Abstract
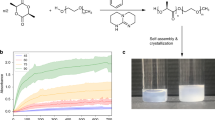
Block copolymerization is already a proven technique to manipulate the properties of polymeric materials. Herein, our work presents a facile synthesis of high molecular weight polyglycolide-b-poly(L-)lactide-b-polyglycolide (b-PLLGA) triblock copolymers by ring-opening polymerization (ROP) in bulk at considerably low temperature. A series of ABA-type triblock copolymers was successfully prepared and microstructural properties were thoroughly investigated against random copolymers of poly(L-lactide-co-glycolide) (r-PLLGA). This synthesis method led to the preparation of high molecular weight b-PLLGAs with highly defined chain microstructure and an intrinsic viscosity [η] was obtained as high as 1.62 dl/g when [M]:[I] was [1]:[1000]. The investigations revealed that the prepared copolymers are in block type chain microstructure, having exclusive thermal transitions and sequence/segment properties. DSC analysis revealed that two glass (Tg) transitions and two melt transitions (Tm) exclusively associated to PGA block and PLLA block. Results were further confirmed by homosequenses of –LLLL- and -GGGG- from 13C-NMR spectroscopy. Tensile results revealed that b-PLLGA have higher tensile strength and Young’s modulus as compared to r-PLLGA. Moreover, b-PLLGAs are more thermally stable and have a wider thermal processing window.







Similar content being viewed by others
References
Ikada Y, Tsuji H (2000) Biodegradable polyesters for medical and ecological applications. Macromol Rapid Commun 21:117–132. https://doi.org/10.1002/(SICI)1521-3927(20000201)21:3<117::AID-MARC117>3.0.CO;2-X
Vert M (2005) Aliphatic polyesters: great degradable polymers that cannot do everything. Biomacromolecules 6:538–546
Gref R, Minamitake Y, Peracchia MT et al (1994) Biodegradable long-circulating polymeric nanospheres. Science 263:1600–1603. https://doi.org/10.1126/SCIENCE.8128245
Miller RA, Brady JM, Cutright DE (1977) Degradation rates of oral resorbable implants (polylactates and polyglycolates): rate modification with changes in PLA/PGA copolymer ratios. J Biomed Mater Res 11:711–719. https://doi.org/10.1002/jbm.820110507
Uhrich KE, Cannizzaro SM, Langer RS, Shakesheff KM (1999) Polymeric systems for controlled drug release. Chem Rev 99:3181–3198. https://doi.org/10.1021/cr940351u
Vey E, Rodger C, Meehan L et al (2012) The impact of chemical composition on the degradation kinetics of poly(lactic-co-glycolic) acid copolymers cast films in phosphate buffer solution. Polym Degrad Stab 97:358–365. https://doi.org/10.1016/j.polymdegradstab.2011.12.010
Weiss R (1991) Implants: how big a risk? Science (80-) 252:1060
Babu A, Amreddy N, Muralidharan R et al (2017) Chemodrug delivery using integrin-targeted PLGA-chitosan nanoparticle for lung cancer therapy. Sci Rep 7:14674. https://doi.org/10.1038/s41598-017-15012-5
Springer C, Benita S, Sherman Y et al (2005) Poly-lactic-glycolic acid microspheres: a biodegradable marker for the diagnosis of aspiration in hamsters. Pediatr Res 58:537–541. https://doi.org/10.1203/01.pdr.0000176910.62067.a0
Zhang H, Mao X, Zhao D et al (2017) Three dimensional printed polylactic acid-hydroxyapatite composite scaffolds for prefabricating vascularized tissue engineered bone: an in vivo bioreactor model. Sci Rep 7:15255. https://doi.org/10.1038/s41598-017-14923-7
Li J (2010) Self-assembled supramolecular hydrogels based on polymer–cyclodextrin inclusion complexes for drug delivery. NPG Asia Mater 2:112–118. https://doi.org/10.1038/asiamat.2010.84
Lin N, Chen G, Huang J et al (2009) Effects of polymer-grafted natural nanocrystals on the structure and mechanical properties of poly(lactic acid): a case of cellulose whisker- graft -polycaprolactone. J Appl Polym Sci 113:3417–3425. https://doi.org/10.1002/app.30308
Ayyoob M, Kim YJ (2018) Effect of chemical composition variant and oxygen plasma treatments on thewettability of PLGA thin films, synthesized by direct copolycondensation. Polymers (Basel) 10:1132. https://doi.org/10.3390/polym10101132
Masutani K, Kobayashi K, Kimura Y, Lee CW (2018) Properties of stereo multi-block polylactides obtained by chain-extension of stereo tri-block polylactides consisting of poly(L-lactide) and poly(D-lactide). J Polym Res 25. https://doi.org/10.1007/s10965-018-1444-3
Baimark Y, Pasee S, Rungseesantivanon W, Prakymoramas N (2019) Flexible and high heat-resistant stereocomplex PLLA-PEG-PLLA/PDLA blends prepared by melt process: effect of chain extension. J Polym Res:26. https://doi.org/10.1007/s10965-019-1881-7
Adams ML, Lavasanifar A, Kwon GS (2003) Amphiphilic block copolymers for drug delivery. J Pharm Sci 92:1343–1355
Hou X, Li Q, Cao A (2014) Solvent annealing-induced microphase-separation of polystyrene-b- polylactide block copolymer aimed at preparation of ordered nanoparticles/block copolymer hybrid thin film. J Polym Res:21. https://doi.org/10.1007/s10965-014-0491-7
Kumar N, Ravikumar MNV, Domb AJ (2001) Biodegradable block copolymers. Adv Drug Deliv Rev 53:23–44
Jeong B, Bae YH, Lee DS, Kim SW (1997) Biodegradable block copolymers as injectable drug-delivery systems. Nature 388:860–862. https://doi.org/10.1038/42218
Cabral H, Miyata K, Osada K, Kataoka K (2018) Block copolymer micelles in nanomedicine applications. Chem Rev 118:6844–6892. https://doi.org/10.1021/acs.chemrev.8b00199
Ramot Y, Haim-Zada M, Domb AJ, Nyska A (2016) Biocompatibility and safety of PLA and its copolymers. Adv Drug Deliv Rev 107:153–162
Cohn D, Younes H (1988) Biodegradable PEO/PLA block copolymers. J Biomed Mater Res 22:993–1009. https://doi.org/10.1002/jbm.820221104
Zhou S, Deng X, Li X et al (2004) Synthesis and characterization of biodegradable low molecular weight aliphatic polyesters and their use in protein-delivery systems. J Appl Polym Sci 91:1848–1856. https://doi.org/10.1002/app.13385
Mohammadi-Rovshandeh J, Farnia SMF, Sarbolouki MN (2002) Synthesis and characterization of ABA triblock and novel multiblock copolymers from ethylene glycol,L-lactide, and ?-caprolactone. J Appl Polym Sci 83:2072–2081. https://doi.org/10.1002/app.10145
Cohn D, Hotovely Salomon A (2005) Designing biodegradable multiblock PCL/PLA thermoplastic elastomers. Biomaterials 26:2297–2305. https://doi.org/10.1016/j.biomaterials.2004.07.052
Stayshich RM, Meyer TY (2010) New insights into poly(lactic- co -glycolic acid) microstructure: using repeating sequence copolymers to decipher complex NMR and thermal behavior. J Am Chem Soc 132:10920–10934. https://doi.org/10.1021/ja102670n
Li J, Stayshich RM, Meyer TY (2011) Exploiting sequence to control the hydrolysis behavior of biodegradable PLGA copolymers. J Am Chem Soc 133:6910–6913. https://doi.org/10.1021/ja200895s
Li J, Rothstein SN, Little SR et al (2012) The effect of monomer order on the hydrolysis of biodegradable poly(lactic-co-glycolic acid) repeating sequence copolymers. J Am Chem Soc 134:16352–16359. https://doi.org/10.1021/ja306866w
Hamad K, Kaseem M, Ayyoob M et al (2018) Polylactic acid blends: the future of green, light and tough. Prog Polym Sci 85:83–127. https://doi.org/10.1016/j.progpolymsci.2018.07.001
Meduri A, Fuoco T, Lamberti M et al (2014) Versatile copolymerization of glycolide and rac-lactide by dimethyl(salicylaldiminato) aluminum compounds. Macromolecules 47:534–543. https://doi.org/10.1021/ma402174y
Yin H, Wang R, Ge H et al (2015) Synthesis and structure control of l -lactic acid-glycolic acid copolymer by homo-copolymerization. J Appl Polym Sci 132:1–6. https://doi.org/10.1002/app.41566
Kaihara S, Matsumura S, Mikos AG, Fisher JP (2007) Synthesis of poly(L-lactide) and polyglycolide by ring-opening polymerization. Nat Protoc 2:2767–2771. https://doi.org/10.1038/nprot.2007.391
Kenley RA, Lee MO, Mahoney TR, Sanders LM (1987) Poly(lactide-co-glycolide) decomposition kinetics in vivo and in vitro introduction polymers find increasing application in the pharma. Macromolecules 20:2398–2403. https://doi.org/10.1021/ma00176a012
Dorgan JR, Janzen J, Knauss DM et al (2005) Fundamental solution and single-chain properties of polylactides. J Polym Sci Part B Polym Phys 43:3100–3111. https://doi.org/10.1002/polb.20577
Jamshidi K, Hyon S-H, Ikada Y (1988) Thermal characterization of polylactides. Polymer (Guildf) 29:2229–2234. https://doi.org/10.1016/0032-3861(88)90116-4
Cam D, Marucci M (1997) Influence of residual monomers and metals on poly (L-lactide) thermal stability. Polymer (Guildf) 38:1879–1884. https://doi.org/10.1016/S0032-3861(96)00711-2
Ayyoob M, Yang X, Park HJ et al (2019) Synthesis of bioresorbable poly(lactic-co-glycolic acid)s through direct polycondensation: an economical substitute for the synthesis of polyglactin via ROP of lactide and glycolide. Fibers Polym 20:887–895. https://doi.org/10.1007/s12221-019-1143-7
Ayyoob M, Lee DH, Kim JH et al (2017) Synthesis of poly(glycolic acids) via solution polycondensation and investigation of their thermal degradation behaviors. Fibers Polym 18:407–415. https://doi.org/10.1007/s12221-017-6889-1
Jeong J, Ayyoob M, Kim J-H et al (2019) In situ formation of PLA-grafted alkoxysilanes for toughening a biodegradable PLA stereocomplex thin film. RSC Adv 9:21748–21759. https://doi.org/10.1039/C9RA03299A
Wei X-F, Bao R-Y, Cao Z-Q et al (2014) Greatly accelerated crystallization of poly(lactic acid): cooperative effect of stereocomplex crystallites and polyethylene glycol. Colloid Polym Sci 292:163–172. https://doi.org/10.1007/s00396-013-3067-x
Kortaberria G, Jimeno A, Arruti P et al (2007) Molecular dynamics of poly(glycolide-co-L-Lactide) copolymer during isothermal cold crystallization. J Appl Polym Sci 106:584–589. https://doi.org/10.1002/app.26604
Zhang J, Duan Y, Domb AJ, Ozaki Y (2010) PLLA mesophase and its phase transition behavior in the PLLA−PEG−PLLA copolymer as revealed by infrared spectroscopy. Macromolecules 43:4240–4246. https://doi.org/10.1021/ma100301h
Ramdhanie LI, Aubuchon SR, Boland ED et al (2006) Thermal and mechanical characterization of electrospun blends of poly(lactic acid) and poly(glycolic acid). Polym J 38:1137–1145. https://doi.org/10.1295/polymj.PJ2006062
Kricheldorf HR, Mang T, Jonté JM (1984) Polylactones. 1. Copolymerization of glycolide and ϵ-caprolactone. Macromolecules 17:2173–2181. https://doi.org/10.1021/ma00140a051
Gao Q, Lan P, Shao H, Hu X (2002) Direct synthesis with melt polycondensation and microstructure analysis of poly(L-lactic acid-co-glycolic acid). Polym J 34:786–793. https://doi.org/10.1295/polymj.34.786
Dobrzyński P, Kasperczyk J, Bero M (1999) Application of calcium acetylacetonate to the polymerization of glycolide and copolymerization of glycolide with ε-caprolactone and L-lactide. Macromolecules 32:4735–4737. https://doi.org/10.1021/ma981969z
Gorrasi G, Meduri A, Rizzarelli P et al (2016) Preparation of poly(glycolide-co-lactide)s through a green process: analysis of structural, thermal, and barrier properties. React Funct Polym 109:70–78. https://doi.org/10.1016/j.reactfunctpolym.2016.10.002
Dobrzynski P, Kasperczyk J, Janeczek H, Bero M (2002) Synthesis of biodegradable glycolide/l-lactide copolymers using iron compounds as initiators. Polymer (Guildf) 43:2595–2601. https://doi.org/10.1016/S0032-3861(02)00079-4
Farah S, Anderson DG, Langer R (2016) Physical and mechanical properties of PLA, and their functions in widespread applications — a comprehensive review. Adv Drug Deliv Rev 107:367–392
Cameron RE, Kamvari-Moghaddam A (2012) Synthetic bioresorbable polymers. In: Durability and reliability of medical polymers. Woodhead Publishing, pp 96–118
Acknowledgments
This work was supported by the Technology Innovation Program, (10077004), “Development of high-strength/bio-resorbable polyglycolic acid-based block copolymers and their reinforced composite with a tensile strength of 120 MPa or higher for spinal fixation” funded by the Ministry of Trade, Industry, & Energy of Korea.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ayyoob, M., Lee, S. & Kim, Y.J. Well-defined high molecular weight polyglycolide-b-poly(L-)lactide-b-polyglycolide triblock copolymers: synthesis, characterization and microstructural analysis. J Polym Res 27, 109 (2020). https://doi.org/10.1007/s10965-019-2001-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10965-019-2001-4