Abstract
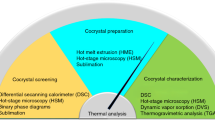
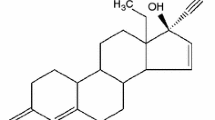
Amorphous form and its characterization using a wide variety of analytical techniques have been known for decades. However, there is no single superior method because of similarities between crystalline mesophases and amorphous forms. The aim of the present study was to characterize and identify solid-state of CKD-519, a new cholesteryl ester transfer protein inhibitor, using a combination of analytical techniques. CKD-519 was analyzed via scanning electron microscope, thermogravimetric/differential thermal analysis (TG/DTA), differential scanning calorimetry (DSC), hot-stage polarized light microscopy (HSPLM), and variable temperature x-ray powder diffraction (VT-XRPD). Endothermic shifts in the baseline before and after the peak with no mass change were observed via TG/DTA and conventional DSC. Enthalpy recovery experiments and modulated-temperature DSC (MDSC) measurement revealed enthalpy recovery superimposed on glass transition. The absence of birefringence in HSPLM micrographs as well as the glass transition and presence of diffuse scattering (halo) in the VT-XRPD pattern of micron-sized particles up to 200 µm indicate that CKD-519 exists in an amorphous form. This combination of analytical techniques may be used to distinguish amorphous forms from crystalline mesophases.










Similar content being viewed by others
References
The United States Pharmacopeia (USP). <696> Characterization of crystalline solids by microcalorimetry and solution calorimetry monograph. In: The United States Pharmacopeia and The National Formulary: The Official Compendia of Standards. USP38-NF33. Rockville, MD: The United States Pharmacopeial Convention Inc.; 2015. pp. 474–7.
Hancock BC, Zografi G. Characteristics and significance of the amorphous state in pharmaceutical systems. J Pharm Sci. 1997;86(1):1–12. https://doi.org/10.1021/js9601896.
Shalaev E, Wu K, Shamblin S, Krzyzaniak JF, Descamps M. Crystalline mesophases: structure, mobility, and pharmaceutical properties. Adv Drug Deliv Rev. 2016;100:194–211. https://doi.org/10.1016/j.addr.2016.04.002.
Thakral S, Terban MW, Thakral NK, Suryanarayanan R. Recent advances in the characterization of amorphous pharmaceuticals by x-ray diffractometry. Adv Drug Deliv Rev. 2016;100:183–93. https://doi.org/10.1016/j.addr.2015.12.013.
Babu NJ, Nangia A. Solubility advantage of amorphous drugs and pharmaceutical cocrystals. Cryst Growth Des. 2011;11(7):2662–79. https://doi.org/10.1021/cg200492w.
Hancock BC, Parks M. What is the true solubility advantage for amorphous pharmaceuticals? Pharm Res. 2000;17(4):397–404. https://doi.org/10.1023/A:1007516718048.
Murdande SB, Pikal MJ, Shanker RM, Bogner RH. Solubility advantage of amorphous pharmaceuticals: II Application of quantitative thermodynamic relationships for prediction of solubility enhancement in structurally diverse insoluble pharmaceuticals. Pharmaceutical Res. 2010;27(12):2704–14. https://doi.org/10.1007/s11095-010-0269-5.
Baird JA, Taylor LS. Evaluation of amorphous solid dispersion properties using thermal analysis techniques. Adv Drug Deliv Rev. 2012;64(5):396–421. https://doi.org/10.1016/j.addr.2011.07.009.
Chieng N, Rades T, Aaltonen J. An overview of recent studies on the analysis of pharmaceutical polymorphs. J Pharm Biomed Anal. 2011;55(4):618–44. https://doi.org/10.1016/j.jpba.2010.12.020.
The United States Pharmacopeia (USP). <695> Crystallinity monograph. In: The United States Pharmacopeia and The National Formulary: The Official Compendia of Standards. USP38-NF33. Rockville, MD: The United States Pharmacopeial Convention Inc.; 2015. pp. 474.
The United States Pharmacopeia (USP). <776> Optical microscopy monograph. In: The United States Pharmacopeia and The National Formulary: The Official Compendia of Standards. USP38-NF33. Rockville, MD: The United States Pharmacopeial Convention Inc.; 2015. pp. 537–40.
The United States Pharmacopeia (USP). <941> Characterization of crystalline and partially crystalline solids by X-ray powder diffraction (XRPD) monograph. In: The United States Pharmacopeia and The National Formulary: The Official Compendia of Standards. USP38-NF33. Rockville, MD: The United States Pharmacopeial Convention Inc.; 2015. pp. 692–8.
Bates S, Zografi G, Engers D, Morris K, Crowley K, Newman A. Analysis of amorphous and nanocrystalline solids from their x-ray diffraction patterns. Pharm Res. 2006;23(10):2333–49. https://doi.org/10.1007/s11095-006-9086-2.
Stevenson CL, Bennett DB, Lechuga-Ballesteros D. Pharmaceutical liquid crystals: the relevance of partially ordered systems. J Pharm Sci. 2005;94(9):1861–80. https://doi.org/10.1002/jps.20435.
Wunderlich B. A classification of molecules, phases, and transitions as recognized by thermal anlysis. Thermochim Acta. 1999;340–1:37–52. https://doi.org/10.1016/S0040-6031(99)00252-X.
Priemel PA, Laitinen R, Barthold S, Grohganz H, Lehto V-P, Rades T, et al. Inhibition of surface crystallisation of amorphous indomethacin particles in physical drug-polymer mixtures. Int J Pharm. 2013;456(2):301–6. https://doi.org/10.1016/j.ijpharm.2013.08.046.
Hancock BC, Shamblin SL. Molecular mobility of amorphous pharmaceuticals determined using differential scanning calorimetry. Thermochim Acta. 2001;380(2):95–107. https://doi.org/10.1016/S0040-6031(01)00663-3.
Moynihan CT, Easteal AJ, Wilder J, Tucker J. Dependence of the glass transition temperature on heating and cooling rate. J Phys Chem. 1974;78(26):2673–7. https://doi.org/10.1021/j100619a008.
Hancock BC, Shamblin SL, Zografi G. Molecular mobility of amorphous pharmaceutical solids below their glass transition temperatures. Pharm Res. 1995;12(6):799–806. https://doi.org/10.1023/a:1016292416526.
Kawakami K, Pikal MJ. Calorimetric investigation of the structural relaxation of amorphous materials: Evaluating validity of the methodologies. J Pharm Sci. 2005;94(5):948–65. https://doi.org/10.1002/jps.20298.
Luthra SA, Hodge IM, Pikal MJ. Investigation of the impact of annealing on global molecular mobility in glasses: optimization for stabilization of amorphous pharmaceuticals. J Pharm Sci. 2008;97(9):3865–82. https://doi.org/10.1002/jps.21255.
Bosma M, ten Brinke G, Ellis TS. Polymer-polymer miscibility and enthalpy relaxations. Macromolecules. 1988;21(5):1465–70. https://doi.org/10.1021/ma00183a041.
Ellis TS. Aromatic polyamide blends: Enthalpy relaxation and its correlation with phase phenomena. Macromolecules. 1990;23(5):1494–503. https://doi.org/10.1021/ma00207a042.
ten Brinke G, Oudhuis L, Ellis TS. The thermal characterization of multi-component systems by enthalpy relaxation. Thermochim Acta. 1994;238:75–98. https://doi.org/10.1016/S0040-6031(94)85207-3.
Liu J, Rigsbee DR, Stotz C, Pikal MJ. Dynamics of pharmaceutical amorphous solids: the study of enthalpy relaxation by isothermal microcalorimetry. J Pharm Sci. 2002;91(8):1853–62. https://doi.org/10.1002/jps.10181.
Hill VL, Craig DQ, Feely LC. Characterisation of spray-dried lactose using modulated differential scanning calorimetry. Int J Pharm. 1998;161(1):95–107. https://doi.org/10.1016/S0378-5173(97)00334-7.
Royall PG, Craig DQ, Doherty C. Characterisation of the glass transition of an amorphous drug using modulated DSC. Pharm Res. 1998;15(7):1117–21. https://doi.org/10.1023/a:1011902816175.
Verdonck E, Schaap K, Thomas LC. A discussion of the principles and applications of modulated temperature DSC (MTDSC). Int J Pharm. 1999;192(1):3–20. https://doi.org/10.1016/S0378-5173(99)00267-7.
Craig DQ, Barsnes M, Royall PG, Kett VL. An evaluation of the use of modulated temperature DSC as a means of assessing the relaxation behaviour of amorphous lactose. Pharm Res. 2000;17(6):696–700. https://doi.org/10.1023/A:1007578131582.
Hutchinson JM, Montserrat S. The application of temperature-modulated DSC to the glass transition region: II Effect of a distribution of relaxation times. Thermochim Acta. 2001;377(1–2):63–84. https://doi.org/10.1016/S0040-6031(01)00542-1.
Simon SL. Temperature-modulated differential scanning calorimetry: Theory and application. Thermochim Acta. 2001;374(1):55–71. https://doi.org/10.1016/S0040-6031(01)00493-2.
Van Den Mooter G, Craig DQ, Royall PG. Characterization of amorphous ketoconazole using modulated temperature differential scanning calorimetry. J Pharm Sci. 2001;90(8):996–1003. https://doi.org/10.1002/jps.1052.
Acknowledgements
This research was supported by Korea Initiative for fostering University of Research and Innovation Program of the National Research Foundation (NRF) funded by the Korean government (MSIT) (No. 2020M3H1A1077095)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Seok, S.H., Kim, JY., Park, CW. et al. Solid-state characterization and amorphous form identification in a small molecule. J Therm Anal Calorim 147, 6301–6308 (2022). https://doi.org/10.1007/s10973-021-10907-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-021-10907-y