Abstract
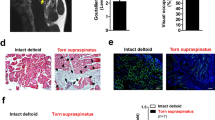
Aquaporin-4 (AQP4) is a selective water channel mediating water transport across cell membranes in skeletal muscles. Recently, it was noted that AQP4 is one of the key molecules regulating muscle morphology. Indeed, the AQP4 accumulation level was stably maintained in hypertrophied skeletal muscles. On the other hand, whether the AQP4 accumulation level is stably maintained in atrophied muscles remains poorly understood. The present study investigated the changes in the AQP4 accumulation level in the atrophied muscles at 2 weeks after denervation. As a result, the accumulation level of AQP4 in the atrophied muscle was significantly decreased compared with that in the control muscle (p < 0.05). Interestingly, the accumulation level of α1-syntrophin, which is an essential factor in regulating the stable accumulation level of AQP4, was stably maintained in the atrophied muscles. On the other hand, the accumulation level of the transient receptor potential vanilloid 4 (TRPV4), which contributes to cell volume control via interaction with AQP4, was significantly increased in the atrophied muscles compared with that in the control muscle (p < 0.05). Therefore, the present study suggested that the imbalance between the AQP4 accumulation level and skeletal muscle volume may be induced in the atrophied muscles by denervation, and the decrease in the accumulation level of AQP4 may be accompanied by defects in the functional and structural relationships with α1-syntrophin and TRPV4.





Similar content being viewed by others
References
Adams ME, Mueller HA, Froehner SC (2001) In vivo requirement of the alpha-syntrophin PDZ domain for the sarcolemmal localization of nNOS and aquaporin-4. J Cell Biol 155:113–122
Basco D, Nicchia GP, Desaphy JF, Camerino DC, Frigeri A, Svelto M (2010) Analysis by two-dimensional Blue Native/SDS-PAGE of membrane protein alterations in rat soleus muscle after hindlimb unloading. Eur J Appl Physiol 110:1215–1224
Basco D, Nicchia GP, D’Alessandro A, Zolla L, Svelto M, Frigeri A (2011) Absence of aquaporin-4 in skeletal muscle alters proteins involved in bioenergetic pathways and calcium handling. PLoS One 6:e19225
Benfenati V, Caprini M, Dovizio M, Mylonakou MN, Ferroni S, Ottersen OP, Amiry-Moghaddam M (2011) An aquaporin-4/transient receptor potential vanilloid 4 (AQP4/TRPV4) complex is essential for cell-volume control in astrocytes. Proc Natl Acad Sci USA 108:2563–2568
Crosbie RH, Dovico SA, Flanagan JD, Chamberlain JS, Ownby CL, Campbell KP (2002) Characterization of aquaporin-4 in muscle and muscular dystrophy. FASEB J 16:943–949
Frigeri A, Nicchia GP, Verbavatz JM, Valenti G, Svelto M (1998) Expression of aquaporin-4 in fast-twitch fibers of mammalian skeletal muscle. J Clin Invest 102:695–703
Frigeri A, Nicchia GP, Nico B, Quondamatteo F, Herken R, Roncali L, Svelto M (2001a) Aquaporin-4 deficiency in skeletal muscle and brain of dystrophic mdx mice. FASEB J 15:90–98
Frigeri A, Nicchia GP, Desaphy JF, Pierno S, De Luca A, Camerino DC, Svelto M (2001b) Muscle loading modulates aquaporin-4 expression in skeletal muscle. FASEB J 15:1282–1284
Frigeri A, Nicchia GP, Balena R, Nico B, Svelto M (2004) Aquaporins in skeletal muscle: reassessment of the functional role of aquaporin-4. FASEB J 18:905–907
Hara H, Wakayama Y, Kojima H, Inoue M, Jimi T, Iijima S, Masaki H (2011) Aquaporin 4 expression in the mdx mouse diaphragm. Acta Histochem Cytochem 44:175–182
Ho TC, Horn NA, Huynh T, Kelava L, Lansman JB (2012) Evidence TRPV4 contributes to mechanosensitive ion channels in mouse skeletal muscle fibers. Channels (Austin) 6:246–254
Ishido M, Nakamura T (2016) Aquaporin-4 protein is stably maintained in the hypertrophied muscles by functional overload. Acta Histochem Cytochem 49:89–95
Jimi T, Wakayama Y, Murahashi M, Shibuya S, Inoue M, Hara H, Matsuzaki Y, Uemura N (2000) Aquaporin 4: lack of mRNA expression in the rat regenerating muscle fiber under denervation. Neurosci Lett 291:93–96
Jimi T, Wakayama Y, Matsuzaki Y, Hara H, Inoue M, Shibuya S (2004) Reduced expression of aquaporin 4 in human muscles with amyotrophic lateral sclerosis and other neurogenic atrophies. Pathol Res Pract 200:203–209
Jo AO, Ryskamp DA, Phuong TT, Verkman AS, Yarishkin O, MacAulay N, Krizaj D (2015) TRPV4 and AQP4 channels synergistically regulate cell volume and calcium homeostasis in retinal muller glia. J Neurosci 35:13525–13537
Kozono D, Yasui M, King LS, Agre P (2002) Aquaporin water channels: atomic structure molecular dynamics meet clinical medicine. J Clin Invest 109:1395–1399
Kunert-Keil C, Bisping F, Kruger J, Brinkmeier H (2006) Tissue-specific expression of TRP channel genes in the mouse and its variation in three different mouse strains. BMC Genomics 7:159
Mola MG, Sparaneo A, Gargano CD, Spray DC, Svelto M, Frigeri A, Scemes E, Nicchia GP (2016) The speed of swelling kinetics modulates cell volume regulation and calcium signaling in astrocytes: A different point of view on the role of aquaporins. Glia 64:139–154
Neely JD, Amiry-Moghaddam M, Ottersen OP, Froehner SC, Agre P, Adams ME (2001) Syntrophin-dependent expression and localization of Aquaporin-4 water channel protein. Proc Natl Acad Sci USA 98:14108–14113
Preston GM, Agre P (1991) Isolation of the cDNA for erythrocyte integral membrane protein of 28 kilodaltons: member of an ancient channel family. Proc Natl Acad Sci USA 88:11110–11114
Preston GM, Carroll TP, Guggino WB, Agre P (1992) Appearance of water channels in Xenopus oocytes expressing red cell CHIP28 protein. Science 256:385–387
Pritschow BW, Lange T, Kasch J, Kunert-Keil C, Liedtke W, Brinkmeier H (2011) Functional TRPV4 channels are expressed in mouse skeletal muscle and can modulate resting Ca2+ influx and muscle fatigue. Pflugers Arch 461:115–122
Verkman AS, Mitra AK (2000) Structure and function of aquaporin water channels. Am J Physiol Renal Physiol 278:F13–F28
Wakayama Y, Jimi T, Inoue M, Kojima H, Murahashi M, Kumagai T, Yamashita S, Hara H, Shibuya S (2002) Reduced aquaporin 4 expression in the muscle plasma membrane of patients with Duchenne muscular dystrophy. Arch Neurol 59:431–437
Wakayama Y, Takahashi J, Shibuya S, Inoue M, Kojima H, Oniki H, Arata S, Hara H, Jimi T, Shioda S, Sunada Y, Ohi H, Shimizu T (2007) Generation of muscle aquaporin 4 overexpressing transgenic mouse: its characterization at RNA and protein levels including freeze-fracture study. Micron 38:257–267
Yang B, Verbavatz JM, Song Y, Vetrivel L, Manley G, Kao WM, Ma T, Verkman AS (2000) Skeletal muscle function and water permeability in aquaporin-4 deficient mice. Am J Physiol Cell Physiol 278:C1108–C1115
Yokota T, Miyagoe Y, Hosaka Y, Tsukita K, Kameya S, Shibuya S, Matsuda R, Wakayama Y, Takeda S (2000) Aquaporin-4 is absent at the sarcolemma and at perivascular astrocyte endfeet in α1-syntrophin knockout mice. Proc Jpn Acad 76:22–27
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ishido, M., Nakamura, T. Marked decrease of aquaporin-4 protein is independent of the changes in α1-syntrophin and TRPV4 levels in response to denervation-induced muscle atrophy in vivo. J Muscle Res Cell Motil 38, 175–181 (2017). https://doi.org/10.1007/s10974-017-9471-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10974-017-9471-y