Abstract
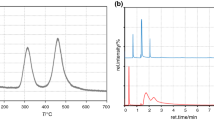
To develop thermal stable flavor of pyrazines, the two new flavor precursors of 3,6-dimethyl-2,5-pyrazinedicarboxylic acid leaf alcohol ester (DPALE) and 3,6-dimethyl-2,5-pyrazinedicarboxylic acid menthyl ester (DPAME) were synthesized by KMnO4 oxidation, acylating chlorination, and esterification reaction. TG, DSC and Py-GC/MS were conducted to investigate the thermal decomposition process and the pyrolysis products of DPALE and DPAME. TG–DTG curves showed that T p of DPALE and DPAME with the largest mass loss rate were 304.9 and 319.9 °C, respectively. The T peak of DPALE and DPAME measured by DSC were 307.1 and 327.7 °C, respectively. The pyrolysis products were qualitatively and semiquantitatively analyzed by Py-GC/MS technology at 300, 600, and 900 °C. The results indicated that 600 °C was better than the other two temperatures to release specific aromas of leaf alcohol, 2-methylpyrazine, 2,5-dimethylpyrazine, menthol, and 3-p-menthene. Finally, the thermal behavior and the thermal decomposition mechanism of the flavor precursors of DPALE and DPAME were discussed. The study on the thermal behavior and pyrolysis products of these two flavor precursors of pyrazine esters would have them applied in food stuffs and tobacco to provide specific flavor of leaf alcohol, menthol, and alkylpyrazines.






Similar content being viewed by others
References
Müller R, Rappert S. Pyrazines: occurrence, formation and biodegradation. Appl Microbiol Biotechnol. 2010;85:1315–20.
Zhang ZH, Yu SZ, Wang ZT, Zhao BL, Hou JW, Yang FJ, Xin WJ. Scavenging effects of tetramethylpyrazine on active oxygen free radicals. Acta Pharmacol Sin. 1994;15:229–31.
Liu SY, Sylvester DM. Antiplatelet activity of tetramethylpyrazine. Thromb Res. 1994;75:51–62.
Mihara S, Masuda H, Tateba H, Tuda T. Olfactive properties of 3-substituted 5-alkyl-2-methylpyrazines. J Agric Food Chem. 1991;39:1262–4.
Talhout R, Opperhuizen A, Van Amsterdam JGC. Sugars as tobacco ingredient: effects on mainstream smoke composition. Food Chem Toxicol. 2006;44:1789–98.
Jiang L, Kubota K. Formation by mechanical stimulus of the flavor compounds in young leaves of Japanese pepper (Xanthoxylum piperitum DC.). J Agric Food Chem. 2001;49:1353–7.
Kobayashi A, Kubota K, Joki Y, Wada E, Wakabayashi M. (Z)-3-Hexenyl-β-D-glucopyranoside in fresh tea leaves as a precursor of green odor. Biosci Biotechnol Biochem. 1994;58:592–3.
Serra S, Fuganti C, Brenna E. Biocatalytic preparation of natural flavours and fragrances. Trends Biotechnol. 2005;23:193–8.
Eccles R. Menthol and related cooling compounds. J Pharm Pharmacol. 1994;46:618–30.
Watson HR, Hems R, Rowsell DG, Spring DJ. New compounds with the menthol cooling effect. J Soc Cosmet Chem. 1978;29:185–200.
Xie WC, Yang XH, Zhang CH, Tan ZC, Xia YM. Investigation on pyrolysis behavior and decomposition compounds of three synthesized glycosidically bond flavor precursors. Thermochim Acta. 2012;532:2–9.
Cardillo P, Gigante L, Lunghi A, Zanirato P. Revisiting the thermal decomposition of five ortho-substituted phenyl azides by calorimetric techniques. J Therm Anal Calorim. 2010;100:191–8.
Bolbukh Y, Tertykh V, Klonos P, Pissis P. DSC study of polyhydroxyethylmethacrylate filled with modified silicas. J Therm Anal Calorim. 2012;108:1111–9.
Ahamad T, Alshehri SM. Thermal degradation and evolved gas analysis of epoxy (DGEBA)/novolac resin blends (ENB) during pyrolysis and combustion. J Therm Anal Calorim. 2013;111:445–51.
Liu X, Gu S, Xu W. Thermal and structural characterization of superfine down powder. J Therm Anal Calorim. 2013;111:259–66.
Saponar A, Popovici EJ, Perhaita I, Nemes G, Cadis AI. Thermal behaviour of some ester derivatives of p-tert-butyl calix [n] arene. J Therm Anal Calorim. 2012;110:349–56.
Worzakowska M. Synthesis, characterization, and thermal properties of new flavor compounds. J Therm Anal Calorim. 2014;116:727–36.
Worzakowska M, Ścigalski P. Synthesis and thermal behavior of linear neryl diesters in inert and oxidative atmosphere. J Therm Anal Calorim. 2014;115:783–92.
Madarász J. Evolved gas analyses on a mixed valence copper (I, II) complex salt with thiosulfate and ammonia by in situ TG-EGA-FTIR and TG/DTA-EGA-MS. J Therm Anal Calorim. 2009;97:111–6.
Czylkowska A. New complexes of heavy lanthanides with 4,4′-bipyridine and trichloroacetates. J Therm Anal Calorim. 2012;110:1299–308.
Xie WC, Gu XH, Tan ZC, Tang J, Wang GY, Luo CR, Sun LX. Thermal decomposition of two synthetic glycosides by TG, DSC and simultaneous Py-GC-MS analysis. J Therm Anal Calorim. 2007;87:505–10.
Xie W, Tang J, Gu X, Luo C, Wang G. Thermal decomposition study of menthyl-glycoside by TGA/SDTA, DSC and simultaneous Py-GC-MS analysis. J Anal Appl Pyrol. 2007;78:180–4.
Zhu P, Sui S, Wang B, Sun K, Sun G. A study of pyrolysis and pyrolysis products of flame-retardant cotton fabrics by DSC, TGA, and PY-GC-MS. J Anal Appl Pyrol. 2004;71:645–55.
Lizarraga E, Zabaleta C, Palop JA. Thermal decomposition and stability of quinoline compounds using thermogravimetry and differential scanning calorimetry. Thermochim Acta. 2005;427:171–4.
Zeng S, Sun S, Liu S, Hu J, He B. Synthesis of α-ionyl-β-D-glucoside and its property of flavor release. J Therm Anal Calorim. 2014;115:1049–56.
Huang YD, Tu W, Yuan YQ, Fan DL. Novel organogelators based on pyrazine-2,5-dicarboxylic acid derivatives and their mesomorphic behaviors. Tetrahedron. 2014;70:1274–82.
Lai M, Zhao B, Bao X, Zhao M, Ji X, Fu P, Zhang Y. Pyrolysates of novel latent fragrant compound 3,6-dimethyl-2,5-pyrazinedicarboxylic acid menthol ester. Chin J Chromatogr. 2015;33:46–51 (In Chinese, with English abstract).
Holmelid B, Kleinert M, Barth T. Reactivity and reaction pathways in thermochemical treatment of selected lignin-like model compounds under hydrogen rich conditions. J Anal Appl Pyrol. 2012;98:37–44.
Paine JB, Pithawalla YB, Naworal JD. Carbohydrate pyrolysis mechanisms from isotopic labeling: part 4. The pyrolysis of d-glucose: the formation of furans. J Anal Appl Pyrol. 2008;83:37–63.
Dai R, Ye L, Luo A, Fu R, Zhang S, Xie G, Jin S. The investigation of pyrolysis mechanism of heptakis [3-O-methyl-2,6-di-O-(methoxybenzyl)]-β-cyclodextrin by Py-GC and Py-GC/MS. J Anal Appl Pyrol. 1997;42:9–19.
Wang S, Liu B, Su Q. Pyrolysis–gas chromatography/mass spectrometry as a useful technique to evaluate the pyrolysis pathways of phenylalanine. J Anal Appl Pyrol. 2004;71:393–403.
Cai JB, Wei SP, Su QD. Thermal decomposition of diphenethyl carbonate by pyrolysis-gas chromatography/mass spectroscopy. Asian J Chem. 2010;22:8192–8.
Acknowledgements
This work was supported by Key Science and Technology Program of Science & Technology Department of Henan Province (Nos. 132102210042, 122102210129).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lai, M., Zhao, B., Ji, X. et al. Thermal behavior of two synthesized flavor precursors of pyrazine esters. J Therm Anal Calorim 123, 479–487 (2016). https://doi.org/10.1007/s10973-015-4886-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-015-4886-4