Abstract
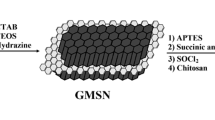
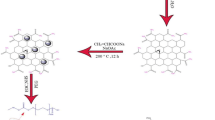
In this study synthesis of a drug delivery system (DDS) is described which has several merits over its counterparts. In order to synthesize this nano-carrier, graphene oxide nano-sheets are used to accommodate MCM-41 nanoparticles. Furthermore Fe3O4 nanoparticles are introduced to this nano-material to produce a traceable nanoparticle. Since cancerous tissues have lower pH than healthy tissues, pH-sensitive oligomers are attached to this nano-material. Finally the nano-carrier is wrapped by a biocompatible shell (PEGylated sodium alginate); this polymeric shell makes the DDS capable of a more controllable drug release. By measuring in vitro situation, ‘loading content%’ and ‘entrapment efficiency%’ proves to be 21 and 93.5 % respectively. In an acidic medium, the drug carrier without a polymeric shell (naked DDS) releases the whole of its drug content in 18 h, while the drug carrier with a polymeric shell (core-shell DDS) releases 45 % of its drug content during 48 h.














Similar content being viewed by others
References
Eid M (2013) J Polym Res 112:1–11
Naahidi S, Jafari M, Edalat F, Raymond K, Khademhosseini A, Chen P (2013) J Control Release 166:182–194
Gillies RJ, Robey I, Gatenby RA (2008) J Nucl Med 49:24–42
Bartil T, Bounekhel M, Cedric C, Jerome R (2007) Acta Pharm 57:301–314
Risbud MK, Hardikar AA, Bhat SV, Bhonde RR (2000) J Control Release 68:23–30
Gupta KC, Kumar MNVR (2000) Polym Int 49:141–146
Singh SK, Singh MK, Kulkarni PP, Sonkar VK, Grácio JJA, Dash D (2012) ACS Nano 6:2731–2740
Deshmukh K, Khatake SM, Joshi GM (2013) J Polym Res 286:1–11
Yang K, Zhang S, Zhang G, Sun X, Lee ST, Liu Z (2010) Nano Lett 10:3318–3323
Wang S (2009) Microporous Mesoporous Mater 117:1–9
Hartmann M (2005) Chem Mater 17:4577–4593
Yiu HHP, Wright PA (2005) J Mater Chem 15:3690–3700
Calero M, Gutierrez L, Salas G, Luengo Y, Lazaro A, Acedo P, Morales MP, Miranda R, Villanueva A (2014) Nanomed Nanotechnol Biol Med 10:733–743
Cornell RM, Schwertmann U (2003) The iron oxides: structure, properties, reactions, occurrences and uses; Wiley: Weinheim,; 2nd Edition, Chapter 1: 3–7
Lu XJ, Forssberg E (2002) Miner Eng 15:755–757
Marcano DC, Kosynkin DV, Berlin JM, Sinitskii A, Sun Z, Slesarev A, Alemany LB, Lu W, Tour JM (2010) ACS Nano 4:4806–4814
Wang G, Yang S, Wei Z, Dong X, Wang H, Qi M (2013) Polym Bull 70:2359–2371
Yin P, Sun N, Deng C, Li Y, Zhang X, Yang P (2013) Proteomics 13:2243–2250
Long D, Li W, Ling L, Miyawaki J, Mochida I, Yoon SH (2010) Langmuir 26:16096–16102
Han Q, Wang Z, Xia J, Chen S, Zhang X, Ding M (2012) Talanta 101:388–395
Gusev VY, Feng X, Bu Z, Haller GL, O’Brien JA (1996) J Phys Chem 100:1985–1988
Zhao D, Huo Q, Feng J, Chmelka BF, Stucky GD (1998) J Am Chem Soc 120:6024–6036
Coates J (2000) Interpretation of infrared spectra, a practical approach. Wiley: Chichester: 10815–10837
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pourjavadi, A., Shakerpoor, A., Tehrani, Z.M. et al. Magnetic graphene oxide mesoporous silica hybrid nanoparticles with dendritic pH sensitive moieties coated by PEGylated alginate-co-poly (acrylic acid) for targeted and controlled drug delivery purposes. J Polym Res 22, 156 (2015). https://doi.org/10.1007/s10965-015-0799-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10965-015-0799-y