Abstract
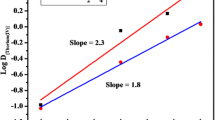
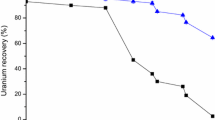
The analytical technique has been developed for the extraction and determination of uranium(VI). This process is based on the ion-pair complex formation of uranium(VI) with 2-octylaminopyridine in xylene. Uranium(VI) is quantitatively extracted by optimizing the parameters of solvent extraction in presence of sodium acetate, such as pH, concentration of weak organic acids, strippant, shaking period, solvent study, and concentration of extractant. A mechanism of extraction was proposed based on the slope ratio analysis method. The interference of various cations and anions was also investigated. The versatility of the developed method was investigated by employing it to binary and ternary mixtures. The robustness of the method was demonstrated by determining uranium(VI) in the bone sample.







Similar content being viewed by others
References
Seaborg GT, Katz JJ (1957) The actinide elements. McGraw-Hill, New York
Madrakian T, Afkhami A, Mousavi A (2007) Spectrophotometric determination of trace amounts of uranium(VI) in water samples after mixed micelle-mediated extraction. Talanta 71:610–614
Miyake M, Sugimara Y, Mayeda M (1970) The Uranium content and the activity ratio 234U/238U in marine organisms and sea water in the western north pacific. J Oceanogr Soc Jpn 26:123–129
Shamsipur M, Saeidi M, Yari A, Yaganeh-Faal A, Mashhadizadeh MH, Azimi G, Naeimi H, Sharghi H (2004) UO22+ Ion-selective membrane electrode based on a Naphthol-Derivative Schiff’s Base 2,2’-[1,2-Ethandiyl bis(nitriloethylidene)]bis(1-naphthalene). Bull Korean Chem Soc 25:629
Shannon SS (1977) The HSSR programme and its relation to the nure effort symposium on hydrogeochemical and stream sediment reconnaissance for uranium in the unites states, grant junction, CO
Carboni M, Abney CW, Liu SB, Lin WB (2013) Highly porous and stable metal-organic frameworks for uranium extraction. Chem Sci 4:2396–2402
Sather AC, Berrymanb OB, Rebek J (2013) Jr. Selective recognition and extraction of the uranyl ion from aqueous solutions with a recyclable chelating resin. Chem Sci 4:3601–3605
El-Taher A (2010) INAA and DNAA for uranium determination in geological samples from Egypt. Appl Radiat Isot 68:1189–1192
El-Taher A, Nossair A, Azzam AH, Kratz KL, Abdel-Halim AS (2004) Determination of traces of uranium and thorium in some egyptian environmental matrices by instrumental neutron activation analysis (pp). J Environ Prot Eng 30:19–30
Afzal M, Hanif J, Saleem M, Hanif I, Ahmed R (1991) Estimation of titanium and iron in uranium by EDXRF using microdroplets on filter paper. J Radioanal Nucl Chem 152:251–259
Shrivastav P, Menon SK, Agrawal YK (2001) Selective extraction and inductively coupled plasma atomic emission spectrophotometric determination of thorium using chromogenic crown ether. J Radioanal Nucl Chem 250:459–464
Freitas MC, Hipolito CS (2007) NAA and PIXE for the determination of the contents of extractable sediment. J Radioanal Nucl Chem 271:179–183
Fujino O, Umetani S, Uenoa E, Shigeta K, Matsuda T (2000) Determination of uranium and thorium in apatite minerals by inductively coupled plasma atomic emission spectrometry with solvent extraction separation into diisobutyl ketone. Anal Chim Acta 420:65–71
Tomé FV, Blanco Rodríguez MP, Lozano JC (2001) Study of the representativity of Uranium and Thorium assays in soil and sediment samples by alpha spectrometry. Appl Radiat Isot 56:393–398
Nakashima T, Taketatsu YT (1992) Determination of uranium(VI) in seawater by ion-exchanger phase absorptiometry with arsenazo-III. Talanta 39:523–527
Gupta KK, Kulkarni PG, Singh RK (1993) Spectrophotometric determination of uranium using ascorbic acid as a chromogenic reagent. Talanta 40:507–510
Hirano Y, Ogawa Y, Ogama K (2003) Simultaneous spectrophotometric determination of uranium and thorium by flow injection analysis using selective masking. Anal Sci 19:303–307
Suresh A, Patre DK, Srinivasan TG, Vasudeva Rao PR (2002) A new procedure for the spectrophotometric determination of uranium(VI) in the presence of a large excess of thorium(IV). Spectrochim Acta Part A 58:341–347
Abbas MN, Homoda AM, Mostafa GAE (2001) First derivative spectrophotometric determination of uranium(VI) and vanadium(V) in natural and saline waters and some synthetic matrices using PAR and cetylpyridinum chloride. Anal Chim Acta 436:223–231
Alyapyshev MY, Babain VA, Antonov NG, Smirnov IV (2006) Extraction of Americium and Europium from perchloric acid solutions with N, N-Dialkyl and N, N, N, N-Tetraalkylpyridine-2,6-dicarboxamides. Russ J Appl Chem 79:1808–1835
Alyapyshev MY, Babain VA, Smirnov IV, Shadrin AY (2006) Separation of americium and europium from solutions of nitric and perchloric acid using dipicolinic acid diamides. Czch J Phys 56:D469–D475
Biswas S, Pathak PN, Singh DK, Roy SB, Manchanda VK (2010) Synergistic extraction of uranium with mixtures of PC88A and neutral oxodonors. J Radioanal Nucl Chem 284:13–19
Rajeshawari B, Dhavale BA, Bangia TR, Mathur JN, Page AG (2002) Role of Cyanex-272 as an extractant for uranium in the determination of rare earths by ICP-AES. J Radioanal Nucl Chem 254:479–483
Marckzenko Z (1976) Spectrophotometric determination of trace elements, 1st edn. Ellis Hardwood Ltd., John Wiley and Sons, Chichester
Bosch NA, Petrukhin OM (1978) 2-octylaminopyridine a new extractant. Zh Anal Chim 33:1805
Sandell EB (1965) Colorimetric determination of traces of metals, 3rd edn. Interscience Publishers Inc, New York
Flaschka HA, Bernard AJ (1972) Chelates in analytical chemistry of elements, vol 4. Marcel Dekker Inc, New York, p 140
Anuse MA, Chavan MB (1984) Studies on extraction separation of platinum metals and gold(III) with pyrimidine thiol. Spectrophotometric determination of palladium(II), osmium(VIII) and ruthenium(III). Chem Anal (Warsaw) 29:409–420
Acknowledgements
The author would like to thanks to UGC for providing teacher fellowship for completion of this work and special thanks to UGC-SAP-DRS-II and DST-FIST level-I for providing instrumentation facilities.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kore, G.D., Zanje, S.B., Kokare, A.N. et al. Liquid–liquid extraction of uranium(VI) from weak sodium acetate medium using 2-octylaminopyridine: real sample analysis. J Radioanal Nucl Chem 329, 975–982 (2021). https://doi.org/10.1007/s10967-021-07828-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-021-07828-3