Abstract
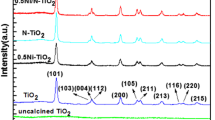
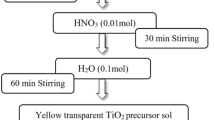
A nitrogen doped TiO2/Ni0.5Zn0.5Fe2O4 core–shell structure nanoparticles was prepared by low temperature sol–gel-hydrothermal process. The characterizations of the catalyst indicate that the Ni0.5Zn0.5Fe2O4 nanocrystals of about 25 nm are well-coated with crystalline N-doped titania. The absorption edges in the diffusion reflectance spectra of TiO0.98N1.02 and TiO1.37N0.63/Ni0.5Zn0.5Fe2O4 shift to visible light region. The core–shell nanocatalysts can effectively photodegrade organic pollutants in the dispersion system and can be recycled easily by an external magnetic field.








Similar content being viewed by others
References
Huang Y, Ho W (2008) Langmuir 24:3510–3516
Livraghi S, Chierotti MR (2008) J Phys Chem C 112:17244–17252
Chi B, Zhao L (2007) J Phys Chem C 111:6189
Zhang Z, Goodall JBM (2009) J Euro Ceram Soc 29:2343–2353
Wu YM, Liu HB (2009) J Phys Chem C 113:14689–14695
Joshi MM, Labhsetwar NK (2009) Appl Catal A: Gen 357:26–33
Kuroda Y, Mori T, Yagi K (2005) Langmuir 21:8026–8034
Yin S, Ihara K (2006) Sol Sta Commun 137:132–137
Wang ZP, Cai WM (2005) Appl Catal B Environ 57:223–231
Li D, Ohashi N (2005) J Solid State Chem 178:3293–3302
Napoli F, Chiesa M (2009) Chem Phys Lett 477:135–138
Izumi Y, Itoi T (2009) J Phys Chem C 113:6706–6718
Emeline AV, Kuzmin GN (2008) Chem Phys Lett 454:279–283
Li D, Haneda H (2005) Mater Sci Eng B 117:67–75
Irie H, Watanabe Y (2003) J Phys Chem B 107:5483–5486
Gole JL, Stout JD (2004) J Phys Chem B 108:1230–1240
Hoffmann MR, Martin ST (1995) Chem Rev 95(1):69–96
Chen XB, Burda C (2004) J Phys Chem B 108:15446–15449
Sakthivel S, Janczarek M (2004) J Phys Chem B 108:19384–19387
Beydoun D, Amal R (2000) J Phys Chem B 104:4387–4396
Gao Y, Chen B (2003) Mater Chem Phys 80:348–355
Beydoun D, Amal R (2002) Mater Sci Eng B Solid 94:71–81
Rana S, Srivastava RS (2005) Mater Sci Eng B 119:144–151
Jeon S, Braun PV (2003) Chem Mater 15:1256–1263
Asilturk M, Sayılkan F (2006) J Hazard Mater B129:164–170
Yu JG, Su YR (2006) J Mol Catal A Chem 258:104–112
Li ZJ, Hou B (2005) J Colloid Interface Sci 288:149–154
Ho WK, Yu JC (2006) J Sol Sta Chem 179:1171–1176
Sayılkan F, Erdemoglu S (2006) Mater Res Bull 41:2276–2285
Kim DS, Kwak SY (2007) Appl Catal A Gen 323:110–118
Fu WY, Yang HB (2006) Mater Lett 60:2723–2727
Hanh N, Quy OK (2003) Phys B Condens Matter 327:382–384
Tang JJ, Wang YJ (2008) Chin J Process Eng 8:172–176 (in Chinese)
Horikawa T, Katoh M (2008) Micro Meso Mater 110:397–404
Li HX, Li JX (2006) J Phys Chem B 110:1559–1565
Asahi R, Morikawa T (2001) Science 293:269–271
Gyorgy E, Perez del Pino A (2003) Surf Coat Technol 173:265–270
Rodriguez JA, Jirsak T (2000) J Phys Chem B 104:319–328
Wang Y, Feng CX (2006) J Mol Catal A Chem 260:1–3
Kim TK, Lee MN (2005) Thin Solid Films 475:171–177
Li YJ, Li XD (2006) Water Res 40:1119–1126
Balaji S, Kalai Selvan R (2005) Mater Sci Eng B 119:119–124
Beydoun D, Amal R (2002) J Mol Catal A Chem 180:193–200
Acknowledgments
We are grateful to the financial supports from the National Natural Science Foundation of China (No. 40830748), the fund of China ‘211’ engineering construction and the open foundation from key laboratory of catalysis and materials science of Hubei in China.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shen, X., Wang, Y.X., Lu, L.Q. et al. Preparation and optical properties of TiO2−x N x /Ni0.5Zn0.5Fe2O4 core–shell nanocatalyst from sol–gel-hydrothermal process. J Sol-Gel Sci Technol 54, 340–346 (2010). https://doi.org/10.1007/s10971-010-2201-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10971-010-2201-1