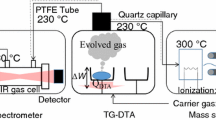
Abstract
The influences of atmospheric CO2 and H2O on the kinetics of the thermal decomposition of zinc carbonate hydroxide, Zn5(CO3)2(OH)6, were investigated by means of controlled rate evolved gas analysis (CREGA) coupled with TG. Although CO2 and H2O were evolved simultaneously in a single mass-loss step of the thermal decomposition, different effects of those evolved gases on the kinetic rate behavior were observed. No distinguished effect of atmospheric CO2 was detected within the possible range of self-generated CO2 concentration. On the other hand, apparent acceleration effect by the increase in the concentration of atmospheric H2O was observed as the reduction of reaction temperature during the course of constant rate thermal decomposition. The catalytic effect was characterized by the decrease in the apparent activation energy for the established reaction with increasing the concentration of atmospheric H2O, accompanied by the partially compensating decrease in the pre-exponential factor.
Similar content being viewed by others
References
N. Koga and H. Tanaka, Thermochim. Acta, 388 (2002) 41.
H. Tanaka and N. Koga, J. Thermal Anal., 36 (1990) 2601.
N. Koga and J. M. Criado, Int. J. Chem. Kinet., 30 (1998) 737.
J. Rouquerol, J. Thermal Anal., 2 (1970) 123.
J. Paulik and F. Paulik, Anal. Chim. Acta, 56 (1971) 328.
O. T. Sorensen and J. Rouquerol, Eds, Sample Controlled Thermal Analysis, Kluwer, Dordrecht 2003.
N. Koga, J. M. Criado and H. Tanaka, Thermochim. Acta, 340–341 (1999) 387.
N. Koga, J. M. Criado and H. Tanaka, J. Therm. Anal. Cal., 60 (2000) 943.
N. Koga and S. Yamada, Int. J. Chem. Kinet., 37 (2005) 346.
N. Koga and H. Tanaka, J. Therm. Anal. Cal., 82 (2005) 725.
N. Koga and Y. Yamane, J. Therm. Anal. Cal., 93 (2008) 963.
S. Yamada and N. Koga, Thermochim. Acta, 431 (2005) 38.
J. M. Criado, L. A. Perez-Maqueda, M. J. Dianez and P. E. Sanchez-Jimenez, J. Therm. Anal. Cal., 87 (2007) 297.
B. Topley and M. L. Smith, J. Chem. Soc., (1935) 321.
D. A. Young, Decomposition of Solids, Pergamon, Oxford 1966.
M. E. Brown, D. Dollimore and A. K. Galwey, Reactions in the Solid State, Elsevier, Amsterdam 1980.
A. K. Galwey and M. E. Brown, Thermal Decomposition of Ionic Solids, Elsevier, Amsterdam 1999.
M. Reading, D. Dollimore, J. Rouquerol and F. Rouquerol, J. Thermal Anal., 29 (1984) 775.
T. Ozawa, J. Thermal Anal., 31 (1986) 547.
N. Koga, Thermochim. Acta, 258 (1995) 145.
T. Ozawa, Bull. Chem. Soc. Jpn., 38 (1965) 1881.
T. Ozawa, J. Thermal Anal., 2 (1970) 301.
T. Ozawa, Thermochim. Acta, 100 (1986) 109.
F. J. Gotor, J. M. Criado, J. Malek and N. Koga, J. Phys. Chem. A, 104 (2000) 10777.
J. M. Criado, L. A. Perez-Maqueda, F. J. Gotor, J. Malek and N. Koga, J. Therm. Anal. Cal., 72 (2003) 901.
D. M. Bates and D. G. Watts, Nonlinear Regression and its Applications, Wiley, New York 1988.
N. Koga, A. Mako, T. Kimizu and Y. Tanaka, Thermochim. Acta, 467 (2008) 11.
N. Koga, Thermochim. Acta, 244 (1994) 1.
A. K. Galwey and M. Mortimer, Int. J. Chem. Kinet., 38 (2006) 464.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yamada, S., Tsukumo, E. & Koga, N. Influences of evolved gases on the thermal decomposition of zinc carbonate hydroxide evaluated by controlled rate evolved gas analysis coupled With TG. J Therm Anal Calorim 95, 489–493 (2009). https://doi.org/10.1007/s10973-008-9272-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-008-9272-z