Abstract
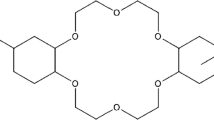
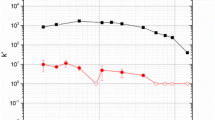
The extraction of 224Ra and 228Ac with dibenzo-21-crown-7 resin was studied in solutions with Rose Bengal and identical solutions without Rose Bengal (blanks) from pH 3 to 12. Batch studies demonstrated that actinium is extracted at high pH (> 10) from both solutions but with a significantly higher extraction from the Rose Bengal solutions (maximum extraction ~ 60,000) than the blank solutions (maximum extraction ~ 2,000). In the Rose Bengal solutions, there was a weak extraction of actinium at lower pH (4–10) as well. There was no uptake of radium from the blank solutions, and a weak extraction at all pH levels from the Rose Bengal solutions. Kinetics studies were performed for actinium only, as radium had no significant extraction. Two column studies are demonstrated for the separation of radium from actinium. These separations are moderately effective with ~ 78% of the actinium recovered with no detectable radium.






Similar content being viewed by others
References
Filosofov D, Lebedev N, Radchenko V, Rakhimov A, Happel S, Roesch F (2015) Behavior of actinium, alkaline and rare earth elements in Sr- resin/mineral acid systems. Solvent Extr Ion Exch 00:1–14
Chiarizia R, Horwitz EP, Dietz M, Cheng Y (1998) Radium separation through complexation by aqueous crown ethers and extraction by dinonylnaphthalenesulfonic acid. React Funct Polym 38(2–3):249–257
Kmak KN, Shaughnessy DA, Vujic J (2021) Extraction of radium and actinium with Pb resin and Rose Bengal. JRNC 328:377–385
Thiele N, Wilson J (2018) Actinum-225 for targeted alpha therapy: coordination chemistry and current chelation approaches. Cancer Biother Radiopharm 33(8):336–347
Gott M, Steinbach J, Mamat C (2016) The radiochemical and radiopharmaceutical applications of radium. Open Chem 14(1):118–129
Bond A, Dietz M, Chiarizia R (2000) Incorporating size selectivity into synergistic solvent extraction: a review of crown ether-containing systems. Ind Eng Chem Res 39(10):3442–3464
Saleh M, Salhin A, Saad B (1995) Determination of lanthanum in monazite sand after selective solvent extraction separation of the picrate with dibenzo-[24]-CROWN-8. Analyst 120:2861–2865
Parham H, Fazeli A (2000) Extraction-spectrophotometric determination of trace amounts of barium by 18-crown-6 and Rose Bengal. Anal Sci 16:575–577
Horwitz E, Dietz M, Rhoads S, Felinto C, Gale N, Houghton J (1994) A lead-selective extraction chromatographic resin and its application to the isolation of lead from geological samples. Anal Chim Acta 292:263–273
Despotopulos JD, Kmak KN, Moody K, Shaughnessy D (2018) "Development of a 212Pb and 212Bi generator for homolog studies of flerovium and moscovium. JRNC 317:473–477
National Nuclear Data Center “NNDC” (2019) Brookhaven National Laboratory. https://www.nndc.bnl.gov/nudat2/indx_dec.jsp. Accessed 22 March 2022
Yankovskaya V, Dovhyi I, Milyutin V, Nekrasova N, Bezhin N, Lyapunov A (2017) Separation of cobalt from thiocyanate solutions by crown ether-based impregnated sorbents. JRNC 314:119–125
Despotopulos J, Kmak K, Gharibyan N, Henderson R, Moody K, Shaughnessy D, Sudowe R (2016) Characterization of the homologs of flerovium with crown ether based extraction chromatography resins: studies in hydrochloric acid. J Radioanal Nucl Chem 310:1201–1207
Horwitz EP, Dietz M, Chiarizia R, Diamond H, Essling A, Graczyk D (1992) Separation and preconcentration of uranium from acidic media by extraction chromatography. Anal Chim Acta 266:25–37
Horwitz EP, Chiarizia R, Dietz M (1992) A novel strontium-selective extraction chromatographic resin. Solvent Extr Ion Exch 10:313–336
Imura H, Saito Y, Ohashi K, Meguro Y, Yoshida Z, Choppin G (1996) Characterization of the Lathanum(III) and Europium(III) tricholoacetate complexes extracted with 18-crown-6. Solvent Extr Ion Exch 14(5):817–832
Zielinska B, Bilewicz A (2004) The hydrolysis of actinium. J Radioanal Nucl Chem 261(1):195–198
Frensdorff H (1971) Stability constants of cyclic polyether complexes with univalent cations. J Am Chem Soc 93(3):600–606
Deblonde G, Zavarin M, Kersting A (2021) The coordination properties and ionic radius of actinium: a 120-year-old enigma. Coord Chem Rev 446:214130
McDowell W, Arnsten B, Case G (1989) The synergistic solvent extraction of radium from alkaline nitrate media by dicyclohexano-21-crown-7 combined with 2-methyl-2-heptyl nonanoic acid equilibrium reactions and metal ion competition. Solvent Extr Ion Exch 7(3):377–393
McDowell W, Moyer B, Case G, Case F (1986) Selectivity in solvent extraction of metal ions by organic cation exchanges synergized by macrocycles: factors relating to macrocycle size and structure. Solvent Extr Ion Exch 4(2):217–236
Acknowledgements
This study was performed under the auspices of the U.S. Department of Energy by Lawrence Livermore National Laboratory under Contract DE-AC52-07NA27344. The authors would like to thank Carlos Valdez for providing the Rose Bengal.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kmak, K.N., Despotopulos, J.D. Extraction of radium and actinium with dibenzo-21-crown-7 resin and Rose Bengal. J Radioanal Nucl Chem 331, 3175–3181 (2022). https://doi.org/10.1007/s10967-022-08368-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-022-08368-0