Abstract
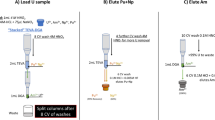
A new method was developed to effectively separate Am(III) from Cm(III). Am(III) was selectively oxidized to Am(V) using a mixture of Na2S2O8, Ag(I), and NaOCl in 0.01 M HNO3. Cm(III) was selectively retained on a DGA resin, while Am(V) had no retention. A separation factor of 110 ± 20 was usually obtained from a single separation. The new separation method was applied to determine Cm isotopes 244, 245 and 246 by accelerator mass spectrometry (AMS) in spent nuclear fuel samples.




Similar content being viewed by others
References
Dares CJ, Lapides AM, Mincher BJ, Meyer TJ (2015) Electrochemical oxidation of 243Am(III) in nitric acid by a terpyridyl-derivatized electrode. Science 350:652–655
Kamoshida M, Fukasawa T (1996) Solvent Extraction of Americium(VI) by Tri-n-Butyl Phosphate. J Nucl Sci Technol 33(5):403–408
Chapron S, Marie C, Pacary V, Duchesne M-T, Arrachart G, Pellet- Rostaing S, Miguirditchian M (2016) Separation of Americium by Liquid-Liquid Extraction Using Diglycolamides Water-Soluble Complexing Agents. Procedia Chem 21:133–139
Geist A, Taylor R, Ekberg C, Guilbaud P, Modolo G, Bourg S (2016) The SACSESS hydrometallurgy domain—an overview. Proc Chem 21:218–222
Burns JD, Shehee TC, Clearfield A, Hobbs DT (2012) Separation of americium from curium by oxidation and ion exchange. Anal Chem 84:6930–6932
Modolo G, Geist A, Miguirditchian M (2015) Reprocessing and recycling of spent. Nucl Fuel Chap. https://doi.org/10.1016/B978-1-78242-212-9.00010-1
Burns JD, Moyer BA (2016) Group hexavalent actinide separations: a new approach to used nuclear fuel recycling. Inorg Chem 55:8913–8919
Whitney SM, Biegalski S, Buchholz B (2007) Analyzing nuclear fuel cycles from isotopic ratios of waste products applicable to measurement by accelerator mass spectrometry. Nucl Sci Eng 157:200–209
Recommended data, Laboratoire National Henri Becquerel, http://www.nucleide.org/DDEP_WG/DDEPdata.htm. Accessd Feb 2018
Mincher BJ, Schmitt NC, Schuetz BK, Shehee TC, Hobbs DT (2015) Recent advances in f-element separations based on a new method for the production of pentavalent americium in acidic solution. RSC Adv 34(5):27205–27210
Poupard D, Journaux B (1990) Determination of picogram quantities of americium and curium by thermal ionization mass spectrometry (TIMS). Radiochim Acta 49:25–28
Chartier F, Aubert M, Pilier M (1999) Determination of Am and Cm in spent nuclear fuels by isotope dilution inductively coupled plasma mass spectrometry and isotope dilution thermal ionization mass spectrometry after separation by high-performance liquid chromatography. Fresenius J Anal Chem 364(4):320–327
Dai X, Christl M, Kramer-Tremblay S, Hans-Arno S (2016) Determination of Atto- to Femtogram Levels of Americium and Curium Isotopes in Large-Volume Urine Samples by Compact Accelerator Mass Spectrometry. Anal Chem 88:2832–2837
Christl M, Dai X, Lachner J, Kramer-Tremblay S, Hans-Arno S (2014) Low energy AMS of americium and curium. Nucl Instrum Meth B 331:225–232
Myasoedov BF, Maryutina TA, Litvina MN, Malikov DA, Kulyako YuM, Spivakov BY, Hill C, Adnet J-M, Lecomte M, Madic C (2005) Americium(III)/curium(III) separation by countercurrent chromatography using malonamide extractants. Radiochim Acta 93:9–15
Šťastná K, John J, Ŝebesta F, Vlk M (2015) Separation of curium from americium using composite sorbents and complexing agent solutions. J Radioanal Nucl Chem 304:349–355
Schweitzer GK, Pesterfield LL (2010) The aqueous chemistry of the elements. Oxford University Press, New York
Runde WH, Mincher BJ (2011) Higher oxidation states of americium: preparation, characterization and use for separations. Chem Rev 111:5723–5741
Glenn AB (1968) Separation of americium from curium by precipitation of K3Am O2(CO3)2. Nucl Appl 4:217–221
Burns JD, Shehee TC, Clearfield A, Hobbs DT (2012) Separation of americium from curium by oxidation and ion exchange. Anal Chem 84:6930–6932
Richards JM, Sudowe R (2016) Separation of americium in high oxidation states from curium utilizing sodium bismuthate. Anal Chem 88:4605–4608
Burns JD, Borkowski M, Clearfield A, Reed DT (2012) Separation of oxidized americium from lanthanides by use of pillared metal(IV) phosphate-phosphonate hybrid materials. Radiochim Acta 100:901–906
Christl M, Vockenhuber C, Kubik PW, Wacker L, Lachner J, Alfimov V, Synal HA (2013) The ETH Zurich AMS facilities: performance parameters and reference materials. Nucl Instrum Meth B 294:29–38
Horwitz EP, McAlister DR, Bond AH, Barrans RE (2005) Novel extraction of chromatographic resins based on tetraalkyldiglycolamides: characterization and potential applications. Solvent Extra Ion Exch 23(3):319–344
Dai X (2011) Isotopic uranium analysis in urine samples by alpha spectrometry. J Radioanal Nucl Chem 289(2):595–600
Separation of Am from rare earths, Eichrom methods (2014) http://www.eichrom.com/eichrom/radiochem/methods/eichrom/index.aspx. Access Feb 2018
Acknowledgements
This research was undertaken for Atomic Energy of Canada limited and funded by the Canadian Federal Science and Technology program for Project 51200.50.18.06.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kazi, Z., Guérin, N., Christl, M. et al. Effective separation of Am(III) and Cm(III) using a DGA resin via the selective oxidation of Am(III) to Am(V). J Radioanal Nucl Chem 321, 227–233 (2019). https://doi.org/10.1007/s10967-019-06571-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-019-06571-0