 2019).
This study used estuarine seascapes in eastern Australia as a model system to test whether, and how, the ecological effects of terrain and artificial seafloor modification (i.e. via dredging) combine to shape the composition of coastal fish assemblages. Estuarine seascapes are typically comprised of a mosaic of natural ecosystems and structurally complex habitat patches (e.g. mangroves, seagrasses, saltmarshes, rock bars, sandbars, log-snags, channels), which provide a variety of high-relief terrain features that support diverse fish assemblages (Gilby et al. 2018; Henderson et al. 2019). Estuaries are, however, also focal points for coastal development and human recreation, and the combined effects of urbanisation, fishing and dredging fragment natural ecosystems and modify seafloor terrain (Teichert et al. 2016; Amorim et al. 2017). The ecological consequences of landscape modification have been well documented and include changes to the composition of estuarine floral and faunal assemblages, reductions in the diversity of fish and invertebrates, as sensitive specialist species are replaced by generalist taxa that can capitalise on opportunities provided in urban habitats, and variation in the rates of important ecological functions (Bishop et al. 2017; Mayer-Pinto et al. 2018; Olds et al. 2018). It is, however, unknown whether the transformation of the three-dimensional properties of urban seascapes (i.e. terrain modification via dredging) modifies the ecological value of seafloor terrain features for fish. Dredging in coastal seascapes typically results in the modification of seafloor terrain and fish habitat by removing consolidated substrates and creating deep shipping channels with uniform profiles (Madricardo et al. 2019; Eidam et al. 2020). Given the significance of terrain variation for fish assemblages in many marine ecosystems (Monk et al. 2010; Pittman and Brown 2011; Moore et al. 2016), it was hypothesised that in urban estuaries that have been subjected to dredging activity, terrain modification changes the way in which fish associate with seafloor terrain features and result in significant declines in fish diversity and harvested fish abundance.
Materials and methods
Study area
Fish assemblages were surveyed from 29 estuaries in eastern Australia (Fig. 1), which stretched over 1000 km from Waterpark Creek in the north to Currumbin Creek in the south (22° 57′ S, 150° 47′ E–28° 07′ S, 153° 29′ E). The focal estuaries are all permanently open to the sea and encompassed a range of natural seascapes with abundant mangroves, rocky reefs and sandbars (e.g. Baffle Creek), and a variety of modified seascapes that are routinely dredged to improve their navigability for shipping (e.g. Brisbane River) (Hossain et al. 2004; Henderson et al. 2019), and are therefore ideal for investigating effects of terrain modification from dredging on estuarine fish assemblages.
Fish surveys
Fish assemblages were surveyed with baited remote underwater video stations (BRUVS). BRUVS were comprised of a high definition GoPro camera mounted on a 5 kg weight, which was attached to a bait bag via a 0.5 m PVC pole (Gilby et al. 2017b). BRUVS are a widely-used video surveillance method for sampling fish from a diversity of marine ecosystems due to the ability to attract species from all trophic groups (Harvey et al. 2007; Rees et al. 2018; Mosman et al. 2020), and in this study were baited with 500 g of Pilchards (Sardinops sagax) (Wraith et al. 2013; Olds et al. 2018). BRUVS were deployed at 10 sites within the marine extent of each estuary (i.e. n = 290) and were spaced at equal distances upstream from the estuary mouth to the point at which salinity reached 30 psu (Practical Salinity Units) (Gilby et al. 2017a). All BRUVS were deployed on one occasion during the austral winter (i.e. temporal variation was not a focus of this study), for 1 h during daylight, within 2 h of high tide, over unvegetated soft sediments in water depths between one and two metres, and within 30 m of adjacent mangroves when they were present, to minimise the potential confounding effects of tide, water depth and seascape context on fish assemblages (Henderson et al. 2017; Olds et al. 2018). Data on fish diversity and abundance was extracted from BRUVS footage using the standard MaxN statistic (i.e. the maximum number of individual species observed in a single frame of the video footage) (Murphy and Jenkins 2010; Gladstone et al. 2012).
Quantifying seafloor terrain
To investigate whether, and how, changes to seafloor terrain from dredging impact fish assemblages, high-resolution (≤ 1 m−1) digital bathymetric models (DBMs) were created for each estuary using depth soundings from a multi-beam acoustic sounder (Lowrance HDS-7 Gen3). Sonar data was processed in ReefMaster 2.0 and corrected to highest astronomical tide (HAT), to standardise DBMs for the possible confounding effects of tidal range, using tide data collected from pressure sensors (INW Smart Sensor) that were also deployed in each estuary (Li et al. 2017). Using the Spatial Analyst and Benthic Terrain Modeler software tools in ArcGIS (Walbridge et al. 2018), surface metrics were applied to the DBMs to quantify structural variation in seafloor terrain. Nine terrain metrics that are consistent predictors of the effects of terrain on fish assemblages in a diversity of marine seascapes were selected to describe variation in the: relief (average depth), morphology (aspect: northness and eastness; plan curvature; and profile curvature), complexity (rugosity and slope) and composition (backscatter: E1 roughness and E2 hardness) of seafloor terrain (Table 1) (Pittman et al. 2009; Borland et al. 2021). Terrain was indexed by quantifying mean values for each metric within a 500 m buffer around each sampling site; this scale was chosen because it encompasses the home ranges of many estuarine fishes, and has been widely used in other studies that report strong spatial effects on the distribution of fishes in similar coastal seascapes (Olds et al. 2012; Gilby et al. 2018). Estuaries were categorised as either natural or dredged based on their dredging history, which was obtained from dredging permits and maps held by the Australian Government (https://apps.des.qld.gov.au/env-authorities/map/). Dredging is permitted in these estuaries on an annual basis, but insufficient data are available to describe the intensity or frequency of these activities. Dredged channels are, however, still prominent features in disturbed coastal landscapes for up to 10 years after operations cease (Borja et al. 2010), and variation in the frequency of ongoing dredging activities is therefore unlikely to alter relationships between fish assemblages and terrain features in modified estuaries. To account for the possible confounding effects of urbanisation (e.g. urban development) and the availability of vegetated habitats (i.e. mangrove forests and seagrass meadows), we also measured the percentage of urban shoreline within the sampled extent of each estuary, and quantified the area of mangrove and seagrass habitats within 500 m buffers surrounding each sampling location, using benthic habitat maps provided by the Queensland Government (following Brook et al. 2018; Yabsley et al. 2020).
Data analysis
A manyGLM was used to test whether, and how, the effects of terrain (indexed by terrain metrics) and modification (a categorical factor delineating either natural or dredged estuaries) combine to shape the composition of fish assemblages in estuarine seascapes. These analyses were conducted within the mvabund package in R (Wang et al. 2012; Warton et al. 2012). ManyGLMs identify correlations between a matrix of multivariate abundance data and a suite of explanatory variables, and identify species that best explain assemblage-level correlations with best-fit variables (Warton et al. 2012). The Pearson’s correlation coefficient was used to test for co-linearity between terrain metrics prior to analysis (following Leitner et al. 2017; Rees et al. 2018). Subsequently, highly colinear (r2 = > 0.7) metrics including northness (correlated with eastness), profile curvature (correlated with plan curvature), rugosity (correlated with slope) and E2 hardness (correlated with E1 roughness) were removed from the analysis. Models tested for possible effects of variation in terrain relief (i.e. average depth), morphology (i.e. northness and plan curvature), complexity (i.e. slope) and composition (i.e. E1 roughness) on fish assemblages, and for interactions between each of these variables and modification (a categorical variable with two levels: natural and dredged). We also included latitude within these models to account for the large latitudinal extent of the study area, and tested for interactions between modification and percentage of urban shoreline, and the area of mangroves and seagrass, to test for the possible confounding effects of urbanisation and habitat availability on fish assemblages in natural and dredged seascapes. Best-fit models were selected using backwards stepwise regression, based on Akaike information criterion (AIC). The “p.uni” function within the mvabund package was then used to identify indicator species that were correlated with significant factors identified by the manyGLM model. Non-metric multidimensional scaling ordinations (nMDS) were then used to visualise effects of variables from the best-fit manyGLM on fish assemblages in natural and dredged estuaries. GLMs were then constructed in R, to further investigate the effects of variables from the best-fit manyGLM on species richness, harvestable fish abundance (i.e. the abundance of species targeted by commercial and/or recreational fishers) and the abundance of indicator species, and to test for variation in the terrain attributes, averaged across the entire sampling extent of each estuary, between natural and dredged seascapes. Harvested fish abundance was selected over total fish abundance because it has direct significance and relevance for coastal managers and spatial conservation planners (Weeks 2017; Brook et al. 2018; Goodridge Gaines et al. 2020). We also ran additional, and identical, manyGLM and generalised linear mixed models (GLMMs) to test for the consistent effects of dredging and terrain on the fish assemblage composition, species richness and harvested fish abundance across our study locations, by including estuary as a random factor using the glmmTMB package, and the “block’ function in mvabund, in R (Wang et al. 2012; Brooks et al. 2017). Multivariate and univariate GLMs and GLMMs were checked for homogeneity of variance, normally distributed residuals, outliers and over-dispersion, and were fitted with either Poisson or Negative Binomial (if models were over-dispersed) distributions, with log link functions. Species accumulation curves were also constructed for natural and modified seascapes using the “specaccum” function within the vegan package in R (Oksanen et al. 2019).
Results
Dredging has substantially altered the terrain of modified estuaries, which were deeper, had steeper sides (i.e. greater slope and plan curvature) and were comprised of substrates with lower roughness (i.e. lower E1 backscatter), than natural estuaries (p = ≤ 0.01; Tables S1 and S2). Natural and dredged estuaries also supported significantly different fish assemblages; fish abundance and species richness were both highest per site in dredged estuaries, but more species were found in natural estuaries overall (Fig. 2, Table 2). A total of 77 species (69 in natural, and 49 in dredged, estuaries), belonging to six functional groups (i.e. 19 piscivores; 6 omnivores; 42 zoobenthivores; 5 herbivores; 4 detritivores and 1 zooplanktivore) were recorded in this study (Table S3). The most abundant species recorded from natural estuaries were yellowfin bream (Acanthopagrus australis) (35%), estuary perchlet (Ambassis marianus) (15%), sea mullet (Mugil cephalus) (7%), striped scat (Selenotoca multifasciata) (4%) and sand whiting (Sillago cilliata) (3%) (Table S3). The most abundant species from dredged estuaries were yellowfin bream (40%), southern herring (Herklotsichthys castelnaui) (15%), estuary perchlet (15%), sea mullet (11%) and weeping toadfish (Torquigener pleurogramma) (3%) (Table S3).
The composition of fish assemblages was shaped by changes in the average depth of estuaries, and by variation in seafloor roughness (i.e. E1 backscatter) and steepness (i.e. plan curvature and slope), but the nature of these terrain effects differed between dredged and natural estuaries (Fig. 3, Tables 2 and S4). In natural estuaries, fish abundance was highest near convex (i.e. high plan curvature) terrain features that were steep (i.e. high slope) and soft (i.e. low roughness) (Fig. 4, Tables 3 and S5). By contrast, in dredged estuaries fish abundance was highest near convex terrain features (i.e. high plan curvature) that were hard (i.e. high roughness) and contained gentle slopes (i.e. low slope) (Fig. 4, Tables 3 and S5). Fish abundance and species richness was always highest near shallow terrain features (i.e. low average depth) in both natural and dredged estuaries (Fig. 4). The influence of dredging and terrain on fish assemblages were consistent across all study locations and were not modified by the random effects of estuary (Tables 2, 3, S4 and S5). No other variable (e.g. latitude, percentage of urban shoreline, aspect, mangrove area, seagrass area) was correlated with variation in the fish assemblage composition (Tables 2 and S4).
Non-metric multidimensional scaling ordinations (nMDS) visualising effects of terrain on fish assemblages in all (A), natural (B) and dredged (C) estuaries. Black lines illustrate the correlation of significant terrain metrics with variation in fish assemblage composition (identified by manyGLM analysis); and dotted lines illustrate correlations between indicator species and significant terrain metrics (identified by “p.uni” analysis). Text in bold illustrate significant interactions between terrain metrics and modification (i.e. a categorical factor delineating either natural or dredged estuaries), identified by the manyGLM analysis
Generalised linear models (GLMs) illustrating the effects of average depth, roughness, slope and plan curvature on species richness, harvested fish abundance and the abundance of significant representative species (identified by “p.uni” analysis) in natural and dredged estuaries. Shaded areas illustrate the 95% confidence intervals
Nine species were good indicators of the effects of seafloor terrain on the spatial distribution of estuarine fishes (Table 4). The abundance of three species (i.e. estuary glassfish, A. marianus; banded toadfish, Marilyna pleurosticta; and common toadfish, Tetractenos hamiltoni) was highest near convex (i.e. high plan curvature) terrain features in natural estuaries, and concave (i.e. low plan curvature) terrain features in dredged estuaries (Fig. 4, Table 4). By contrast, the abundance of estuary stingray (Hemitrygon fluviorum) was highest near concave (i.e. low plan curvature) terrain features in both dredged and natural estuaries (Fig. 4, Table 4). The abundance of common silverbiddy (Gerres subfasciatus) was highest near hard (i.e. high roughness) terrain features in dredged estuaries, and soft (i.e. low roughness) terrain features in natural seascapes (Fig. 4, Table 4). By contrast, the abundance of striped scat and common toadfish was highest near soft (i.e. low roughness) terrain features in both dredged and natural estuaries (Fig. 4, Table 4). The abundance of yellowfin bream was highest near steep (i.e. high slope) terrain features in natural seascapes, and gentle (i.e. low slope) terrain features in dredged estuaries (Fig. 4, Table 4). The abundance of two species (i.e. common silverbiddy and banded toadfish) were highest near deep (i.e. high average depth) terrain features, and the abundance of three species (i.e. yellowfin bream; sea mullet; and blue catfish, Neoarius graeffei) was highest near shallow (i.e. low average depth) terrain features, in both natural and dredged estuaries (Fig. 4, Table 4).
Discussion
Landscape modification alters the condition of ecosystems and the structure of terrain features, and this can lead to declines in biodiversity and ecosystem functioning (Heery et al. 2017; Berger-Tal and Saltz 2019). These negative consequences of fragmentation and urbanisation have been widely documented, both on land and in the sea (Fischer and Lindenmayer 2007; Bulleri and Chapman 2010), but the ecological consequences of terrain modification remain largely unknown (e.g. Rochette et al. 2010). Our findings show that the composition of fish assemblages in estuaries is tightly linked to the three-dimensional properties of the seafloor and demonstrate for the first time globally how dredging activities can fundamentally reshape the ecological significance of terrain features (Borland et al. 2021). Fish diversity and the abundance of harvested species was strongly correlated with variation in seafloor depth, plan curvature, roughness and slope, and were always highest in the shallow areas of estuaries, and adjacent to terrain features with convex sides (i.e. increased plan curvature). However, changes to the roughness (i.e. reduced backscatter) and steepness (i.e. increased slope) of modified estuaries from repeated dredging operations altered the way in which assemblages use prominent terrain features. In natural seascapes, harvested fish abundance and diversity were highest in areas with lower roughness and steeper slopes, whereas in dredged estuaries harvested fish abundance and diversity were highest in areas with higher roughness and gentler slopes. These findings highlight the significance of bathymetric variation for animal assemblages in estuaries and illustrate how the transformation of seafloor terrain from dredging can alter the ecological values of benthic habitats, and cause species to utilise fundamentally different terrain features, in modified seascapes (Todd et al. 2014; Macura et al. 2019; Borland et al. 2021).
It is widely accepted that coastal regions contain some of the most degraded landscapes and seascapes globally, and these impacts have been linked to changes in the productivity, functioning and ecological values of a diversity of ecosystems (Bishop et al. 2017; Heery et al. 2017). Coastal seascapes are transformed in many ways (e.g. shoreline hardening, urban development, dredging) and this typically results in declines in both the diversity and abundance of populations and assemblages, but some forms of modification also benefit generalist species that take advantage of newly created habitats (Waltham and Connolly 2011; Heery et al. 2018; Mayer-Pinto et al. 2018; Todd et al. 2019). Our results show that landscape modification alters the ecological value of terrain features for fish, but surprisingly both abundance and diversity were highest in dredged estuaries. Dredging simplifies landscape heterogeneity by removing structurally complex terrain features (e.g. subtidal rocky structures), and this typically has negative impacts on species diversity via the local extirpation of sensitive species, and through the redistribution of mobile taxa to less modified areas of urban landscapes (Rehitha et al. 2017; Piló et al. 2019). The homogenisation of coastal terrain features can, however, also favour species with diverse habitat requirements and broad diets (e.g. sea gulls, sea bream, polychaetes) (Fuirst et al. 2018; Piló et al. 2019; Henderson et al. 2020). These generalist taxa can rapidly colonise modified habitats, out-compete specialised species for resources, and thrive in modified landscapes and seascapes that also offer a reprieve from their natural predators (Ewers and Didham 2006; Estes et al. 2011; Henderson et al. 2020; Meng et al. 2020). The ecological effects of terrain reprofiling from dredging might, therefore, have a range of structural and functional consequences for biodiversity, food-webs and ecosystem health, not all of which are negative, and these should be considered when implementing conservation and restoration initiatives in modified seascapes (Fraser et al. 2017; Wenger et al. 2018).
The spatial distribution of animals in most landscapes is shaped by the composition and complexity of terrain features (Simonson et al. 2014; Pygas et al. 2020). Here, we demonstrate the significance of terrain variation for fish in estuarine seascapes, for the first time, and highlight the importance of changes in depth, roughness, slope and plan curvature for fish assemblages. This finding concurs with the results of research in other seascapes, which are characterised by deeper water and consolidated structure (i.e. coral and rocky reefs, continental shelves, the deep sea), and extends our understanding of the ecological significance of prominent terrain features into shallow coastal waters (Pittman et al. 2009; Smoliński and Radtke 2017; Oyafuso et al. 2017; Stamoulis et al. 2018; Borland et al. 2021). In most seascapes, fish diversity and abundance is linked to the presence of highly sloping, and rough, terrain features (e.g. pinnacles, canyons, reefs, seamounts) because these areas support a diversity of prey and afford heterogenous refugia from predators (Stamoulis et al. 2018; Weijerman et al. 2019). Fish also seek refuge within convex terrain features (e.g. mounds, undulations) that offer shelter from hydrodynamic activity and from the foraging activity of larger predators (Quattrini et al. 2012; Pirtle et al. 2017). Estuarine seascapes contain a diversity of terrain features that provide important foraging, refuge and spawning habitats for many fish species (Bradley et al. 2017; Henderson et al. 2019). Terrain variation is provided by a mosaic of deep channels, channel banks, rock bars, oyster and rocky reefs, and urban infrastructure, and also by changes in the architecture of low-profile ridges and depressions over intertidal flats (Becker et al. 2017; Bassett et al. 2018). Shallow sand and mud flats are important foraging grounds for zoobenthivores and benthic piscivores, like the banded toadfish, blue catfish, common silverbiddy and yellowfin bream we recorded, and provide nursery habitat for a diversity of juvenile fishes (Nicolas et al. 2007; Trimoreau et al. 2013; Pirtle et al. 2017). In this study, harvested fish abundance and diversity were negatively linked to the depth of terrain features. The significance of depth was consistent across both natural and modified estuaries but might be expected to change as estuarine seascapes become deeper, and more homogenous, and as the transition between intertidal and subtidal habitats become more abrupt, with increases in the ecological footprint of dredging operations (Rochette et al. 2010; Borland et al. 2021).
The modification of seafloor terrain in dredged estuaries changed how fish assemblages associated with benthic habitats, resulting in a greater harvested fish abundance and diversity in areas with higher roughness and gentler slopes. Dredging directly alters the morphology and complexity of the seafloor with the explicit intention of removing rough terrain features (e.g. rock bars and reefs with high backscatter) and creating deeper waterways for vessel passage (e.g. deep channels with steeply sloping sides) (Madricardo et al. 2019; Eidam et al. 2020). Seascapes that are frequently dredged are also characterised by consistently greater depths, higher turbidity, more extreme hydrodynamic conditions (e.g. stronger currents and larger waves), and increased boat traffic, which is associated with a greater frequency of collisions, and louder and more continuous noise (Todd et al. 2014; Wenger et al. 2017). The combined effects of these impacts change the way in which species use coastal habitats (Bilkovic 2011; Wakefield et al. 2013; Wenger et al. 2017; Ferrari et al. 2018), and we suggest that many taxa might utilise flats and sandbars with shallow depths and gentle slopes (i.e. the opposite of dredged channels) to avoid the diversity of stressors that are concentrated in the dredged channels of modified estuaries. The morphology of the seafloor is, however, also altered by each dredging event, with repetitive extraction decreasing the stability of softer benthic habitats and therefore improving the relative habitability of rougher terrain features (e.g. rock bars, reefs and rock-walls with high backscatter and curvature), which persist through dredging events (e.g. Wakefield et al. 2013). In this study, the abundance of three species (i.e. yellowfin bream, slope; silverbiddy, backscatter; estuary ray, curvature) were correlated with changes in the slope, roughness and curvature of the seafloor in dredged estuaries. The spatial distribution of these taxa is likely a response to the aforementioned effects of the cumulative stressors in dredged channels, but as all are zoobenthivores that feed in a range of habitats, it is also probably shaped by the diversity and abundance of epifaunal and infaunal invertebrates (e.g. molluscs, crustaceans, worms) that are common in the soft sediments, shallow rock bars and fringing urban structures of highly modified estuaries (Rehitha et al. 2017; Piló et al. 2019; Henderson et al. 2020).
Our findings provide the first empirical evidence that dredging fundamentally alters the ecological significance of three-dimensional terrain features in coastal seascapes. We show that variation in the relief, complexity, composition and morphology of terrain features shaped the composition of fish assemblages in both natural and dredged seascapes. Intensive dredging operations, however, directly altered these bathymetric properties of the seafloor and this changed how fish assemblages respond to variation in the relief, complexity and morphology of benthic habitats. Fish diversity and harvested fish abundance switched from being correlated with lower terrain roughness and steeper slopes in natural estuaries to being linked to higher roughness and gentler slopes in dredged estuaries. These contrasting relationships with terrain variation were completely opposite in natural and dredged estuaries, and suggest that the effects of landscape modification, and landform simplification from repeated dredging, might be pervasive in modified seascapes and likely extend well beyond the footprint of dredging operations. These results have important implications for urban planning and coastal management in a diversity of shallow coastal seascapes because they highlight the ecological significance of terrain variation for fish assemblages and demonstrate how the extraction and reprofiling of sediments can reshape how species utilise habitats within modified landscapes. Our results also suggest that urban planners and coastal managers should utilise bathymetric maps to describe the ecological consequences of dredging, and other human disturbances, that modify the three-dimensional properties of modified seascapes. The key challenge now is for researchers to identify the ecological and functional consequences of variation in the spatial characteristics (e.g. size, position, terrain modification) of dredged channels, and whether the effects of dredging and terrain differ between species size-classes and life-stages, and among nursery ecosystems. The ecological impacts of changes in seafloor terrain are, however, rarely considered in coastal management, and we suggest that their inclusion might result in a diversity of benefits ranging from improved species conservation outcomes to enhanced responses to ecosystem restoration, but highly modified seascapes might require different management actions to their natural counterparts.
Data availability
Data will be made available from the USC Research Bank.
Code availability
Not applicable.
References
Amorim E, Ramos S, Elliott M, Franco A, Bordalo AA (2017) Habitat loss and gain: Influence on habitat attractiveness for estuarine fish communities. Estuar Coast Shelf Sci 197:244–257
Arizaga J, Amat JA, Monge-Ganuzas M (2017) The negative effect of dredging and dumping on shorebirds at a coastal wetland in northern Spain. J Nat Conserv 37:1–7
Badgley C, Smiley TM, Terry R, Davis EB, DeSantis LR, Fox DL, Hopkins SS, Jezkova T, Matocq MD, Matzke N, McGuire JL, Mulch A, Riddle BR, Roth VL, Samuels JX, Strömberg CAE, Yanites BJ (2017) Biodiversity and topographic complexity: modern and geohistorical perspectives. Trends Ecol Evol 32(3):211–226
Bassett M, Lindholm J, Garza C, Kvitek R, Wilson-Vandenberg D (2018) Lingcod (Ophiodon elongatus) habitat associations in California: implications for conservation and management. Environ Biol Fish 101(1):203–213
Becker A, Whitfield AK, Cowley PD, Cole VJ (2017) Does water depth influence size composition of estuary-associated fish? Distributions revealed using mobile acoustic-camera transects along the channel of a small shallow estuary. Mar Freshw Res 68(11):2163–2169
Berger-Tal O, Saltz D (2019) Invisible barriers: anthropogenic impacts on inter- and intra-specific interactions as drivers of landscape-independent fragmentation. Philos Trans R Soc B. https://doi.org/10.1098/rstb.2018.0049
Bilkovic DM (2011) Response of tidal creek fish communities to dredging and coastal development pressures in a shallow-water estuary. Estuar Coasts 34(1):129–147
Bishop MJ, Mayer-Pinto M, Airoldi L, Firth LB, Morris RL, Loke LH, Hawkins SJ, Naylor LA, Coleman RA, Chee SY, Dafforn KA (2017) Effects of ocean sprawl on ecological connectivity: impacts and solutions. J Exp Mar Biol Ecol 492:7–30
Bolam SG, McIlwaine P, Garcia C (2021) Marine macrofaunal traits responses to dredged material disposal. Mar Pollut Bull 168:112412
Borja Á, Dauer DM, Elliott M, Simenstad CA (2010) Medium- and long-term recovery of estuarine and coastal ecosystems: patterns, rates and restoration effectiveness. Estuar Coasts 33(6):1249–1260
Borland HP, Gilby BL, Henderson CJ, Leon JX, Schlacher TA, Connolly RM, Pittman SJ, Sheaves M, Olds AD (2021) The influence of seafloor terrain on fish and fisheries: a global synthesis. Fish Fish 22(4):707–734
Bouchet PJ, Meeuwig JJ, Salgado Kent CP, Letessier TB, Jenner CK (2015) Topographic determinants of mobile vertebrate predator hotspots: current knowledge and future directions. Biol Rev 90(3):699–728
Bradley M, Baker R, Sheaves M (2017) Hidden components in tropical seascapes: deep-estuary habitats support unique fish assemblages. Estuar Coasts 40(4):1195–1206
Brook TW, Gilby BL, Olds AD, Connolly RM, Henderson CJ, Schlacher TA (2018) The effects of shoreline armouring on estuarine fish are contingent upon the broader urbanisation context. Mar Ecol Prog Ser 605:195–206
Brooks ME, Kristensen K, Van Benthem KJ, Magnusson A, Berg CW, Nielsen A, Skaug HJ, Machler M, Bolker BM (2017) glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R J 9:400
Bulleri F, Chapman MG (2010) The introduction of coastal infrastructure as a driver of change in marine environments. J Appl Ecol 47(1):26–35
Carse A, Lewis JA (2020) New horizons for dredging research: the ecology and politics of harbor deepening in the southeastern United States. Wires Water 7(6):e1485
Dafforn KA, Glasby TM, Airoldi L, Rivero NK, Mayer-Pinto M, Johnston EL (2015) Marine urbanization: an ecological framework for designing multifunctional artificial structures. Front Ecol Environ 13(2):82–90
Davies AB, Asner GP (2014) Advances in animal ecology from 3D-LiDAR ecosystem mapping. Trends Ecol Evol 29(12):681–691
Dobrowski SZ (2011) A climatic basis for microrefugia: the influence of terrain on climate. Glob Change Biol 17(2):1022–1035
Drabble R (2012) Monitoring of east channel dredge areas benthic fish population and its implications. Mar Pollut Bull 64(2):363–372
Eidam EF, Sutherland DA, Ralston DK, Dye B, Conroy T, Schmitt J, Ruggiero P, Wood J (2020) Impacts of 150 years of shoreline and bathymetric change in the Coos Estuary, Oregon, USA. Estuar Coasts. https://doi.org/10.1007/s12237-020-00732-1
Einoder LD, Southwell DM, Lahoz-Monfort JJ, Gillespie GR, Fisher A, Wintle BA (2018) Occupancy and detectability modelling of vertebrates in northern Australia using multiple sampling methods. PLoS ONE. https://doi.org/10.1371/journal.pone.0203304
Erftemeijer PL, Lewis RR 3rd (2006) Environmental impacts of dredging on seagrasses: a review. Mar Pollut Bull 52(12):1553–1572
Estes JA, Terborgh J, Brashares JS, Power ME, Berger J, Bond WJ, Carpenter SR, Essington TE, Holt RD, Jackson JBC, Marquis RJ, Oksanen L, Oksanen T, Paine RT, Pikitch EK, Ripple WJ, Sandin SA, Scheffer M, Schoener TW, Shurin JB, Sinclair ARE, Soulé ME, Virtanen R, Wardle DA (2011) Trophic downgrading of planet earth. Science 333(6040):301–306
Ewers RM, Didham RK (2006) Confounding factors in the detection of species responses to habitat fragmentation. Biol Rev Camb Philos Soc 81(1):117–142
Ferrari R, Malcolm HA, Byrne M, Friedman A, Williams SB, Schultz A, Jordan AR, Figueira WF (2018) Habitat structural complexity metrics improve predictions of fish abundance and distribution. Ecography 41(7):1077–1091
Fischer J, Lindenmayer DB (2007) Landscape modification and habitat fragmentation: a synthesis. Glob Ecol Biogeogr 16(3):265–280
Flaspohler DJ, Giardina CP, Asner GP, Hart P, Price J, Lyons CKA, Castaneda X (2010) Long-term effects of fragmentation and fragment properties on bird species richness in Hawaiian forests. Biol Conserv 143(2):280–288
Fraser MW, Short J, Kendrick G, McLean D, Keesing J, Byrne M, Caley MJ, Clarke D, Davis AR, Erftemeijer PLA, Field S, Gustin-Craig S, Huisman J, Keough M, Lavery PS, Masini R, McMahon K, Mengersen K, Rasheed M, Statton J, Stoddart J, Wu P (2017) Effects of dredging on critical ecological processes for marine invertebrates, seagrasses and macroalgae, and the potential for management with environmental windows using Western Australia as a case study. Ecol Ind 78:229–242
Fuirst M, Veit RR, Hahn M, Dheilly N, Thorne LH (2018) Effects of urbanization on the foraging ecology and microbiota of the generalist seabird Larus argentatus. PLoS ONE. https://doi.org/10.1371/journal.pone.0209200
Gilby BL, Olds AD, Yabsley NA, Connolly RM, Maxwell PS, Schlacher TA (2017a) Enhancing the performance of marine reserves in estuaries: JUST add water. Biol Conserv 210:1–7
Gilby BL, Olds AD, Connolly RM, Yabsley NA, Maxwell PS, Tibbetts IR, Schoeman DS, Schlacher TA (2017b) Umbrellas can work under water: Using threatened species as indicator and management surrogates can improve coastal conservation. Estuar Coast Shelf Sci 199:132–140
Gilby BL, Olds AD, Connolly RM, Maxwell PS, Henderson CJ, Schlacher TA (2018) Seagrass meadows shape fish assemblages across estuarine seascapes. Mar Ecol Prog Ser 588:179–189
Gladstone W, Lindfield S, Coleman M, Kelaher B (2012) Optimisation of baited remote underwater video sampling designs for estuarine fish assemblages. J Exp Mar Biol Ecol 429:28–35
Goodridge Gaines LA, Olds AD, Henderson CJ, Connolly RM, Schlacher TA, Jones TR, Gilby BL (2020) Linking ecosystem condition and landscape context in the conservation of ecosystem multifunctionality. Biol Conserv 243:108479
Grimm NB, Faeth SH, Golubiewski NE, Redman CL, Wu J, Bai X, Briggs JM (2008) Global change and the ecology of cities. Science 319(5864):756–760
Harvey ES, Cappo M, Butler JJ, Hall N, Kendrick GA (2007) Bait attraction affects the performance of remote underwater video stations in assessment of demersal fish community structure. Mar Ecol Prog Ser 350:245–254
Heery EC, Bishop MJ, Critchley LP, Bugnot AB, Airoldi L, Mayer-Pinto M, Sheehan EV, Coleman RA, Loke LH, Johnston EL, Komyakova V, Morris RL, Strain EMA, Naylor LA, Dafforn KA (2017) Identifying the consequences of ocean sprawl for sedimentary habitats. J Exp Mar Biol Ecol 492:31–48
Heery EC, Olsen AY, Feist BE, Sebens KP (2018) Urbanization-related distribution patterns and habitat-use by the marine mesopredator, giant Pacific octopus (Enteroctopus dofleini). Urban Ecosyst 21(4):707–719
Henderson CJ, Gilby BL, Schlacher TA, Connolly RM, Sheaves M, Flint N, Borland HP, Olds AD (2019) Contrasting effects of mangroves and armoured shorelines on fish assemblages in tropical estuarine seascapes. ICES J Mar Sci 76(4):1052–1061
Henderson CJ, Olds AD, Lee SY, Gilby BL, Maxwell PS, Connolly RM, Stevens T (2017) Marine reserves and seascape context shape fish assemblages in seagrass ecosystems. Mar Ecol Prog Ser 566:135–144. https://doi.org/10.3354/meps12048
Henderson CJ, Gilby BL, Schlacher TA, Connolly RM, Sheaves M, Maxwell PS, Flint N, Borland HP, Martin TS, Gorissen B, Olds AD (2020) Landscape transformation alters functional diversity in coastal seascapes. Ecography 43(1):138–148
Hossain S, Eyre BD, McKee LJ (2004) Impacts of dredging on dry season suspended sediment concentration in the Brisbane River estuary, Queensland, Australia. Estuar Coast Shelf Sci 61(3):539–545
Huijbers CM, Schlacher TA, Schoeman DS, Olds AD, Weston MA, Connolly RM (2015) Limited functional redundancy in vertebrate scavenger guilds fails to compensate for the loss of raptors from urbanized sandy beaches. Divers Distrib 21(1):55–63
Johnson MTJ, Munshi-South J (2017) Evolution of life in urban environments. Science. https://doi.org/10.1126/science.aam8327
Kowarik I (2011) Novel urban ecosystems, biodiversity, and conservation. Environ Pollut 159(8–9):1974–1983
Laundré JW, Hernández L, Ripple WJ (2010) The landscape of fear: ecological implications of being afraid. Open Ecol J. https://doi.org/10.2174/1874213001003030001]
Leitner AB, Neuheimer AB, Donlon E, Smith CR, Drazen JC (2017) Environmental and bathymetric influences on abyssal bait-attending communities of the Clarion Clipperton Zone. Deep Sea Res Part I 125:65–80
Lepczyk CA, Aronson MFJ, Evans KL, Goddard MA, Lerman SB, MacIvor JS (2017) Biodiversity in the city: fundamental questions for understanding the ecology of urban green spaces for biodiversity conservation. Bioscience 67(9):799–807
Lepczyk CA, Wedding LM, Asner GP, Pittman SJ, Goulden T, Linderman MA, Gang J, Wright R (2021) Advancing landscape and seascape ecology from a 2D to a 3D science. Bioscience. https://doi.org/10.1093/biosci/biab001
Li D, Tang C, Xia C, Zhang H (2017) Acoustic mapping and classification of benthic habitat using unsupervised learning in artificial reef water. Estuar Coast Shelf Sci 185:11–21
Macura B, Bystrom P, Airoldi L, Eriksson BK, Rudstam L, Stottrup JG (2019) Impact of structural habitat modifications in coastal temperate systems on fish recruitment: a systematic review. Environ Evid. https://doi.org/10.1186/s13750-019-0157-3
Madricardo F, Foglini F, Campiani E, Grande V, Catenacci E, Petrizzo A, Kruss A, Toso C, Trincardi F (2019) Assessing the human footprint on the sea-floor of coastal systems: the case of the Venice Lagoon, Italy. Sci Rep 9(1):6615
Marley SA, Salgado Kent CP, Erbe C (2017) Occupancy of bottlenose dolphins (Tursiops aduncus) in relation to vessel traffic, dredging, and environmental variables within a highly urbanised estuary. Hydrobiologia 792(1):243–263
Mayer-Pinto M, Cole VJ, Johnston EL, Bugnot A, Hurst H, Airoldi L, Glasby TM, Dafforn KA (2018) Functional and structural responses to marine urbanisation. Environ Res Lett. https://doi.org/10.1088/1748-9326/aa98a5
McCauley DJ, Pinsky ML, Palumbi SR, Estes JA, Joyce FH, Warner RR (2015) Marine defaunation: animal loss in the global ocean. Science. https://doi.org/10.1126/science.1255641
McLean KA, Trainor AM, Asner GP, Crofoot MC, Hopkins ME, Campbell CJ, Martin RE, Knapp DE, Jansen PA (2016) Movement patterns of three arboreal primates in a neotropical moist forest explained by LiDAR-estimated canopy structure. Landsc Ecol 31(8):1849–1862
Meng X, Chen J, Li Z, Liu Z, Jiang X, Ge Y, Cooper KM, Xie Z (2020) Degraded functional structure of macroinvertebrates caused by commercial sand dredging practices in a flood plain lake. Environ Pollut. https://doi.org/10.1016/j.envpol.2020.114415
Monk J, Ierodiaconou D, Versace VL, Bellgrove A, Harvey E, Rattray A, Laurenson L, Quinn GP (2010) Habitat suitability for marine fishes using presence-only modelling and multibeam sonar. Mar Ecol Prog Ser 420:157–174
Moore C, Drazen JC, Radford BT, Kelley C, Newman SJ (2016) Improving essential fish habitat designation to support sustainable ecosystem-based fisheries management. Mar Policy 69:32–41
Mosman JD, Henderson CJ, Olds AD, Gilby BL, Schlacher TA (2020) Seascape connectivity exerts differing effects for fish assemblages in distinct habitats of the surf zones of ocean beaches. ICES J Mar Sci. https://doi.org/10.1093/icesjms/fsaa018
Müller J, Brandl R (2009) Assessing biodiversity by remote sensing in mountainous terrain: the potential of LiDAR to predict forest beetle assemblages. J Appl Ecol 46(4):897–905
Munsch SH, Cordell JR, Toft JD (2017) Effects of shoreline armouring and overwater structures on coastal and estuarine fish: opportunities for habitat improvement. J Appl Ecol 54(5):1373–1384
Murphy HM, Jenkins GP (2010) Observational methods used in marine spatial monitoring of fishes and associated habitats: a review. Mar Freshw Res 61(2):236–252
Nicolas D, Le Loc’h F, Désaunay Y, Hamon D, Blanchet A, Le Pape O (2007) Relationships between benthic macrofauna and habitat suitability for juvenile common sole (Solea solea, L.) in the Vilaine estuary (Bay of Biscay, France) nursery ground. Estuar Coast Shelf Sci 73(3–4):639–650
Noriega R, Schlacher TA, Smeuninx B (2012) Reductions in ghost crab populations reflect urbanization of beaches and dunes. J Coast Res 28(1):123–131
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O’Hara RB, Simpson GL, Solymos P, Stevens MH et al (2019) Vegan: community ecology package. R package version 2.5–6. https://cran.r-project.org/web/packages/vegan/vegan.pdf. Accessed 10 Dec 2019
Olds AD, Connolly RM, Pitt KA, Maxwell PS (2012) Primacy of seascape connectivity effects in structuring coral reef fish assemblages. Mar Ecol Prog Ser 462:191–203
Olds AD, Frohloff BA, Gilby BL, Connolly RM, Yabsley NA, Maxwell PS, Henderson CJ, Schlacher TA (2018) Urbanisation supplements ecosystem functioning in disturbed estuaries. Ecography 41(12):2104–2113. https://doi.org/10.1111/ecog.03551
Osborne FE, Olds AD, Schlacher TA, Henderson CJ, Martin TS, Connolly RM, Maxwell PS, Gilby BL (2021) Human modifications to estuaries correlate with the morphology and functional roles of coastal fish. Mar Environ Res 170:105443
Oyafuso ZS, Drazen JC, Moore CH, Franklin EC (2017) Habitat-based species distribution modelling of the Hawaiian deepwater snapper-grouper complex. Fish Res 195:19–27
Pérez-Ruzafa A, García-Charton JA, Barcala E, Marcos C (2006) Changes in benthic fish assemblages as a consequence of coastal works in a coastal lagoon: the Mar Menor (Spain, Western Mediterranean). Mar Pollut Bull 53(1):107–120
Pickett ST, Cadenasso ML, Grove JM, Boone CG, Groffman PM, Irwin E, Kaushal SS, Marshall V, McGrath BP, Nilon CH, Pouyat RV, Szlavecz K, Troy A, Warren P (2011) Urban ecological systems: scientific foundations and a decade of progress. J Environ Manage 92(3):331–362
Piló D, Carvalho AN, Pereira F, Coelho HE, Gaspar MB (2019) Evaluation of macrobenthic community responses to dredging through a multimetric approach: effective or apparent recovery? Ecol Ind 96:656–668
Pirtle JL, Shotwell SK, Zimmermann M, Reid JA, Golden N (2017) Habitat suitability models for groundfish in the Gulf of Alaska. Deep-Sea Res Part II: Top Stud Oceanogr 165:303–321
Pittman SJ, Brown KA (2011) Multi-scale approach for predicting fish species distributions across coral reef seascapes. PLoS ONE 6(5):e20583
Pittman SJ, Costa BM, Battista TA (2009) Using lidar bathymetry and boosted regression trees to predict the diversity and abundance of fish and corals. J Coast Res 25(6):27–38
Pygas DR, Ferrari R, Figueira WF (2020) Review and meta-analysis of the importance of remotely sensed habitat structural complexity in marine ecology. Estuar Coast Shelf Sci 235:106468
Quattrini AM, Ross SW, Carlson MCT, Nizinski MS (2012) Megafaunal-habitat associations at a deep-sea coral mound off North Carolina, USA. Mar Biol 159(5):1079–1094
Rees MJ, Knott NA, Neilson J, Linklater M, Osterloh I, Jordan A, Davis AR (2018) Accounting for habitat structural complexity improves the assessment of performance in no-take marine reserves. Biol Conserv 224:100–110
Rehitha TV, Ullas N, Vineetha G, Benny PY, Madhu NV, Revichandran C (2017) Impact of maintenance dredging on macrobenthic community structure of a tropical estuary. Ocean Coast Manage 144:71–82
Rochette S, Rivot E, Morin J, Mackinson S, Riou P, Le Pape O (2010) Effect of nursery habitat degradation on flatfish population: application to Solea solea in the Eastern Channel (Western Europe). J Sea Res 64(1–2):34–44
Seto KC, Güneralp B, Hutyra LR (2012) Global forecasts of urban expansion to 2030 and direct impacts on biodiversity and carbon pools. Proc Natl Acad Sci USA 109(40):16083–16088
Simonson WD, Allen HD, Coomes DA (2014) Applications of airborne lidar for the assessment of animal species diversity. Methods Ecol Evol 5(8):719–729
Smoliński S, Radtke K (2017) Spatial prediction of demersal fish diversity in the Baltic Sea: comparison of machine learning and regression-based techniques. ICES J Mar Sci 74(1):102–111
Song XP, Hansen MC, Stehman SV, Potapov PV, Tyukavina A, Vermote EF, Townshend JR (2018) Global land change from 1982 to 2016. Nature 560(7720):639–643
Stamoulis KA, Delevaux JM, Williams ID, Poti M, Lecky J, Costa B, Kendall MS, Pittman SJ, Donovan MK, Wedding LM, Friedlander AM (2018) Seascape models reveal places to focus coastal fisheries management. Ecol Appl 28(4):910–925
Strain EM, Olabarria C, Mayer-Pinto M, Cumbo V, Morris RL, Bugnot AB, Dafforn KA, Heery E, Firth LB, Brooks PR, Bishop MJ (2018) Eco-engineering urban infrastructure for marine and coastal biodiversity: which interventions have the greatest ecological benefit? J Appl Ecol 55(1):426–441
Teichert N, Borja A, Chust G, Uriarte A, Lepage M (2016) Restoring fish ecological quality in estuaries: implication of interactive and cumulative effects among anthropogenic stressors. Sci Total Environ 542:383–393
Todd VLG, Todd IB, Gardiner JC, Morrin ECN, MacPherson NA, DiMarzio NA, Thomsen F (2014) A review of impacts of marine dredging activities on marine mammals. ICES J Mar Sci 72(2):328–340
Todd PA, Heery EC, Loke LHL, Thurstan RH, Kotze DJ, Swan C (2019) Towards an urban marine ecology: characterizing the drivers, patterns and processes of marine ecosystems in coastal cities. Oikos 128(9):1215–1242
Trimoreau E, Archambault B, Brind’Amour A, Lepage M, Guitton J, Le Pape O (2013) A quantitative estimate of the function of soft-bottom sheltered coastal areas as essential flatfish nursery habitat. Estuar Coast Shelf Sci 133:193–205
Wakefield CB, Lewis PD, Coutts TB, Fairclough DV, Langlois TJ (2013) Fish assemblages associated with natural and anthropogenically-modified habitats in a marine embayment: comparison of baited videos and opera-house traps. PLoS ONE. https://doi.org/10.1371/journal.pone.0059959
Walbridge S, Slocum N, Pobuda M, Wright JD (2018) Unified geomorphological analysis workflows with Benthic Terrain Modeler. Geosciences 8(3):94–117
Waltham NJ, Connolly RM (2011) Global extent and distribution of artificial, residential waterways in estuaries. Estuar Coast Shelf Sci 94(2):192–197
Wang Y, Naumann U, Wright ST, Warton DI (2012) mvabund– an R package for model-based analysis of multivariate abundance data. Methods Ecol Evol 3(3):471–474
Warton DI, Wright ST, Wang Y (2012) Distance-based multivariate analyses confound location and dispersion effects. Methods Ecol Evol 3(1):89–101
Weeks R (2017) Incorporating seascape connectivity in conservation prioritisation. PLoS ONE. https://doi.org/10.1371/journal.pone.0182396
Weijerman M, Grüss A, Dove D, Asher J, Williams ID, Kelley C, Drazen JTC (2019) Shining a light on the composition and distribution patterns of mesophotic and subphotic fish communities in Hawaii. Mar Ecol Prog Ser 630:161–182
Wenger AS, Harvey E, Wilson S, Rawson C, Newman SJ, Clarke D, Saunders BJ, Browne N, Travers MJ, Mcilwain JL, Erftemeijer PLA, Hobbs JPA, Mclean D, Depczynski M, Evans RD (2017) A critical analysis of the direct effects of dredging on fish. Fish Fish 18(5):967–985
Wenger AS, Rawson CA, Wilson S, Newman SJ, Travers MJ, Atkinson S, Browne N, Clarke D, Depczynski M, Erftemeijer PL, Evans RD, Hobbs J-PA, McIlwain JL, McLean DL, Saunders BJ, Harvey E (2018) Management strategies to minimize the dredging impacts of coastal development on fish and fisheries. Conserv Lett. https://doi.org/10.1111/conl.12572
Wraith J, Lynch T, Minchinton TE, Broad A, Davis AR (2013) Bait type affects fish assemblages and feeding guilds observed at baited remote underwater video stations. Mar Ecol Prog Ser 477:189–99. https://doi.org/10.3354/meps10137
Yabsley NA, Gilby BL, Schlacher TA, Henderson CJ, Connolly RM, Maxwell PS, Olds AD (2020) Landscape context and nutrients modify the effects of coastal urbanisation. Mar Environ Res. https://doi.org/10.1016/j.marenvres.2020.104936
Acknowledgements
Funding for this project was provided by the Queensland Department of Agriculture and Fisheries (DAF1498CQ8-3), Healthy Land and Water and the Sea World Research and Rescue Foundation. The authors would like to thank Felicity Osborne, Olivia Kimber, Jesse Mosman, Sarah Thackwray and Nicholas Yabsley for their help in the field and laboratory.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions.
Author information
Authors and Affiliations
Contributions
HB, BLG, CH and AO conceived the research project and designed the study; BLG, RC, MS and AO resourced the project; HB, CH, BLG, BG, NO, AR and AO collected the data; HB, BLG, AR and AO analysed the data; HB, CH, BLG, RC, BG, NO, AR, SP, MS and AO interpreted results and compiled the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Borland, H.P., Gilby, B.L., Henderson, C.J. et al. Dredging fundamentally reshapes the ecological significance of 3D terrain features for fish in estuarine seascapes. Landsc Ecol 37, 1385–1400 (2022). https://doi.org/10.1007/s10980-021-01394-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-021-01394-5