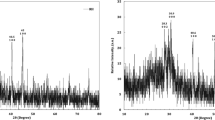
Abstract
The present research work presents the study of cost effective cellulose fibers as sorbent for the U(VI) removal from nuclear wastewaters. Alkali-treated cellulose fibers (ATCFs) were extracted from alkaline peroxide mechanical pulp board by using CH3COOH and NaOH treatment. Sorption capability was ascertained by conducting batch experiments. The kinetic sorption of U(VI) on ATCFs could be described by the pseudo-second-order rate equation well. The results demonstrated that the sorption of U(VI) to ATCFs was strongly dependent of pH and weakly dependent of ionic strength. U(VI) sorption on ATCFs was mainly dominated by the outer-sphere complexation.










Similar content being viewed by others
References
Wang XK, Chen CL, Hu WP, Ding AP, Xu D, Zhou X (2005) Sorption of 243Am(III) to multi-wall carbon nanotubes. Environ Sci Technol 39:2856–2860
Zhao YG, Li JX, Zhang SW, Wang XK (2014) Amidoxime-functionalized magnetic mesoporous silica for selective sorption of U(VI). RSC Adv 4:32710–32717
Zhao YG, Li JX, Zhao LP, Zhang SW, Huang YS, Wang XL, Wang XK (2014) Synthesis of amidoxime-functionalized Fe3O4@SiO2 core-shell magnetic microspheres for highly efficient sorption of U(VI). Chem Eng J 235:275–283
Dabrowski A, Hubicki Z, Podkoscielny P, Robens E (2004) Selective removal of the heavy metal ions from waters and industrial wastewaters by ion-exchange method. Chemosphere 56:91–106
Zhao YG, Li JX, Zhang SW, Chen H, Shao DD (2013) Efficient enrichment of uranium(VI) on amidoximated magnetite/graphene oxide composites. RSC Adv 3:18952–18959
Yuan LY, Liu YL, Shi WQ, Li ZJ, Lan JH, Feng YX, Zhao YL, Yuan YL, Chai ZF (2012) A novel mesoporous material for uranium extraction, dihydroimidazole functionalized SBA-15. J Mater Chem 22:17019–17026
Seyhan SB, Nursel PB (2013) The uranium recovery from aqueous solutions using amidoxime modified cellulose derivatives. IV. Recovery of uranium by amidoximated hydroxypropyl methylcellulose. Cellulose 20:827–839
Melo DQ, Vidal CB, Silva ALD (2014) Removal of Cd2+, Cu2+, Ni2+, and Pb2+ Ions from aqueous solutions using tururi fibers as an adsorbent. J Appl Polym Sci 131:40883–40895
Shibi IG, Anirudhan TS (2002) Synthesis, characterization and application as a mercury(II) sorbent of banana stalk (musa paradisiaca)-polyacrylamide grafted copolymer bearing carboxyl groups. Ind Eng Chem Res 41:5341–5352
Li W, Zhao X, Liu SX (2013) Preparation of long and entangled nanocellulose fibers aerogels from poplar alkaline peroxide mechanical pulp and its magnetic functional property as matrix. Carbohyd Polym 94:278–285
Zhang J, Zhai JJ, Zhao FZ, Tao ZY (1999) Study of soil humic substances by cross-polarization magic angle spinning 13C nuclear magnetic resonance and pyrolysis-capillary gas chromatography. Anal Chim Acta 378:177–182
Chen CL, Hu J, Xu D, Tan XL, Meng YD, Wang XK (2008) Surface complexation modeling of Sr(II) and Eu(III) adsorption onto oxidized multiwall carbon nanotubes. J Colloid Interface Sci 323:33–41
Shao DD, Hou GS, Li JX, Wen T, Ren XM, Wang XK (2014) PANI/GO as a super adsorbent for the selective adsorption of uranium(VI). Chem Eng J 255:604–612
Nishiyama Y, Sugiyama J, Chanzy H, Langan P (2002) Crystal structure and hydrogen bonding system in cellulose Iβ from synchrotron X-ray and neutron fiber diffraction. J Am Chem Soc 124:9074–9082
Gao Y, Shao ZY, Xiao ZH (2015) U(VI) sorption on illite: effect of pH, ionic strength, humic acid and temperature. J Radioanal Nucl Chem 303:867–876
Sun YB, Wang Q, Chen CL, Tan XL, Wang XK (2012) Interaction between Eu(III) and graphene oxide nanosheets investigated by batch and extended X-ray absorption fine structure spectroscopy and by modeling techniques. Environ Sci Technol 46:6020–6027
Yang SB, Hu J, Chen CL, Shao DD, Wang XK (2011) Mutual effect of Pb(II) and humic acid adsorption onto multiwalled carbon nanotubes/poly(acrylamide) composites from aqueous solution. Environ Sci Technol 45:3621–3627
Ren XM, Wang SW, Yang ST, Li JX (2010) Influence of contact time, pH, soil humic/fulvic acids, ionic strength and temperature on sorption of U(VI) onto MX-80 bentonite. J Radioanal Nucl Chem 283:253–259
Shao DD, Jiang ZQ, Wang XK, Li JX, Meng YD (2009) Plasma induced grafting carboxymethyl cellulose on multiwalled carbon nanotubes for the removal of UO2 2+ from aqueous solution. J Phys Chem B 113:860–864
Sheng GD, Yang ST, Sheng J, Hu J, Tan X, Wang XK (2011) Macroscopic and microscopic investigation of Ni(II) sequestration on diatomite by batch, XPS, and EXAFS techniques. Environ Sci Technol 45:7718–7726
Kowal-Fouchard A, Drot R, Simoni E, Ehrhardt JJ (2004) Use of spectroscopic techniques for uranium(VI)/montmorillonite interaction modeling. Environ Sci Technol 38:1399–1407
Niu Z, Fan QH, Wang W, Xu J, Chen L, Wu W (2009) Effect of pH, ionic strength and humic acid on the sorption of uranium(VI) to attapulgite. Appl Radiat Isot 67:1582–1590
Chen CL, Hu J, Shao DD, Li JX, Wang XK (2009) Adsorption behavior of multiwall carbon nanotube/iron oxide magnetic composites for Ni(II) and Sr(II). J Hazard Mater 164:923–928
Yang ST, Li JX, Shao DD, Hu J, Wang XK (2009) Adsorption of Ni(II) on oxidized multi-walled carbon nanotubes, effect of contact time, pH, foreign ions and PAA. J Hazard Mater 166:109–116
Xu D, Tan XL, Chen CL, Wang XK (2008) Removal of Pb(II) from aqueous solution by oxidized multiwalled carbon nanotubes. J Hazard Mater 154:407–417
Xu D, Tan XL, Chen CL, Wang XK (2008) Adsorption of Pb(II) from aqueous solution to MX-80 bentonite: effect of pH, ionic strength, foreign ions and temperature. Appl Clay Sci 41:37–46
Yan WL, Bai RB (2005) Adsorption of lead and humic acid on chitosan hydrogel beads. Water Res 39:688–698
Montavon G, Markai S, Andres Y, Grambow B (2002) Complexation studies of Eu(III) with alumina-bound polymaleic acid: effect of organic polymer loading and metal ion concentration. Environ Sci Technol 36:3303–3309
Zhao GX, Li JX, Ren XM, Chen CL, Wang XK (2011) Few-layered graphene oxide nanosheets as superior sorbents for heavy metal ion pollution management. Environ Sci Technol 45:10454–10462
Tan XL, Fang M, Chen CL, Yu SM, Wang XK (2008) Counterion effects of nickel and sodium dodecylbenzene sulfonate adsorption to multiwalled carbon nanotubes in aqueous solution. Carbon 46:1741–1750
Liu X, Xu YB, Jin RC, Yin P, Sun LX, Liang T, Gao SM (2014) Facile synthesis of hierarchical Fe4(P2O7)3 for removal of U(VI). J Mol Liq 200:311–318
Fan QH, Hao LM, Wang CL, Zheng Z, Liu CL, Wu WS (2014) The adsorption behavior of U(VI) on granite. Environ Sci Proc Impact 16:534–541
Du YF, Yin ZX, Wu HY, Li P, Qi W, Wu WS (2015) Sorption of U(VI) on magnetic illite: effects of pH, ions, humic substances and temperature. J Radioanal Nucl Chem 304:793–804
Xiao J, Chen YT, Zhao WH, Xu JB (2013) Sorption behavior of U(VI) onto Chinese bentonite: effect of pH, ionic strength, temperature and humic acid. J Mol Liq 188:178–185
Zong PF, Wang H, Pan H, Zhao YL, He CH (2013) Application of NKF-6 zeolite for the removal of U(VI) from aqueous solution. J Radioanal Nucl Chem 295:1969–1979
Zhao YL, Zong PF, Li YH, Li K, Zhao XH, Wang H, Liu SH, Sun YB, He CH (2015) Fabrication of oxidized multiwalled carbon nanotubes for the immobilization of U(VI) from aqueous solutions. J Radioanal Nucl Chem 305:361–369
Pan S, Zhang Y, Shen H, Hu M (2012) An intensive study on the magnetic effect of mercapto-functionalized nano-magnetic Fe3O4 polymers and their adsorption mechanism for the removal of Hg(II) from aqueous solution. Chem Eng J 210:564–574
Sari A, Tuzen M (2009) Biosorption of As(III) and As(V) from aqueous solution by macrofungus (inonotus hispidus) biomass: equilibrium and kinetic studies. J Hazard Mater 164:1372–1378
Acknowledgments
The National Natural Science Foundation of China (41273134 and 21477133) is acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhu, K., Chen, C., Li, J. et al. Alkali-treated cellulose fibers for U(VI) separation and enrichment. J Radioanal Nucl Chem 308, 981–990 (2016). https://doi.org/10.1007/s10967-015-4527-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-015-4527-1