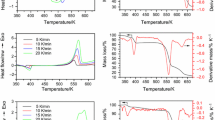
Abstract
The constant-volume combustion energies of the lead salts of 2-hydroxy-3,5-dinitropyridine (2HDNPPb) and 4-hydroxy-3,5-dinitropyridine (4HDNPPb), ΔU c (2HDNPPb(s) and 4HDNPP(s)), were determined as –4441.92±2.43 and –4515.74±1.92 kJ mol–1 , respectively, at 298.15 K. Their standard enthalpies of combustion, Δc m H θ(2HDNPPb(s) and 4HDNPPb(s), 298.15 K), and standard enthalpies of formation, Δr m H θ(2HDNPPb(s) and 4HDNPPb(s), 298.15 K) were as –4425.81±2.43, –4499.63±1.92 kJ mol–1 and –870.43±2.76, –796.65±2.32 kJ mol–1 , respectively. As two combustion catalysts, 2HDNPPb and 4HDNPPb can enhance the burning rate and reduce the pressure exponent of RDX–CMDB propellant.
Similar content being viewed by others
References
FQ Zhao P Chen Y Luo RE Zhang SW Li MZ Deng YM Zheng (2003) Chinese J. Explosives Propellants 26 1 Occurrence Handle1:CAS:528:DC%2BD2cXivFeqs70%3D
P Chen FQ Zhao Y Luo RZ Hu YM Zheng MZ Deng Y Gao (2004) Acta Chim. Sinica 62 1197 Occurrence Handle1:CAS:528:DC%2BD2cXlvVOnsL8%3D
A Singnor E Scoffone L Biondi S Bezzi (1963) Gazz Chim. Ital. 93 65
SP Chen XW Yang SL Gao RZ Hu QZ Shi (2004) J. Therm. Anal. Cal. 76 265 Occurrence Handle1:CAS:528:DC%2BD2cXjvFSrsrY%3D Occurrence Handle10.1023/B:JTAN.0000027825.55555.a6
XF Yang XW Yang LZ Sun (1986) Chem. J. Chin.Univ. 7 363 Occurrence Handle1:CAS:528:DyaL2sXls1KqsA%3D%3D
MM Popiv (1954) Thermometry and Calorimetry Moscow University Publishing House Moscow 382
X. F. Guo, W. B. Yang and S. Y. Guo, CN Patent 1034 200A, 1989-07-26.
JD Cox (1978) J. Chem. Thermodyn. 10 903 Occurrence Handle1:CAS:528:DyaE1cXmt1WqsLg%3D Occurrence Handle10.1016/0021-9614(78)90050-2
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fengqi, Z., Hongxu, G., Yang, L. et al. Constant-volume combustion energy of the lead salts of 2HDNPPb and 4HDNPPb and their effect on combustion characteristics of RDX–CMDB propellant. J Therm Anal Calorim 85, 791–794 (2006). https://doi.org/10.1007/s10973-005-7455-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-005-7455-4