Abstract
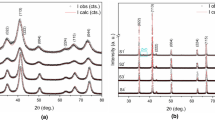
Nanosize Co1−x Zn x Fe2O4 (x = 0, 0.1, 0.3, and 0.4) have been synthesized by the precursor combustion technique via autocatalytic combustion of the mixed-metal fumarato-hydrazinate precursors. A key feature of these precursors is that they decompose autocatalytically once ignited to give the monophasic nanocrystalline ferrite. This fact is confirmed by X-ray powder diffraction analysis. The thermal decomposition pattern of the precursors has been studied by thermogravimetric and differential thermal analysis. The precursors have also been characterized by FTIR and chemical analysis to fix the chemical composition. The Curie temperature (T c) of the “as-prepared” oxide was determined by alternating current susceptibility measurements.





Similar content being viewed by others
References
Ramana Reddy AV, Ranga Mohan G, Ravinder D, Boyanov BS. High-frequency dielectric behaviour of polycrystalline zinc substituted cobalt ferrites. J Mat Sci. 1999;34:3169–76.
Josyulu OS, Sobhanadri J. DC conductivity and dielectric behaviour of cobalt-zinc ferrites. Phy Stat Sol (a). 1980;59:323–9.
Veverka M, Veverka P, Jirak Z, Kaman O, Knizek K, Marysko M, Pollert E, Zaveta K. Synthesis and magnetic properties of Co1−x Zn x Fe2O4 nanoparticles as materials for magnetic fluid hyperthermia. J Magn Magn Mater. 2010;322:2386–9.
Matsushita N, Ichinose M, Nakagawa S, Naoe M. Co–Zn ferrite films prepared by facing targets sputtering system for longitudinal recording layer. J Magn Magn Mater. 1999;193:68–70.
Mukherjee K, Majumdar SB. Hydrogen sensing characteristics of wet chemical synthesized tailored Mg0.5Zn0.5Fe2O4 nanostructures. Nanotechnology. 2010;21:255504.
Gedam NN, Padole PR, Rithe SK, Chaudhari GN. Ammonia gas sensor based on a spinel semiconductor, Co0.8Ni0.2Fe2O4 nanomaterial. J Sol-Gel Sci Tech. 2009;50:296–300.
Chen Z, Gao L. Synthesis and magnetic properties of CoFe2O4 nanoparticles by using PEG as surfactant additive. Mat Sci Eng B. 2007;141:82–6.
Vital A, Angermann A, Dittmann R, Graule T, Topfer J. Highly sinter-active (Mg–Cu)–Zn ferrite nanoparticles prepared by flame spray synthesis. Acta Mater. 2007;55:1955–64.
Hua ZH, Chen RS, Li CL, Yang SG, Lu M, Gu XB, Du YW. CoFe2O4 nanowire arrays prepared by template-electrodeposition method and further oxidization. J Alloys Compd. 2007;427:199–203.
Thakur S, Katyal SC, Singh M. Structural and magnetic properties of nano nickel–zinc ferrite synthesized by reverse micelle technique. J Magn Magn Mat. 2009;321:1–7.
Maensiri S, Masingboon C, Boonchom B, Seraphin S. A simple route to synthesize nickel ferrite (NiFe2O4) nanoparticles using egg white. Scripta Mater. 2007;56(9):797–800.
Jiang J. A facile method to the Ni0.8Co0.2Fe2O4 nanocrystalline via a refluxing route in ethylene glycol. Mater Lett. 2007;61:3239–42.
Singhal S, Singh J, Barthwal SK, Chandra K. Preparation and characterization of nanosize nickel-substituted cobalt ferrites (Co1-x Ni x Fe2O4). J Sol State Chem. 2005;178:3183–9.
Verenkar VMS, Rane KS. Thermal and electrothermal analysis (ETA) of Iron (II) carboxylato-hydrazinates Part I – Ferrous fumarato-hydrazinate and ferrous succinato-hydrazinate. In: Dharwadkar SR, Bharadwaj SR, Mukherjee SK, Sood DD, editors. Proceedings of the 10th national symposium on thermal analysis, thermans. Kanpur: Indian Thermal Analysis Society; 1995. p. 171–4.
Verenkar VMS, Rane KS. Synthesis, characterization and thermal analysis of ferrous malato-hydrazinate. In: Ravindran PV, Sudersanan M, Misra NL, Venugopal V, editors. Proceedings of the 12th national symposium on thermal analysis, thermans. Gorakhpur: Indian Thermal Analysis Society; 2000. p. 194–7.
Sawant SY, Verenkar VMS, Mojumdar SC. Preparation, thermal, XRD, chemical and FTIR spectral analysis of NiMn2O4 nanoparticles and respective precursor. J Therm Anal Calorim. 2007;90:669–72.
Gonsalves LR, Verenkar VMS, Mojumdar SC. Preparation and characterization of Co0.5Zn0.5Fe2(C4H2O4)3.6N2H4 A precursor to prepare Co0.5Zn0.5Fe2O4 nanoparticles. J Therm Anal Calorim. 2009;96(1):53–7.
Gonsalves LR, Verenkar VMS, Mojumdar SC. Synthesis of cobalt nickel ferrite nanoparticles via autocatalytic decomposition of the precursor. J Therm Anal Calorim. 2010;100:789–92.
Gonsalves LR, Verenkar VMS, Mojumdar SC. Synthesis and characterization of Co0.8Zn0.2Fe2O4 nanoparticles. J Therm Anal Calorim. 2011;104:869–73.
More A, Verenkar VMS, Mojumdar SC. Nickel ferrite nanoparticles synthesis from novel fumarato-hydrazinate precursor. J Therm Anal Calorim. 2008;94(1):63–7.
Porob RA, Khan SZ, Mojumdar SC, Verenkar VMS. Synthesis, TG, DSC and infrared spectral study of NiMn2(C4H4O4)3.6N2H4: a precursor for NiMn2O4 nano-particles. J Therm Anal Calorim. 2006;86(3):605–8.
Sawant SY, Kannan KR, Verenkar VMS. Synthesis, characterisation and thermal analysis of nickel manganese fumarato-hydrazinate. In: Pillai CGS, Ramakumar KL, Ravindran PV, Venugopal V, editors. Proceedings of the 13th national symposium on thermal analysis, B.A.R.C. Mumbai: Indian Thermal Analysis Society; 2002. p. 154–5.
Jeffery GH, Bassett J, Mendham J, Denney RC. Vogel’s text book of quantitative inorganic analysis. 5th ed. England: Longman; 1989.
Braibanti A, Dallavalle F, Pellinghelli MA, Leporati E. The nitrogen–nitrogen stretching band in hydrazine derivatives and complexes. Inorg Chem. 1968;7:1430–3.
Gul IH, Abbasi AZ, Amin F, Anis-ur-Rehman M, Maqsood A. Structural, magnetic and electric properties of Co1−x Zn x Fe2O4 synthesized by co-precipitation method. J Magn Magn Mater. 2007;311:494–9.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gonsalves, L.R., Verenkar, V.M.S. Synthesis and thermal studies of the cobalt zinc ferrous fumarato-hydrazinate. J Therm Anal Calorim 108, 871–875 (2012). https://doi.org/10.1007/s10973-012-2343-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-012-2343-1