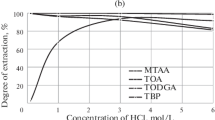
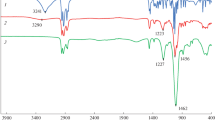
Abstract
Di-(2-ethylhexyl)phosphoric acid, HDEHP, is a well-known extractant used for metal ion extraction in an industrial scale and for research work. Understanding metal extraction mechanisms is crucial for accurate interpretation of data obtained from solvent extraction experiments. The extraction mechanism of many metals by HDEHP has been determined. However, the mechanism for Tc(IV) extraction by HDEHP has not been specifically described in the literature and various assumptions have been made on the identity of the extracted Tc(IV) complex, without convincing demonstration. This work reports that TcO(OH)(DEHP)(HDEHP) is the Tc(IV) complex extracted by HDEHP into dodecane from an aqueous solution of 3 M NaClO4 at pH 2, with one HDEHP dimer participating in the extraction and one proton undergoing transfer from the HDEHP into the aqueous phase. The proposed mechanism agrees with other established metal hydroxide mechanisms and follow overall extraction trends for tetravalent metals with HDEHP.
Similar content being viewed by others
References
El-Sweify FH, Kamel MW (1996) Studies on the extraction behavior of Zr(IV), Ce(III), Th(IV) and U(VI) from aqueous solutions of Arzenazo-I with HDEHP, HTTA, TDA and TCMA. J Radioanal Nucl Chem 207:369–382
Lohithakshan KV, Patil P, Aggarwal SK (2014) Solvent extraction studies of plutonium(IV) and americium(III) in room temperature ionic liquid (RTIL) by di-2-ethyl hexyl phosphoric acid (HDEHP) as extractant. J Radioanal Nucl Chem 301:153–157. https://doi.org/10.1007/s10967-014-3119-9
Reddy L, Reddy B, Harikrishna K, Venugopal Chetty K (1994) Solvent extraction of Hf(IV) from mixed electrolyte solutions into di-2-ethylhexylphosphoric acid (HDEHP). J Radioanal Nucl Chem 178:173–178
Shakir K (1992) Extraction of certain actinide and lanthinide elements from different acid media by polyurethane foams loaded with di-2-ethylhexylphosphoric acid (HDEHP). J Radioanal Nucl Chem 162:371–380
Islam F, Biswas RK (1980) Kinetics of solvent extraction of metal ions with HDEHP - II Kinetics and mechanism of solvent extraction of V(IV) from acid aqueous solutions with bis-(2-ethylhexyl) phosphoric acid in benzene. Journal of ionrganic and nuclear chemistry 42:421–429
Rydberg Michael; Musikas, Claude; Choppin, Gregory R. J cox, (2004) Solvent Extraction Principles and Practice. Marcel Dekker, New York
Peppard DF, Mason GW, McCarty S (1960) Extraction of thorium(IV) by di-esters of orthophosphoric acid, (GO)2PO(OH). J Inorg Nucl Chem 1:138–150
Cecilie M, Hiscox B, Nash KL (2012) Characterization of HDEHP-lanthanide complexes formed in a non-polar organic phase using 31 P NMR and ESI-MS. Dalton Trans 41:1054–1064. https://doi.org/10.1039/c1dt11534k
Lieser KH (1993) Technetium in the nuclear fuel cycle, in medicine and in the environment. Radiochim Acta 63:5–8
Hess NJ, Xia Y, Rai D, Conradson SD (2004) Thermodynamic model for the solubility of TcO2·xH2O(am) in the aqueous Tc(IV) - Na+- Cl– H+- OH– H2O system. J Solution Chem 33:199–226. https://doi.org/10.1023/B:JOSL.0000030285.11512.1f
Guillaumont R, Fanghanel T, Neck V et al (2003) Update on the chemical thermodynamics of uranium, neptunium, plutonium, americium and technetium. Chemical Thermodynamics 5:919
Yalçintaş E, Gaona X, Altmaier M et al (2016) Thermodynamic description of Tc(IV) solubility and hydrolysis in dilute to concentrated NaCl, MgCl2and CaCl2solutions. Dalton Trans 45:8916–8936. https://doi.org/10.1039/c6dt00973e
Omoto T, Wall NA (2017) Evaluation of vanadium(IV) as a non-radioactive surrogate for technetium(IV) by comparison of stability constants for polyamino polycarboxylate ligand complexation. J Solution Chem 46:1981–1994. https://doi.org/10.1007/s10953-017-0680-1
Wall NA, Karunathilake N, Dong W (2013) Interactions of Tc(IV) with citrate in NaCl media. Radiochim Acta. https://doi.org/10.1524/ract.2013.2001
Wall NA, Mathews SA (2005) Sustainability of humic acids in the presence of magnesium oxide. Appl Geochem. https://doi.org/10.1016/j.apgeochem.2005.04.004
Boggs MA, Islam M, Dong W, Wall NA (2013) Complexation of Tc(IV) with EDTA at varying ionic strength of NaCl. Radiochim Acta. https://doi.org/10.1524/ract.2013.2000
Xia Y, Hess NJ, Felmy AR (2006) Stability constants of technetium(IV) oxalate complexes as a function of ionic strength. Radiochim Acta 94:137–141. https://doi.org/10.1524/ract.2006.94.3.137
Parker TG, Omoto T, Dickens SM et al (2018) Complexation of Tc(IV) with SO42- in NaCl Medium. J Solution Chem 47:1192–1201. https://doi.org/10.1007/s10953-018-0785-1
Shannon BYRD, H M, Baur NH, et al (1976) Revised effective ionic radii and systematic studies of interatomie distances in halides and chaleogenides
Hu G, Chen D, Wang L et al (2014) Extraction of vanadium from chloride solution with high concentration of iron by solvent extraction using D2EHPA. Separ Purif Technol 125:59–65. https://doi.org/10.1016/j.seppur.2014.01.031
Xing-bin LI, Chang WEI, Jun WU et al (2012) Thermodynamics and mechanism of vanadium (IV) extraction from sulphate medium with D2EHPA, EHEHPA and CYANEX 272 in kerosene. Trans Nonferr Met Soc China 22:461–466. https://doi.org/10.1016/S1003-6326(11)61199-0
Zhang Y, Zhang T, Lv G et al (2016) Synergistic extraction of vanadium ( IV ) in sulfuric acid media using a mixture of D2EHPA and EHEHPA. Hydrometallurgy 166:87–93. https://doi.org/10.1016/j.hydromet.2016.09.003
Biswas RK, Begum DA (1998) Solvent extraction of tetravalent titanium from chloride solution by di-2-ethylhexyl phosphoric acid in kerosene. Hydrometallurgy 1:263–274
Liem DH, Sinegribova O (1971) Synergistic and antagonistic effects in th extraction of Hf(IV) by dibutylphosphate (HDBP) and di(2-ethyl-hexyl)phosphate (HDEHP) in the presence of trioctylphosphineoxide (TOPO) or trioctylamine (TOA) in hexane and toluene. Acta Chem Scand 25:301–322
Vermaak V, Krieg HM, De Beer L, Van der Westhuizen D (2018) Mechanistic study of hafnium and zirconium extraction with organophosphorous extractants. Solvent Extr Ion Exch 36:150–161. https://doi.org/10.1080/07366299.2018.1431078
Abdel-Fattah AA, Ali SM, El-Sweify FH (2002) Thermodynamics of the solvent extraction of Zr ( IV ) by Amberlite LA-2, TBP and HDEHP from different aqueous media. J Radioanal Nucl Chem 253:465–475
Koladkar DV, Dhadke PM (2002) Extraction and separation of Th (IV) and U (VI) from nitric acid media using PIA-8 and HDEHP. J Radioanal Nucl Chem 253:297–302
Peppard DF, Mason GW (1963) Some mechanisms of extraction of M(II), (III), (IV), and (VI) metals by acidic organophosphorous extractants. Nucl Sci Eng 16:382–399
Ali SM (2008) Extraction thermodynamics of Th (IV) in various aqueous organic systems. J Chem Thermodyn 40:798–805. https://doi.org/10.1016/j.jct.2008.01.013
Joseph Steigman WCE (1992) The Chemistry of Technetium in Medicine
Tang J, C.M. W, (1986) Solvent extraction of lanthanides with a crown ether carboxylic acid. J Anal Chem 58:3233–3235. https://doi.org/10.1021/ac00127a068
Friend MT, Eiroa-Lledo C, Lecrivain LM et al (2018) Thermodynamic parameters for the complexation of technetium(IV) with EDTA. Radiochim Acta 106:963–970. https://doi.org/10.1515/ract-2018-2992
Stary J (1964) Composition and stability of metal chelates. In: The solvent extraction of metal chelates. pp 4–20
Favre-Reguillon A, Fiaty K, Laurent P et al (2007) Solid/liquid extraction of zirconium and hafnium in hydrochloric acid aqueous solution with anion exchange resin s kinetic study and equilibrium analyses. Ind Eng Chem Res 46:1286–1291. https://doi.org/10.1021/ie060444p
Rydberg J (1956) Studies on the formation of composite complexes by means of an extractive technique. J R Netherl Chem Soc 75:737–742
Rossotti F, Rossotti H (1961) The determination of stability constants
Rydberg J (1954) Studies on the extraction of metal complexes. XII-A Arkic Kemi 8:101
Andersson C, Andersson SO, Liljenzin JO et al (1969) Solvvent extraction studies by the AKUFVE Method 1969.pdf. Acta Chem Scand 23:2781–2796
There H (1976) Hydrolysis of Cu (II)
Ronald N. S, Malcolm R. D (1978) The hydrolysis of metal ions. Part 1. Copper (II). JCS Daltion 232–235
Hess NJ, Qafoku O, Xia Y, et al (2008) Thermodynamic Model for the Solubility of TcO 2 · x H 2 O in Aqueous Oxalate Systems. 1471–1487. https://doi.org/10.1007/s10953-008-9328-5
Parker L, Lledo EC, Tabada DL, Wall DE, Wall NA, TGL (2019) Comparison of the solvent extraction behavior of ReO4- and TcO4-. Radiochim Acta. https://doi.org/10.1515/ract-2019-0006
Partridge JA, Jensen RC (1969) Purification of di-(2-ethylhexyl)phosphoric acid by precipitation of copper(II) di-(2-ethylhexyl)phosphate. J Inorg Nucl Chem 31:2587–2589
J⊘rgensen CK, Schwochau K, (1965) Comparative interpretation of absorption spectra of technetium (iv) and rhenium (iv) hexahalides. Zeitschrift fur Naturforschung - Section A J Phys Sci 20:65–75. https://doi.org/10.1515/zna-1965-0112
Gu B, Dong W, Liang L, Wall NA (2011) Dissolution of Technetium ( IV ) Oxide by Natural and Synthetic Organic Ligands under both Reducing and Oxidizing Conditions. 4771–4777. https://doi.org/10.1021/es200110y
Yalcintas E, Gaona X, Scheinost AC et al (2015) Redox chemistry of Tc(VII)/Tc(IV) in dilute to concentrated NaCl and MgCl2solutions. Radiochim Acta 103:57–72. https://doi.org/10.1515/ract-2014-2272
Sillén LG (1956) On equilibria with polynuclear complexes. Recueil des travaux chimiques des Pays-Bas 75:705–710
Banda R, Lee MS (2015) Solvent extraction for the separation of Zr and Hf from aqueous solutions. Separ Purif Rev 44:199–215. https://doi.org/10.1080/15422119.2014.920876
Miralles N, Sastre A, Martinez M, Aguilar M (1992) The aggregation of organophosphorus acid compounds in toluene. Anal Sci 8:773–777
Acknowledgements
This work was funded by the U.S. National Nuclear Security Administration, under the SSAA Grant DE-NA0002916.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Eiroa-Lledo, C., Wall, D.E. & Wall, N.A. Extraction mechanism of Tc(IV) by Di-(2-ethylhexyl)phosphoric acid (HDEHP). J Radioanal Nucl Chem 331, 2751–2760 (2022). https://doi.org/10.1007/s10967-022-08303-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-022-08303-3