Abstract
Metapopulation models typically assume that suitable habitats occupied by local populations and unsuitable matrix separating them form a ‘black-and-white’ landscape mosaic, in which dispersal is primarily determined by the spatial configuration of habitat patches. In reality, however, the matrix composition is also likely to influence dispersal. Using intensive mark-recapture surveys we investigated inter-patch movements in Maculinea (Phengaris) nausithous and M. teleius occurring sympatrically in six metapopulations. Three of these metapopulations had the matrix dominated by forest, an inhospitable environment for grassland butterflies, whereas in the remaining three the matrix was mostly composed of open environments. Dispersal parameters derived with the Virtual Migration model revealed significant differences between both groups of metapopulations. Both species had a lower propensity to emigrate from their natal habitat patches, and they suffered substantially higher dispersal mortality in the metapopulations with forest matrix. On the other hand, mean dispersal distances were roughly an order of magnitude longer in forest matrix as compared with open landscapes (ca. 500–1,500 vs. 100–200 m). Our results suggest that inhospitable forest matrix induces strong selection against dispersal, leading to a reduced emigration rate. At the same time, the selection may promote emigrants with good dispersal abilities, which are able to perform long-distance movements. Thus, while it is generally believed that a matrix structurally similar to the habitat of a species should improve the functional connectivity of habitat patches, our findings imply that this may not necessarily be the case.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Dispersal is a key process for species survival in fragmented landscapes (Fahrig 2003; Bowne and Bowers 2004; Reed 2004). It enables gene flow, preventing local populations from genetic variability loss due to genetic drift and inbreeding (O’Grady et al. 2006; Hanski 2009; Lowe and Allendorf 2010). It also provides rescue effects for declining populations, and it makes possible colonisation of vacant habitat patches, thus ensuring metapopulation persistence despite occasional extinctions of local populations (Hanski et al. 1996; Gonzalez et al. 1998; Poethke et al. 2003). The role of dispersal in metapopulation functioning has been investigated in countless modelling studies (Hanski 1999; Clobert et al. 2004; Bowler and Benton 2005).
In most metapopulation models landscape is perceived as a ‘black and white’ mosaic of suitable habitat patches and unsuitable environment separating them, called matrix (e.g. Hanski 1994; With and King 1999; Zollner and Lima 1999). Consequently, the exchange of individuals among local populations is assumed to be a function of the dispersal traits of a given species and the spatial configuration of their habitat patches versus the matrix. Only recently more attention has been paid to the fact that matrix can be highly variable and its character is likely to affect dispersal as well (Prevedello and Vieira 2010; Eycott et al. 2012; Zeller et al. 2012). In particular, differences in dispersal patterns may depend on whether environments forming the matrix are structurally similar to the habitats of a species (hereafter termed hospitable matrix for the sake of brevity) or clearly distinct from them (inhospitable matrix).
In the case of grassland butterflies major differences may be expected between dispersal in forests and in open environments (Sutcliffe and Thomas 1996; Roland et al. 2000; Ricketts 2001). The latter may include meadows of other types than required by a species or simply lacking essential resources (especially foodplants), but also fallow lands, agricultural fields, road margins or even low density residential areas. When moving through such environments grassland butterflies encounter conditions fairly similar to those experienced within their natal habitat patches. In contrast, flying through forest involves facing conditions that are strikingly different in several aspects. The most obvious distinction is that there is little direct sunlight, which alone may discourage many individuals from entering forest and affect the flight activity of those that have ventured into moving through forest (Kingsolver 1985; Dreisig 1995). Moreover, compared with open environments forests are typically characterised by the low availability of nectar plants, which serve as essential sources of energy utilised for flight by many butterfly species (Brown and Chippendale 1974; Sacktor 1975; Kammer and Heinrich 1978).
Since the aforementioned effects on mobility are predominantly negative, one may predict reduced dispersal of grassland butterflies in forested landscapes. We tested the above prediction by comparing dispersal parameter estimates, including emigration rate, mean dispersal distance, and dispersal mortality, among several metapopulations with either mostly open matrix and or mostly forested one. As model organisms we used two species of specialist grassland butterflies, Maculinea (=Phengaris) nausithous and M. teleius, the metapopulations of which were intensively surveyed with mark-recapture methods.
Methods
Study species and their field surveys
Maculinea are highly specialised myrmecophilous butterflies, requiring specific foodplants and specific Myrmica host ants to complete their life cycle (Thomas 1995). The host ants are typically scarce but widely distributed, while the foodplants are highly abundant but occur in patches, which can thus be regarded as Maculinea habitat patches (Nowicki et al. 2005c, 2007; Anton et al. 2008). Consequently, Maculinea often form classic metapopulation systems (Nowicki et al. 2007; Dierks and Fischer 2009; but see Nowicki et al. 2009). M. nausithous and M. teleius typically occur sympatrically in wet meadows, sharing the same larval foodplant, Sanguisorba officinalis, which is also a primary nectar source for their adults (Elmes and Thomas 1992; Thomas 1995). Other nectar plants occasionally used by both species, including Vicia cracca, Betonica officinalis, Cirsium arvense, and Veronica longifolia (Thomas 1984; Sielezniew and Stankiewicz-Fiedurek 2013; authors’ unpubl. data) grow commonly within grasslands and fallow lands, but very rarely occur in forests.
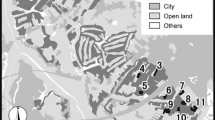
Both species were surveyed with mark-recapture methods in six metapopulations located in the Czech Republic, Germany, Poland, and Slovenia (Fig. 1). Butterflies were captured with entomological nets, individually marked with numbers written on the underside of their hind wing using permanent markers, and immediately released at the place of capture. For each capture we recorded the butterfly number, its species and sex, as well as the habitat patch number. The sampling was conducted daily to every second day (with few gaps due to unfavourable weather) between 9:00 and 17:00. Sampling intensity on particular habitat patches was adjusted to their area and butterfly numbers in order to ensure uniform capture probabilities across all the patches within a particular metapopulation.
In each metapopulation an intensive survey, in which mark-recapture sampling was conducted at a large number of habitat patches throughout the entire flight period, i.e. roughly from early July to late August, was performed in 1 year which was different for each metapopulation (see Table 1). Although mark-recapture studies were also carried out in the investigated metapopulations in other years, they were not comprehensive enough for dispersal analysis, because they were limited to too few local populations (cf. Stettmer et al. 2001; Nowicki et al. 2013). A clear exception in this respect was the metapopulation near the Czech town of Přelouč, which was intensively surveyed for seven consecutive years (Nowicki and Vrabec 2011). However, except for 1 year these surveys were restricted to the relatively small core fragment of the metapopulation. Thus for the sake of consistency in the spatial extent of the investigated metapopulations, we have not included these surveys in the present analysis, especially that the Přelouč metapopulation was the smallest.
The information about the study sites is summarised in Table 1. The investigated metapopulations clearly differed in their matrix composition. In three of them (Přelouč, Kraków, Teisendorf) the matrix consisted predominantly of open lands, including meadows, fallow lands and fields. The remaining three (Dečin, Steigerwald, Slovenske Gorice hereafter Gorice) had a matrix dominated by forest. The proportion of forest within the minimum convex polygons encompassing all the Maculinea habitat patches in a particular metapopulation was below 20 % in the former group, while in the latter it reached ca. 50–70 %. We note that the proportion of forest was little changed if we extended the minimum convex polygons to include 500-m or 1-km buffer zones around each habitat patch. Obviously, a high proportion of forest in the matrix does not necessarily imply that dispersing butterflies often need to cross forest fragments, if most of them are located in marginal parts of a metapopulation. Hence, in order to account not only for the amount of forest in the matrix, but also for its location versus habitat patches, we calculated the proportions of forest along cross sections of the lines linking the centres of habitat patches in each metapopulation. Nevertheless, the results were almost identical to the proportions of forest in the matrix area (Table 1).
Other inhospitable landuse types, such as urban areas and waters (rivers and lakes), had a consistently low proportion of the matrix area, reaching only a few percent at all the study sites. The investigated metapopulations had fairly similar spatial dimensions, which corresponded to similar ranges of potential inter-patch movement distances, except for the Přelouč one, which was approximately half the size (Fig. 1; Table 1). Moreover, habitat patch sizes were also comparable across all the metapopulations, although very large patches, exceeding 10 ha, existed only in Kraków and Steigerwald (Table 1).
In an earlier study we found that dispersal within metapopulations of Maculinea butterflies is negatively affected by their strong spatial isolation (Bonelli et al. 2013). We also demonstrated positive density-dependence of emigration rate (but not of other dispersal parameters), leading to its sharp increase at densities exceeding carrying capacity (Nowicki and Vrabec 2011). In this context, it is important to stress that none of the investigated metapopulations was strongly isolated, with neighbouring metapopulations being located 2–4 km away in each case (Table 1). Such isolation distances are close to the maximum movement distances recorded for Maculinea butterflies (Nowicki et al. 2005b; see also the “Results” section). Consequently, inter-metapopulation movements are likely to occur, but only sporadically, and thus it is valid to restrict dispersal analyses to the investigated metapopulations (cf. Bonelli et al. 2013). Besides, the available data from other years indicate that in the years used in the present study butterfly abundances in all the metapopulations were at their normal levels below carrying capacities. Consequently, neither of the aforementioned effects is likely to influence our estimates of dispersal parameters.
Dispersal analysis
In the original data sets many land fragments covered with S. officinalis foodplants were regarded as separate habitat patches based on, for example, different land ownership, even though they were directly adjacent to each other (cf. Hovestadt et al. 2011). Thus, to ensure that habitat patches are defined in a uniform way across all the metapopulations, for the purpose of dispersal analysis we have pooled together all the patches that were separated by less than 50 m. The 50-m threshold was adopted after Nowicki et al. (2007), who found that such a distance is enough make local populations of Maculinea butterflies demographically independent. Pooling together closely lying patches also allowed disregarding short-distance movements between them, which are likely to represent daily routine flights rather than genuine dispersal (Hovestadt et al. 2011).
The mark-recapture data collected were analysed with the Virtual Migration (VM) model (Hanski et al. 2000), which is a well-established standard for investigating dispersal in metapopulations. Since the rationale and a detailed description of the model have been provided elsewhere (Hanski et al. 2000; Petit et al. 2001), in the present paper we only briefly outline its features. Dispersal within a metapopulation is modelled using six parameters, which include: (i) mortality in habitat patches (μp); (ii) emigration propensity (η), defined as daily emigration rate scaled to 1 ha patch; (iii) emigration scaling with patch area (ζem,); (iv) immigration scaling with target patch area (ζim); (v) scaling of dispersal mortality with natal patch connectivity (λ); and (vi) distance dependence of dispersal (α).
Mortality in habitat patches is independent of dispersal and constrained to be constant across all the patches within a metapopulation. Emigration propensity reflects the emigration rate scaled to an imaginary 1-ha patch. Both emigration and immigration are assumed to depend on patch area (A j ), with the power relationship being negative for emigration (\( E_{j} = \eta A_{j}^{{\zeta_{\text{em}} }} \), where ζem < 0, η represents daily emigration rate from 1 ha patch) and positive for immigration (\( I_{j} \sim A_{j}^{{\zeta_{\text{em}} }} \), where ζim > 0). The probability of successful dispersal (dispersal survival, φmj ) is modelled to increase sigmoidally with the natal patch connectivity (S j , defined as in Hanski 1994): \( \varphi_{{{\text{m}}j}} = {{S_{j}^{2} } \mathord{\left/ {\vphantom {{S_{j}^{2} } {(\lambda + S_{j}^{2} )}}} \right. \kern-0pt} {(\lambda + S_{j}^{2} )}} \). The square root of λ is thus the equivalent of patch connectivity, for which half of emigrants from the patch die during dispersal. The α parameter defines the dispersal kernel.
We opted for the negative exponential function (NEF) as the kernel (as in Hanski et al. 2000), in which mean dispersal distance (measured in km) corresponds to 1/α, as it was found to describe movements of Maculinea butterflies quite well in previous studies (Hovestadt and Nowicki 2008; Nowicki and Vrabec 2011). Nevertheless, the estimates of all other VM model parameters changed hardly at all, when we attempted the inverse power function (IPF), preferred as the kernel by some authors (Schtickzelle et al. 2006; Fric et al. 2010). The inter-patch movement distances used for fitting the kernel were measured between centres of patches, which is a standard approach (Hanski et al. 2000; Matter et al. 2005; Hovestadt et al. 2011). Alternative solutions, such as applying edge-to-edge distances or dividing centre-to-centre distances into within-patch and within-matrix fractions, for which separate kernels are fitted (see Matter et al. 2004), would in fact change very little as patch dimensions were typically small as compared with inter-patch distances.
The analysis was conducted using the VM2 program (Hanski et al. 2000). Its goodness-of-fit tests indicated that the VM model fitted our mark-recapture data well. The observed numbers of emigrants, immigrants, and residents were not significantly different from those predicted by the model, except for the small number of patches with few (<10) captures recorded. However, low numbers of captures are well known to bias the goodness-of-fit tests toward more significant values (Schtickzelle et al. 2006).
The VM2 program allows the estimation of the VM model parameters together with their 95 % confidence intervals. The parameter estimates are expressed in uniform units and thus they can be used for comparisons between metapopulations, with non-overlapping 95 % confidence intervals indicating statistically significant differences between the estimates (Schtickzelle and Baguette 2003; Schtickzelle et al. 2006). The accuracy of the model estimates is not affected by sample size, though their precision may be reduced in the case of a small sample (Nowicki and Vrabec 2011). It must be stressed that while the estimation of the VM model parameters requires spatial information (area and location) for all the habitat patches within a metapopulation, not all of them need to be sampled with mark-recapture. It is enough that the sampling has been conducted in at least ca. 10 patches (Hanski et al. 2000; Petit et al. 2001), which was the case in our metapopulations (Table 1). We derived the parameter estimates separately for both species in each metapopulation. In addition, we calculated the weighted means and their confidence intervals (Sokal and Rohlf 2012) for both species in ‘open-land matrix’ metapopulations (Přelouč, Kraków, Teisendorf) and ‘forest matrix’ metapopulations (Dečin, Gorice, Steigerwald), with weights being the numbers of butterfly captures.
Results
Altogether, we recorded 4,287 M. nausithous and 2,700 M. teleius individuals, which were captured 6,318 and 4,015 times respectively. The sample sizes for particular metapopulations are given in Table 2. Except for the Kraków region, in which M. teleius captures prevailed, in all the other metapopulations M. nausitous was more abundant, which leads to a better precision of the VM model estimates derived for the latter species (Fig. 2).
Comparison of the estimates of the Virtual Migration model parameters (presented with 95 % confidence intervals) derived for the investigated metapopulations of Maculinea butterflies: solid squares M. nausithous; empty squares M. teleius. In both species the weighted means obtained for metapopulations with open landscape matrix and those with forest-dominated matrix were significantly different (P < 0.05) in all the parameters except for emigration and immigration scalings. Note the logarithmic scale used in the case of dispersal mortality scaling; zeros were depicted in place of 0.001 values for the sake of presentation clarity
It is noteworthy that the estimates of the three main dispersal parameters of interest, i.e. emigration rate, dispersal distance, and dispersal-related mortality, were highly repeatable among the three metapopulations with open-land matrix as well as among the three metapopulations with predominantly forest matrix. Emigration propensity reached 8–12 % individuals per day in the former group of metapopulations, whereas it was only 3–4 %, i.e. approximately three times lower, in the latter group (Fig. 2b).
High proportions of forest in the matrix also resulted in greatly increased mortalities of dispersing butterflies. Although poor precision of dispersal mortality scaling estimates (a common problem with this parameter, which is the most difficult one to derive with the VM model) did not allow detecting statistically significant differences for any particular metapopulation, the weighted means for the open-land matrix metapopulations and those with the matrix dominated by forest proved to be significantly different (Fig. 2e). Moreover, in absolute terms the differences were quite strong (Fig. 3). The mortality scaling estimates for the open landscapes were hardly (if at all) above zero, indicating the proportions of unsuccessful dispersers were negligible and reached at most 5 % in M. nausithous and 8 % in M. teleius (in the Kraków metapopulation). In turn, with the connectivity of local habitat patches accounted for, the parameter values obtained for the metapopulations with highly forested matrix corresponded to the overall disperser mortality of 28 % (in the case of M. teleius in Gorice) to even 43 % (in the case of M. teleius in Steigerwald).
The most prominent results of our analysis were those concerning dispersal distances. Somewhat in contrast with the reduced emigration and the increased dispersal-related mortality, we found that the mean inter-patch movement distances (1/α of Fig. 2d) covered by both Maculinea species in the highly forested regions were almost an order of magnitude longer than those recorded in the open landscapes (ca. 500–1,500 vs. ca. 100–200 m). Similarly, while in the open landscapes the maximum observed movement distances were 960 m for M. teleius (in Kraków) and 1,007 m for M. nausithous (in Přelouč), in the highly forested regions we recorded altogether 18 and 58 over 1-km movements for the two species, with the longest ones reaching 2.94 and 5.03 km respectively. Interspecific comparisons revealed that M. nausithous appeared to be slightly more mobile than M. teleius in the open landscapes, as indicated by consistently (though not significantly) higher emigration rates and longer movement distances, but not in the highly forested regions.
Mortality within habitat patches turned out to be significantly higher in the open-land metapopulations than in those with highly forested matrix, when the weighted mean values were compared (Fig. 2a). However, the difference in fact stemmed from particularly high values recorded for both species in a single metapopulation, namely the Kraków one, which in this respect was clearly distinct from all the others. More importantly, the estimates for both species were highly concordant (Pearson’s r = 0.814; df = 4; P = 0.0489), implying that within-patch mortality experienced by the investigated butterflies was presumably influenced by factors specific to each metapopulation. There were no clear patterns in both emigration and immigration scaling parameters in relation to matrix composition (Fig. 2c, f). Although emigration scaling estimates appeared to be generally lower (but note their relatively wide 95 % confidence intervals) in the metapopulations with highly forested matrix, this is understandable since the effect of patch area on emigration becomes hard to detect when there is little emigration.
Discussion
Our findings clearly demonstrate that dispersal within metapopulations of Maculinea butterflies is affected by matrix composition. The principal dispersal parameters, i.e. emigration rate, mean dispersal distance, and dispersal-related mortality, clearly differed between open landscapes and highly forested ones. Even though applying a rigorous experimental design is not possible in large-scale ecological research (Oksanen 2004; Schtickzelle et al. 2006), the reliability of our results is enhanced by the high repeatability of the parameter estimates obtained in both landscape types. In turn, non-dispersal mortality, which translates into life expectancy of adult butterflies living in their habitat patches, was most likely shaped by case-specific factors. We believe that the underlying reason was the variation in weather patterns among the investigated metapopulations, which is a typical driver of butterfly life expectancy (Casula and Nichols 2003; Nowicki et al. 2009; Matter et al. 2011). Interestingly, however, distinctively short adult life expectancies were recorded in earlier studies on M. teleius and M. nausithous in the region (Nowicki et al. 2005a, b), which implies that they may either be heritable traits or reflect less favourable climatic conditions.
The documented differences in the proportions of unsuccessful dispersers demonstrate that forest matrix induces high dispersal mortality (Fig. 3). Admittedly, when applying the Virtual Migration model one cannot distinguish emigration outside the study area from dispersal mortality. Due to relatively long dispersal distances in forest matrix it is possible that some emigrants successfully moved to neighbouring localities. However, the proportions of emigrants that are likely to reach other metapopulations are very low. The VM model simulations based on the dispersal kernel estimates and the distances separating the metapopulations revealed that they should not exceed 5–6 %. Hence, although our estimates of dispersal mortality may be positively biased, the biases are slight only, and with these biases accounted for, the dispersal mortality in forest matrix still greatly exceeds the levels estimated for open landscapes. Consequently, it may be expected that forest matrix leads to a strong selection against dispersing individuals.
The decrease in emigration rate in highly forested regions is in perfect agreement with the above prediction. The proximate mechanism behind the reduced emigration is probably butterfly tendency to avoid crossing habitat patch edges bordered by forest. Although Maculinea butterflies do not refrain from the edges of their habitat patches, and may even prefer to use the edges (Batáry et al. 2009; Kőrösi et al. 2012; Nowicki et al. 2013), they have been found to avoid crossing the edges, especially high contrast ones. Among various types of patch edges examined by Skórka et al. (2013), forest ones were crossed the least frequently by M. teleius. The avoidance of crossing high contrast edges between meadows and forests was also observed for several other butterfly species (Sutcliffe and Thomas 1996; Ries and Debinski 2001; Ross et al. 2005; Schultz et al. 2012).
The negative impact of inhospitable forest matrix on the numbers of meadow butterflies that undertake dispersal is not surprising. The phenomenon that animals are less likely to leave their patches if the adjacent environment is structurally dissimilar to the patch habitat was reported in many species representing various taxons (see review by Eycott et al. 2012). Consequently, it is believed that matrix resembling the species’ habitat enhances functional connectivity of habitat patches in fragmented landscapes (Prevedello and Vieira 2010). However, our results concerning the inter-patch movement distances of Maculinea butterflies do not support the above statement.
The mean dispersal distances in highly forested regions were several times longer than those recorded in open landscapes. Such hugely increased dispersal distances in inhospitable matrix may seem to be in sharp contrast to the predicted selection against dispersive individuals. On the other hand, it is reasonable to expect that such a selection would be a disruptive one (sensu Rueffler et al. 2006): it should act primarily against poor dispersers that undertake emigration, whereas both individuals showing strong fidelity to their patches as well as emigrants with good dispersal abilities should be favoured. Ultimately, such conditions could even result in the selection of rare types of individuals specifically fit to carry the risks of dispersal through inhospitable matrix (Roff 1994; Fronhofer et al. 2011). The evidence for the existence of two different classes of movements in Maculinea butterflies has been found by Hovestadt et al. (2011).
Nevertheless, the substantial increase in inter-patch movement distances cannot be fully attributed to the fact that dispersal is performed only by the individuals best adapted to it. Since we found roughly a threefold decrease in emigration in the forested landscapes as compared with the open ones, it can be responsible for at most a similar increase in dispersal distances, while the estimated increase reached almost an order of magnitude. Furthermore, while the increase in dispersal distances as a result of selective pressure is an attractive hypothesis, and in our opinion, quite a plausible one, our study alone is far from being enough to prove it. Without a translocation experiment, which would confirm that butterflies originating from highly forested landscapes maintain long dispersal distances even when moved to open matrix regions, one has to assume that the pattern we have observed may equally well derive from purely behavioural mechanisms, namely different movement rules in forests and open lands. A possible explanation may be the fact that butterflies moving through inhospitable matrix tend to fly continuously and follow relatively straight paths, rather than to perform short and tortuous flights as they do within habitat patches and similar environments (Schultz 1998; Schtickzelle et al. 2007; Kuefler et al. 2010; Skórka et al. 2013). A particularly interesting illustration of the above pattern is the study by Kuefler et al. (2010), who found that the displacement rate of the wet forest butterfly, Satyrodes appalachia, was fastest in open environments, where the flights were the longest and straightest, and the slowest within forests due to short and sinuous movements. At first glance the findings of Kuefler et al. (2010) and our findings appear to be conflicting, but actually they jointly indicate that butterfly movement routines are determined by the structural similarity of matrix to the species’ habitat rather than by the matrix structure alone.
In recent years there has been a growing number of studies focused on assessing permeability (or its inverse, i.e. resistance) of various matrix types to movements of animal species of interest (see reviews by Kindlman and Burel 2008; Zeller et al. 2012). They typically attempt to quantify this property with a single metric. The outcome of our study implies that it may be a conceptually flawed approach, because the suitability of a matrix type for animal dispersal is a two-dimensional property, reflecting both the probability of entering the matrix type (which affects emigration rate) and the ease and/or motivation to move through it (which affects dispersal distances). Moreover, there is probably a negative relationship between the matrix characteristics that promote emigration and those enhancing dispersal distances (see also Kuefler et al. 2010). Consequently, both aspects of dispersal, i.e. how many individuals disperse and how far they get, as well as the factors influencing them need to be evaluated separately in the assessments of functional connectivity within metapopulations.
References
Anton C, Musche M, Hula V, Settele J (2008) Myrmica host-ants limit the density of the ant-predatory large blue Maculinea nausithous. J Insect Conserv 12:511–517
Batáry P, Kőrösi A, Örvössy N, Köver S, Peregovits L (2009) Species-specific distribution of two sympatric Maculinea butterflies across different meadow edges. J Insect Conserv 13:223–230
Bonelli S, Vrabec V, Witek M, Barbero F, Patricelli D, Nowicki P (2013) Selection on dispersal in isolated butterfly metapopulations. Popul Ecol 55:469–478
Bowler DE, Benton TG (2005) Causes and consequences of animal dispersal strategies: relating individual behaviour to spatial dynamics. Biol Rev 80:205–225
Bowne DR, Bowers MA (2004) Interpatch movements in spatially structured populations: a literature review. Landscape Ecol 19:1–20
Brown JJ, Chippendale GM (1974) Migration of the monarch butterfly, Danaus plexippus: energy sources. J Insect Physiol 20:1117–1130
Casula P, Nichols JD (2003) Temporal variability of local abundance, sex ratio and activity in the Sardinian chalk hill blue butterfly. Oecologia 136:374–382
Clobert J, Ims RA, Rousset F (2004) Causes, mechanisms and consequences of dispersal. In: Hanski I, Gaggiotti OE (eds) Ecology, genetics and evolution of metapopulation. Elsevier Academic Press, San Diego, pp 307–335
Dierks A, Fischer K (2009) Habitat requirements and niche selection of Maculinea nausithous and M. teleius (Lepidoptera: Lycaenidae) within a large sympatric metapopulation. Biodivers Conserv 18:3663–3676
Dreisig H (1995) Thermoregulation and flight activity in territorial-male graylings, Hipparchia semele (Satyridae), and large skippers, Ochlodes venata (Hesperiidae). Oecologia 101:169–176
Elmes GW, Thomas JA (1992) The complexity of species conservation: interactions between Maculinea butterflies and their ant hosts. Biodivers Conserv 1:155–169
Eycott AE, Stewart GB, Buyung-Ali LM, Bowler DE, Watts K, Pullin AS (2012) A meta-analysis on the impact of different matrix structures on species movement rates. Landscape Ecol 27:1263–1278
Fahrig L (2003) Effects of habitat fragmentation on biodiversity. Ann Rev Ecol Evol Syst 34:487–515
Fric Z, Hula V, Klimova M, Zimmermann K, Konvicka M (2010) Dispersal of four fritillary butterflies within identical landscape. Ecol Res 25:543–552
Fronhofer E, Kubisch A, Hovestadt T, Poethke HJ (2011) Assortative mating counteracts the evolution of dispersal polymorphisms. Evolution 65:2461–2469
Gonzalez A, Lawton JH, Gilbert FS, Blackburn TM, Evans-Freke I (1998) Metapopulation dynamics, abundance, and distribution in a microecosystem. Science 281:2045–2047
Hanski I (1994) A practical model of metapopulation dynamics. J Anim Ecol 63:151–162
Hanski I (1999) Metapopulation ecology. Oxford University Press, Oxford
Hanski I (2009) Eco-evolutionary spatial dynamics in the Glanville fritillary butterfly. PNAS 108:14397–14404
Hanski I, Moilanen A, Gyllenberg M (1996) Minimum viable metapopulation size. Am Nat 147:527–541
Hanski I, Alho J, Moilanen A (2000) Estimating the parameters of survival and migration of individuals in metapopulations. Ecology 81:239–251
Hovestadt T, Nowicki P (2008) Investigating movement within irregularly shaped patches: analysis of MRR data using randomisation procedures. Isr J Ecol Evol 54:137–154
Hovestadt T, Binzenhöfer B, Nowicki P, Settele J (2011) Do all inter-patch movements represent dispersal? A mixed kernel study of butterfly mobility in fragmented landscapes. J Anim Ecol 80:1070–1077
Kammer AE, Heinrich B (1978) Insect flight metabolism. Adv Insect Physiol 13:133–228
Kindlmann P, Burel F (2008) Connectivity measures: a review. Landscape Ecol 23:879–890
Kingsolver JG (1985) Thermal ecology of Pieris butterflies (Lepidoptera: Pieridae): a new mechanism of behavioural thermoregulation. Oecologia 66:540–545
Kőrösi A, Örvössy N, Batáry P, Harnos A, Peregovits L (2012) Different habitat selection by two sympatric Maculinea butterflies at small spatial scale. Insect Conserv Divers 5:118–125
Kuefler D, Hudgens B, Haddad NM, Morris WF, Thurgate N (2010) The conflicting role of matrix habitats as conduits and barriers for dispersal. Ecology 91:944–950
Lowe WH, Allendorf FW (2010) What can genetics tell us about population connectivity? Mol Ecol 19:3038–3051
Matter SF, Roland J, Moilanen A, Hanski I (2004) Migration and survival of Parnassius smintheus: detecting effects of habitat for individual butterflies. Ecol Appl 14:1526–1534
Matter SF, Roslin T, Roland J (2005) Predicting immigration in patchy landscapes: effects of spatial scale, patch size and isolation. Oikos 111:359–367
Matter SF, Doyle A, Illerbrun K, Wheeler J, Roland J (2011) An assessment of direct and indirect effects of climate change for populations of the Rocky Mountain Apollo butterfly (Parnassius smintheus Doubleday). Insect Sci 18:385–392
Nowicki P, Vrabec V (2011) Evidence for positive density-dependent emigration in butterfly metapopulations. Oecologia 167:657–665
Nowicki P, Richter A, Glinka U, Holzschuh A, Toelke U, Henle K, Woyciechowski M, Settele J (2005a) Less input same output: simplified approach for population size assessment in Lepidoptera. Popul Ecol 47:203–212
Nowicki P, Settele J, Thomas JA, Woyciechowski M (2005b) A review of population structure of Maculinea butterflies. In: Settele J, Kuhn E, Thomas JA (eds) Studies in the ecology and conservation of butterflies in Europe, vol 2. Species ecology along a European gradient: Maculinea butterflies as a model. Pensoft Publishers, Sofia, pp 144–149
Nowicki P, Witek M, Skórka P, Settele J, Woyciechowski M (2005c) Population ecology of the endangered butterflies Maculinea teleius and M. nausithous, and its implications for conservation. Popul Ecol 47:193–202
Nowicki P, Pepkowska A, Kudlek J, Skórka P, Witek M, Settele J, Woyciechowski M (2007) From metapopulation theory to conservation recommendations: lessons from spatial occurrence and abundance patterns of Maculinea butterflies. Biol Conserv 140:119–129
Nowicki P, Bonelli S, Barbero F, Balletto E (2009) Relative importance of density-dependent regulation and environmental stochasticity for butterfly population dynamics. Oecologia 161:227–239
Nowicki P, Halecki W, Kalarus K (2013) All natural habitat edges matter equally for endangered Maculinea butterflies. J Insect Conserv 17:139–146
O’Grady JJ, Brook BW, Reed DH, Ballou JD, Tonkyn DW, Frankham R (2006) Realistic levels of inbreeding depression strongly affect extinction risk in wild populations. Biol Conserv 133:42–51
Oksanen L (2004) The devil lies in details: reply to Stuart Hurlbert. Oikos 104:598–605
Petit S, Moilanen A, Hanski I, Baguette M (2001) Metapopulation dynamics of the bog fritillary butterfly: movements between habitat patches. Oikos 92:491–500
Poethke HJ, Hovestadt T, Mitesser O (2003) Local extinction and the evolution of dispersal rates: causes and correlations. Am Nat 161:631–640
Prevedello JA, Vieira MV (2010) Does the type of matrix matter? A quantitative review of the evidence. Biodivers Conserv 19:1205–1223
Reed DH (2004) Extinction risk in fragmented habitats. Anim Conserv 7:181–191
Ricketts TH (2001) The matrix matters: effective isolation in fragmented landscapes. Am Nat 158:87–99
Ries L, Debinski DM (2001) Butterflies responses to habitat edges in the highly fragmented prairies of Central Iowa. J Anim Ecol 70:840–852
Roff DA (1994) Habitat persistence and the evolution of wing dimorphism in insects. Am Nat 144:772–798
Roland J, Keyghobadi N, Fownes S (2000) Alpine Parnassius butterfly dispersal: effects of landscape and population size. Ecology 81:1642–1653
Ross JA, Matter SF, Roland J (2005) Edge avoidance and movement of the butterfly Parnassius smintheus in matrix and non-matrix habitat. Landscape Ecol 20:127–135
Rueffler C, Van Dooren TJM, Leimar O, Abrams PA (2006) Disruptive selection and then what? TREE 21:238–245
Sacktor B (1975) Biochemistry of insect flight. 1. Utilization of fuels by muscle. In: Candy DJ, Kilby BA (eds) Insect biochemistry and function. Chapman and Hall, London, pp 1–88
Schtickzelle N, Baguette M (2003) Behavioural responses to habitat patch boundaries restrict dispersal and generate emigration–patch area relationships in fragmented landscapes. J Anim Ecol 72:533–545
Schtickzelle N, Mennechez G, Baguette M (2006) Dispersal depression with habitat fragmentation in the bog fritillary butterfly. Ecology 87:1057–1065
Schtickzelle N, Joiris A, van Dyke H, Baguette M (2007) Quantitative analysis of changes in movement behaviour within and outside habitat in a specialised butterfly. BMC Evol Biol 7:4. doi:10.1186/1471-2148-7-4
Schultz CB (1998) Dispersal behavior and its implications for reserve design for a rare Oregon butterfly. Conserv Biol 12:284–292
Schultz CB, Franco AMA, Crone EE (2012) Response of butterflies to structural and resource boundaries. J Anim Ecol 81:724–734
Sielezniew M, Stankiewicz-Fiedurek AM (2013) Behavioural evidence for a putative oviposition-deterring pheromone in the butterfly, Phengaris (Maculinea) teleius (Lepidoptera: Lycaenidae). Eur J Entomol 110:71–80
Skórka P, Nowicki P, Lenda M, Witek M, Śliwińska EB, Settele J, Woyciechowski M (2013) Different flight behaviour of the endangered scarce large blue butterfly Phengaris teleius (Lepidoptera: Lycaenidae) within and outside its habitat patches. Landscape Ecol 28:533–546
Sokal RR, Rohlf FJ (2012) Biometry: the principles and practice of statistics in biological research, 4th edn. WH Freeman and Co., New York
Stettmer C, Binzenhöfer B, Hartmann P (2001) Habitatmanagement und Schutzmassnahmen für die Ameisenbläulinge Glaucopsyche teleius und Glaucopsyche nausithous. Teil 1: populationsdynamik, Ausbreitungsverhalten und Biotopverbund. Natur und Landschaft 76:278–287
Sutcliffe OL, Thomas CD (1996) Open corridors appear to facilitate dispersal by the ringlet butterfly (Aphantopus hyperantus) between woodland clearings. Conserv Biol 10:1359–1365
Thomas JA (1984) The behaviour and habitat requirements of Maculinea nausithous (the Dusky Large Blue) and M. teleius (the Scarce Large Blue) in France. Biol Conserv 28:325–347
Thomas JA (1995) The ecology and conservation of Maculinea arion and other European species of Large Blue Butterfly. In: Pullin AS (ed) Ecology and conservation of butterflies. Chapman and Hall, London, pp 180–197
With KA, King AW (1999) Dispersal success on fractal landscapes: a consequence of lacunarity thresholds. Landscape Ecol 14:73–82
Zeller KA, McGarigal K, Whiteley AR (2012) Estimating landscape resistance to movement: a review. Landscape Ecol 27:777–797
Zollner PA, Lima SL (1999) Search strategies for landscape level interpatch movements. Ecology 80:1019–1030
Acknowledgments
We are grateful to several institutions that financed the surveys as well as to numerous volunteers who helped in the fieldwork. The data analysis was funded by the Polish National Science Centre Grant No. N304 064139 and by the European Commission within its project SCALES (FP7 contract no. 226852). TH and JS were supported by the EU FP6 BiodivERsAEranet through its projects TenLamas and CLIMIT respectively; the latter being financed by DLR-BMBF (Germany), NERC and DEFRA (UK), ANR (France), Formas and Swedish EPA (Sweden). Norma Orlowski improved the English of the manuscript, and two anonymous reviewers provided useful comments to its earlier version.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Nowicki, P., Vrabec, V., Binzenhöfer, B. et al. Butterfly dispersal in inhospitable matrix: rare, risky, but long-distance. Landscape Ecol 29, 401–412 (2014). https://doi.org/10.1007/s10980-013-9971-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-013-9971-0