Abstract
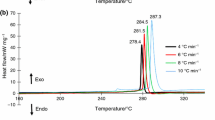
In order to study the effect of aluminum powder and Mg(BH4)2 on the thermal safety of energetic materials, the mixtures that RDX mixed with different proportions of Al/Mg(BH4)2 were produced by using the molding powder method. The thermal decomposition curves of mixed explosives were obtained by differential scanning calorimetry (DSC) and adiabatic accelerated calorimetry (ARC), the kinetic parameters were calculated, and the risk of the thermal runaway was evaluated. The following main conclusions were drawn. Under the premise of a certain RDX quality, the decomposition heat of adding the same mass of Al is 1.15 times that of adding the same mass of Mg(BH4)2. Adding Mg(BH4)2 alone will reduce the thermal stability of RDX, while adding Al/Mg(BH4)2 mixture will improve the thermal stability of RDX. The mass ratio of Al/Mg(BH4)2 is less than 2:1, which can improve the performance of mixed explosives to a certain extent, and the mixed explosive as a whole achieve better thermal safety. This study provides important information for improving the safety of storage, transportation, and use of RDX-based mixed explosives.





Similar content being viewed by others
Abbreviations
- DSC:
-
Differential scanning calorimetry
- ARC:
-
Adiabatic accelerated calorimetry
- ICTAC:
-
International confederation for thermal analysis and calorimetry
- TMRad :
-
Time to maximum reaction rate under adiabatic condition
- T on :
-
Temperature of the initial reaction, K
- T off :
-
Temperature of the end reaction, K
- ∆H :
-
Reaction enthalpy, J g−1
- β :
-
Heating rate, K min−1
- T p :
-
Peak temperature of the exothermic peak, K
- R :
-
Ideal gas constant, J·mol−1 K−1
- E :
-
Apparent activation energy, J mol−1
- A :
-
Denotes pre-exponential factor, s−1
- r :
-
Correlation coefficients
- ∆T ad :
-
Adiabatic temperature rise, °C
- ∆H r :
-
Specific heat release, J g−1
- ∆T ad * :
-
Adiabatic temperature rise corrected by ф
- ∆Hr * :
-
Modified specific heat release corrected by ф
- C p :
-
Specific heat capacity, J g−1 °C−1
- V':
-
Specific gas yield of the sample, mL g−1
- P 1 :
-
Pressure in the heating stage at the same temperature, bar
- P 2 :
-
Pressure in the cooling stage at the same temperature, bar
- P 0 :
-
Normal pressure, 1.01 bar
- T 0 :
-
Normal temperature, 25 °C
- T :
-
Selected temperature for calculation of gas production, °C
- m :
-
Sample mass, g
References
Wang XH, Li XJ, Yan HH, Qu YD, Sun GL, Xie XH, Zhang YJ. Research of thermal decomposition kinetic characteristic of emulsion explosive base containing Fe and Mn elements. J Therm Anal Calorim. 2008;91:545–50.
Zhou J, Ding L, An J, Zhu Y, Liang Y. Study on the thermal behaviors of nano-Al based fuel air explosive. J Therm Anal Calorim. 2017;130:1111–6.
Bari R, Denton AA, Fondren ZT, McKenna GB, Simon SL. Acceleration of decomposition of CL-20 explosive under nanoconfinement. J Therm Anal Calorim. 2020;140:2649–55.
Talawar MB, Jangid SK, Nath T, Sinha RK, Asthana SN. New directions in the science and technology of advanced sheet explosive formulations and the key energetic materials used in the processing of sheet explosives: emerging trends. J Hazard Mater. 2015;300:307–21.
Douglass DL. Formation and dissociation of magnesium alloy hydrides and their use for fuel storage in hydrogen car. Metall Trans A Phys Metall Mater Sci. 1975;6:2179–89.
Zhang S, Lee LH, Sun Y, Liu Y. IOP In 2020 ASIA conference on geological research and environmental technology Asia conference on geological research and environmental technology (GRET), 2021:632.
Chlopek K, Frommen C, Leon A, Zabara O, Fichtner M. Synthesis and properties of magnesium tetrahydroborate, Mg(BH4)2. J Mater Chem. 2007;17:3496–503.
Hanada N, Chopek K, Frommen C, Lohstroh W, Fichtner M. Thermal decomposition of Mg(BH4)2 under He flow and H-2 pressure. J Mater Chem. 2008;18:2611–4.
Li HW, Kikuchi K, Nakamori Y, Ohba N, Miwa K, Towata S, Orimo S. Dehydriding and rehydriding processes of well-crystallized Mg(BH4)2 accompanying with formation of intermediate compounds. Acta Mater. 2008;56:1342–7.
Makhaev VD, Borisov AP, Antsyshkina AS, Sadikov GG. Synthesis and properties of new magnesium tetrahydroborate complexes: the crystal structure of (Ph4P)2[Mg(BH4)4]. Russ J Inorg Chem. 2004;49:323–30.
van Setten MJ, Lohstroh W, Fichtner M. A new phase in the decomposition of Mg(BH4)2: first-principles simulated annealing. J Mater Chem. 2009;19:7081–7.
Abusaidi H, Ghaieni HR. Thermal analysis and kinetic decomposition of Nitro-functionalized hydroxyl-terminated polybutadiene bonded explosive. J Therm Anal Calorim. 2017;127:2301–6.
Luo L, Guo P, Jin B, Xiao Y, Zhang Q, Chu S, Peng R. An isothermal decomposition dynamics research instrument and its application in HMX/TNT/Al composite explosive. J Therm Anal Calorim. 2020;139:2265–72.
Cao C, Chen W, Jhang W, Chung Y, Lin W. Thermal decomposition and evaluation thermokinetic parameters for explosive type. J Therm Anal Calorim. 2021;144:443–54.
Yue Y, et al. Effect of Mg(BH4)2 on safety of desensitizing RDX. China Saf Sci J. 2016;26:125–9.
Wei YJ, Cheng LP, Yao M, Peng JH. Effect of MgH2 and Mg(BH4)2 on thermal decomposition process of ammonium nitrate. Chin J Explos Propellants. 2015;38:59–68.
Yao M, Ding W, Rao G, Chen L, Peng J. Effects of MgH2/Mg(BH4)2 powders on the mechanical sensitivity of conventional explosive compounds. Propell Explos Pyrot. 2018;43:274–9.
Wang HJ, Liu SS. In Mechatronics and materials processing I, PTS 1–3; Zhang LC; Zhang CL; Chen Z (Eds) International conference on mechatronics and materials processing (ICMMP 2011), 2011:328–330.
An J, Ju R, Zeng J, Luo Y, Luo H, Ma H. Different inhibition mechanisms and safety effects of TPU and EVA on thermal decomposition of RDX. J Therm Anal Calorim. 2022;147:11261–72.
Schroederd MA Critical analysis of nitramine decomposition data: product distributions from HMX and RDX decomposition, Army Ballistic research lab., Aberdeen Proving Ground, MD., 1985.
Jiang Z, et al. Effects of nanometer metal powders on thermal decomposition characteristics of RDX. J Nanjing Univ Sci Technol. 2002;23:258.
Jiang J, Wei J, Leng H, Li Q, Chou K. Effect of Al on the hydrogen storage properties of Mg(BH4)2. Int J Hydrogen Energ. 2013;38:10919–25.
Huang G, Zou Y, Luo W, Xiao M, Han D, Wang S, Meng Y. Nonisothermal crystallization behavior and kinetics of poly( L-lactide-co-propylene carbonate). J Therm Anal Calorim. 2015;121:877–83.
Wang S, Kossoy AA, Yao Y, Chen L, Chen W. Kinetics-based simulation approach to evaluate thermal hazards of benzaldehyde oxime by DSC tests. Thermochim Acta. 2017;655:319–25.
Kissinger HE. Reaction kinetics in differential thermal analysis. Anal Chem. 1957;29:1702–6.
Zhou J, Yu A, Suetor CG, Liang X, Hua M, Pan X, Ni L, Jiang J. Risk assessment of polyarylether polymerization process. J Therm Anal Calorim. 2021;144:295–303.
Wang W, Su W, Jiao Z, Wang Q. Thermal hazard analysis of inorganic peroxide initiators with varying water concentrations. J Therm Anal Calorim. 2021;146:1111–20.
Acknowledgements
The author thanks the teacher for his guidance and relatives and friends for their care and help.
Funding
This work was supported by the National Nature Science Foundation of China [Grant number 51704302].
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chang, C., Wei, X., Ren, J. et al. Effect of different proportions of Mg(BH4)2/Al on thermal decomposition of RDX. J Therm Anal Calorim 148, 4689–4698 (2023). https://doi.org/10.1007/s10973-023-12140-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-023-12140-1