Abstract
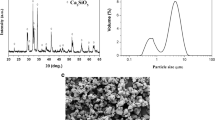
Endodontic calcium silicate-based cement is the most used materials in root-end filling and/or repairing perforations. However, the relatively high cost associated with these cement is a drawback that necessitates improvements in their manufacturing process through the development of alternatives. This study used differential scanning calorimetry (DSC) and high-temperature X-ray powder diffraction observations of the tricalcium silicate (C3S) crystalline phase to identify and quantify important parameters of hydration reactions such as the degree of hydration. A C3S reference was prepared with pure chemical reagents to develop a novel biomaterial variety of calcium silicate-based cement which was predefined to preserve the significant properties and clinical applications typical of calcium silicates. A methodology of cycled synthesis was used to obtain almost pure, highly crystalline C3S phase. With temperatures that can reach 1450–1500 °C, the thermal processing of the mixture of raw materials gives rise to thermochemical reactions of the formation of the main phases: 2CaO·SiO2 (C2S), 3CaO·SiO2 (C3S), and 3CaO·Al2O3 (C3A). The formation of the C3S phase is aided by the fusion of calcium aluminates (C3A). The use of Al2O3 and MgO as liquid phase-forming agents and the stabilization of the phases in cooling, allowed the liquid phase to provide the greatest precipitation of the C3S solid phase of interest. The X-ray diffraction confirmed the formation of the highly C3S crystalline phase in the material after cooling at the end of the five thermal cycles. The results obtained indicated that such a material could be derived through bottom-up synthesis routes that aim to attain the proper balance between the C3S phase and the use of other cementitious materials.




Similar content being viewed by others
Availability of data and materials
All authors agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
References
Schilder H. Filling root canals in three dimensions. J Endod. 2006. https://doi.org/10.1016/j.joen.2006.02.007.
Parirokh M, Torabinejad M, Dummer PMH. Mineral trioxide aggregate and other bioactive endodontic cements: an updated overview—part I: vital pulp therapy. Int Endod J. 2018. https://doi.org/10.1111/iej.12841.
Bernardes RA, de Amorim CA, Junior D, Pereira LO, Duarte MAH, Moraes IG, Bramante CM. Evaluation of the flow rate of 3 endodontic sealers: Sealer 26, AH Plus, and MTA Obtura. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2010. https://doi.org/10.1016/j.tripleo.2009.08.038.
Torabinejad M, Watson TF, Ford TRP. Sealing ability of a mineral trioxide aggregate when used as a root end filling material. J Endot. 1993. https://doi.org/10.1016/S0099-2399(06)80271-2.
Parirokh M, Torabinejad M. Mineral trioxide aggregate: a comprehensive literature review—part III: clinical applications, drawbacks, and mechanism of action. J Endod. 2010. https://doi.org/10.1016/j.joen.2009.09.009.
Camilleri J. Characterization and hydration kinetics of tricalcium silicate cement for use as a dental biomaterial. Dent Mater. 2011. https://doi.org/10.1016/j.dental.2011.04.010.
Formosa LM, Mallia B, Camilleri J. The effect of curing conditions on the physical properties of tricalcium silicate cement for use as a dental biomaterial. Int Endod J. 2012. https://doi.org/10.1111/j.1365-2591.2011.01980.x.
Roberts HW, Toth JM, Berzins DW, Charlton DG. Mineral trioxide aggregate material use in endodontic treatment: a review of the literature. Dent Mater. 2008. https://doi.org/10.1016/j.dental.2007.04.007.
Pina S, Vieira SI, Rego P, Torres PMC, da Cruz e Silva OAB, da Cruz e Silva EF, Ferreira JMF. Biological responses of brushite-forming Zn- and ZnSr-substituted beta-tricalcium phosphate bone cements. Eur Cell Mater. 2010; https://doi.org/https://doi.org/10.22203/ecm.v020a14
Hamdan Alkhraisat M, Moseke C, Blanco L, Barralet JE, Lopez-Carbacos E, Gbureck U. Strontium modified biocements with zero order release kinetics. Biomaterials. 2008. https://doi.org/10.1016/j.biomaterials.2008.08.026.
Sarkar NK, Caicedo R, Ritwik P, Moiseyeva R, Kawashima I. Physicochemical basis of the biologic properties of mineral trioxide aggregate. J Endod. 2005. https://doi.org/10.1097/01.don.0000133155.04468.41.
Reyes-Carmona JF, Felippe MS, Felippe WT. Biomineralization ability and interaction of mineral trioxide aggregate and white portland cement with dentin in a phosphate-containing fluid. J Endod. 2009. https://doi.org/10.1016/j.joen.2009.02.011.
Tay FR, Pashley DH, Rueggeberg FA, Loushine RJ, Weller RN. Calcium phosphate phase transformation produced by the interaction of the portland cement component of white mineral trioxide aggregate with a phosphate-containing fluid. J Endod. 2007. https://doi.org/10.1016/j.joen.2007.07.008.
Xuereb M, Vella P, Damidot D, Sammut CV, Camilleri J. In situ assessment of the setting of tricalcium silicate-based sealers using a dentin pressure model. J Endod. 2015. https://doi.org/10.1016/j.joen.2014.09.015.
Candeiro GTM, Moura-Netto C, D’Almeida-Couto RS, Azambuja-Júnior N, Marques MM, Cai S, Gavini G. Cytotoxicity, genotoxicity and antibacterial effectiveness of a bioceramic endodontic sealer. Int Endod J. 2016. https://doi.org/10.1111/iej.12523.
Duarte MAH, Marciano MA, Vivan RR, Tanomaru Filho M, Tanomaru JMG, Camilleri J. Tricalcium silicate-based cements: properties and modifications. Braz Oral Res. 2018. https://doi.org/10.1590/1807-3107bor-2018.vol32.0070.
Camilleri J, Sorrentino F, Damidot D. Investigation of the hydration and bioactivity of radiopacified tricalcium silicate cement. Biodentine MTA Angelus Dent Mater. 2013. https://doi.org/10.1016/j.dental.2013.03.007.
Yang F, Liu X, Zhao Y, Zhang Y, Wang P, Robinson I, Chen B. Investigation of three-dimensional microstructure of tricalcium silicate (C3S) by electron microscopy. Materials (Basel). 2018. https://doi.org/10.3390/ma11071110.
Ramachandran VS. Estimation of tricalcium silicate through polymorphic transformation. J Therm Anal Calorim. 1971. https://doi.org/10.1007/BF01904682.
Nocun-Wczelik W. Thermogravimetric studies of the tricalcium silicate hydration in the presence of solid additives. J Therm Anal Calorim. 1990. https://doi.org/10.1007/BF01914134.
Murzyn P, Malata G, Wiśniewska J, Kapeluszna E, Nocuń-Wczelik W. Characterization of 40-year-old calcium silicate pastes by thermal methods and other techniques. J Therm Anal Calorim. 2019. https://doi.org/10.1007/s10973-019-08519-8.
Turkyilmazoglu M. Single phase nanofluids in fluid mechanics and their hydrodynamic linear stability analysis. Comp Method Prog Biomed. 2020. https://doi.org/10.1016/j.cmpb.2019.105171.
Turkyilmazoglu M. Nanoliquid film flow due to a moving substrate and heat transfer. Eur Phys J Plus. 2020. https://doi.org/10.1140/epjp/s13360-020-00812-y.
Staněk T, Sulovský P. The influence of the alite polymorphism on the strength of the Portland cement. Cem Concr Res. 2002. https://doi.org/10.1016/S0008-8846(02)00756-1.
Taylor HFW. Cement chemistry. Cem. Chem. 2nd ed. London: Thomas Telford Services Ltd; 1997.
Hewlett P. Lea’s chemistry of cement and concrete. 4th ed. Oxford: Butterworth-Heinemann; 2003.
Velez K, Maximilien S, Damidot D, Fantozzi G, Sorrentino F. Determination by nanoindentation of elastic modulus and hardness of pure constituents of Portland cement clinker. Cem Concr Res. 2001. https://doi.org/10.1016/S0008-8846(00)00505-6.
Hong H, Fu Z, Min X. Effect of cooling performance on the mineralogical character of Portland cement clinker. Cem Concr Res. 2001. https://doi.org/10.1016/S0008-8846(00)00445-2.
Maki I, Fukuda K, Imura T, Yoshida H, Ito S. Formation of belite clusters from quartz grains in portland cement clinker. Cem Concr Res. 1995. https://doi.org/10.1016/0008-8846(95)00074-M.
Li X, Xu W, Wang S, Tang M, Shen X. Effect of SO3 and MgO on Portland cement clinker: formation of clinker phases and alite polymorphism. Constr Build Mater. 2014. https://doi.org/10.1016/j.conbuildmat.2014.02.029.
Ichikawa M, Kanaya M. Effects of minor components and heating rates on the fine textures of alite in portland cement clinker. Cem Concr Res. 1997. https://doi.org/10.1016/S0008-8846(97)00103-8.
Staněk T, Sulovský P. The influence of phosphorous pentoxide on the phase composition and formation of Portland clinker. Mater Charact. 2009. https://doi.org/10.1016/j.matchar.2008.11.013.
Simão L, Jiusti J, Lóh NJ, Hotza D, Raupp-Pereira F, Labrincha JA, Montedo ORK. Waste-containing clinkers: valorization of alternative mineral sources from pulp and paper mills. Process Saf Environ Prot. 2017. https://doi.org/10.1016/j.psep.2017.03.038.
Maki I, Fukuda K, Seki S, Tanioka T. Impurity distribution during crystal growth of alite in portland cement clinker. J Am Ceram Soc. 1991. https://doi.org/10.1111/j.1151-2916.1991.tb08263.x.
Formosa LM, Mallia B, Bull T, Camilleri J. The microstructure and surface morphology of radiopaque tricalcium silicate cement exposed to different curing conditions. Dent Mater. 2012. https://doi.org/10.1016/j.dental.2012.02.006.
Andrade AS, Silva GF, Camilleri J, Cerri ES, Guerreiro-Tanomaru JM, Cerri PS, Tanomaru-Filho M. Tissue response and immunoexpression of interleukin 6 promoted by tricalcium silicate-based repair materials after subcutaneous implantation in rats. J Endod. 2018. https://doi.org/10.1016/j.joen.2017.12.006.
Acknowledgements
The authors are very grateful to Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES/Brazil) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq/Brazil; process n. 308669/2016-9; 307761/2019-3) for supporting this work.
Funding
All authors declare that there are no sources of funding, financial or nonfinancial interests.
Author information
Authors and Affiliations
Contributions
All authors have made substantial contributions to the conception or design of the work; acquisition, analysis, and interpretation of data; drafted the work or substantively revised it; and approved the submitted version, including the substantially modified version after making revision requested by the reviewers.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Consent for publication
All authors approve the version to be published.
Ethics approval and consent to participate
The authors agree with the content and consent to submit the manuscript. All authors obtained consent from the responsible authorities at the institute/organization where the work has been carried out before the work is submitted.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bernardi, A.V., Raupp-Pereira, F., Possolli, N.M. et al. Evolution of tricalcium silicate crystalline phase by differential scanning calorimetry for the development of endodontic calcium silicate-based cements. J Therm Anal Calorim 147, 2083–2090 (2022). https://doi.org/10.1007/s10973-021-10581-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-021-10581-0