Abstract
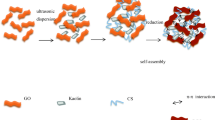
A novel bio-adsorbent, composite hydrogel beads, composed of polyvinyl alcohol (PVA), chitosan (CS) and graphene oxide (GO) was prepared by an instantaneous gelation method. The micromorphology and thermal stability of the PVA/CS/GO hydrogel beads were characterized by scanning electronic microscope (SEM) and thermo gravimetric analysis (TGA) respectively. Adsorption of Cu(II) onto PVA/CS/GO hydrogel beads was investigated with respect to pH, initial concentration, temperature and adsorption time. Adsorption data were well matched by Langmuir isotherm and pseudo-second-order kinetic model at the optimum pH 5.5. The maximum adsorption capacities of PVA/CS/GO hydrogel beads were found to be 162 mg g−1 for Cu(II) at 30 °C, which was much higher than that of PVA/CS hydrogel. Thermodynamic studies indicated that the adsorption was spontaneous and endothermic process in nature. Besides, desorption efficiency and reusability of the adsorbents were assessed on basis of six consecutive adsorption-desorption cycles. Based on these studies, it can be seen that PVA/CS/GO hydrogel beads will be a potential recyclable adsorbent for removal of hazardous metal ions in waste water.










Similar content being viewed by others
References
Zhang Q, Gao Y, Zhai YA, Liu FQ, Gao G (2008) Carbohydr Polym 73:359–363
Brower JB, Ryan RL, Pazirandeh M (1997) Environ Sci Technol 31:2910–2914
Amjal M, Khan AH, Ahmad S (1998) Water Res 32:3085–3091
Li N, Bai N (2005) Sep Purif Technol 45:237–247
Zeng X, Ruckenstein E (1998) J Membr Sci 148(2):195–205
Garg VK, Gupta R, Yadav AB, Kumar R (2003) Bioresour Technol 89:121–124
Allen SJ, McKay G, Porter JF (2004) J Colloid Interface Sci 280(2):322–333
Yi JZ, Zhang LM (2008) Bioresour Technol 99:2182–2186
Akkaya R, Ulusoy U (2008) J Hazard Mater 151:380–388
Li A, Zhang JP, Wang AQ (2007) Bioresour Technol 98:327–332
Yao QX, Xie JJ, Liu JX, Kang HM, Liu Y (2014) J Polym Res 21:465
Wang WB, Kang YR, Wang AQ (2013) J Polym Res 20:101
Liu DG, Li ZH, Ma ZS (2013) Ind Eng Chem Res 52:11036–11044
Cathell MD, Szewczyk JC, Bui FA, Weber CA, Wolever JD, Kang J, Schauer CL (2008) Biomacromolecules 9:289–295
Pandey S, Mishra SB (2011) J Colloid Interface Sci 361:509–520
Li H, Bi S, Liu L, Dong W, Wang X (2011) Desalination 278:397–404
Fan L, Luo C, Lv Z, Lu F, Qiu H (2011) J Hazard Mater 194:193–201
Liu Z, Wang HS, Liu C, Jiang YJ, Yu G, Mu XD, Wang XY (2012) Chem Commun 48:7350–7352
Shen C, Wang YJ, Xu JH, Luo GS (2013) Chem Eng J 229:217–224
Pongjanyakul T, Priprem A, Puttipipatkhachorn S (2005) J Control Release 107:343–356
Wu TM, Wu CY (2006) Polym Degrad Stab 91:2198–2204
Wang SF, Shen L, Tong YJ, Chen L, Phang IY, Lim PQ, Liu TX (2005) Polym Degrad Stab 90:123–131
Saravanan D, Gomathi T, Sudha PN (2013) Int J Biol Macromol 53:67–71
Nesic AR, Velickovic SJ, Antonovic DG (2012) J Hazard Mater 209–210:256–263
Xu D, Wu K, Zhang Q, Hu H, Xi K, Chen Q, Yu X, Chen J, Jia X (2010) Polymer 51:1926–1933
Zhu HY, Fu YQ, Jiang R, Yao J, Xiao L, Zeng GM (2012) Bioresour Technol 105:24–30
Sobahi TR, Makki MSI, Abdelaal MY (2013) J Saudi Chem Soc 17:245–250
Li XL, Li YF, Zhang SD, Ye ZF (2012) Chem Eng J 183:88–97
Chen CH, Wang FY, Mao CF, Yang CH (2007) J Appl Polym Sci 105:1086–1092
Kumar K, Tripathi BP, Shahi VK (2009) J Hazard Mater 172:1041–1048
Dreyer DR, Park S, Bielawski CW, Ruoff RS (2010) Chem Soc Rev 39:228
Madadrang CJ, Kim HY, Gao GH, Wang N, Zhu J, Feng H, Gorring M, Kasner ML, Hou SF (2012) ACS Appl Mater Interfaces 4:1186–1193
Hummers WS, Offeman RE (1958) J Am Chem Soc 80:1339–1339
Marcano DC, Kosynkin DV, Berlin JM, Sinitskii A, Sun ZZ, Slesarev A, Alemany LB, Lu W, Tour JM (2010) ACS Nano 8:4806–4814
Zhang J, Zhang CQ, Madbouly SA (2015) J Appl Polym Sci 132:41751
Becerril HA, Mao J, Liu Z, Stoltenberg RM, Bao Z, Chen Y (2008) ACS Nano 2:463–470
Stankovich S, Dikin DA, Piner RD, Kohlhaas KA, Kleinhammes A, Jia Y, Wu Y, Nguyen SBT, Ruoff RS (2007) Carbon 45:1558–1565
Sankararamakrishnan N, Sharma AK, Sanghi R (2007) J Hazard Mater 148:353–359
Langmuir I (1918) J Am Chem Soc 40:1361–1403
Freundlich H (1932) Trans Faraday Soc 28:195–201
Aksu Z, Tunc O (2005) Process Biochem 40:831–847
Treybal RE. (1981) Mass-Transfer Operations third ed, McGraw-Hill.
Salehi E, Madaeni SS, Rajabi L, Vatanpour V, Derakhshan AA, Zinadini S, Ghorabi S, Ahmadi Monfared H (2012) Sep Purif Technol 89:309–319
Lia XL, Lia YF, Ye ZF (2011) Chem Eng J 178:60–68
Hu X, Wang J, Liu Y, Li X, Zeng G, Bao Z, Zeng X, Chen A, Long F (2011) J Hazard Mater 185:306–314
Li XL, Qi YX, Li YF, Zhang Y, He XH, Wang YH (2013) Bioresour Technol 142:611–619
Acknowledgments
This work was supported by the Research Project of Chinese Ministry of Education (No.113034A), National Natural Science Foundation of China (51373070), Program for New Century Excellent Talents in University (NCET-12-0884).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Li, L., Wang, Z., Ma, P. et al. Preparation of polyvinyl alcohol/chitosan hydrogel compounded with graphene oxide to enhance the adsorption properties for Cu(II) in aqueous solution. J Polym Res 22, 150 (2015). https://doi.org/10.1007/s10965-015-0794-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10965-015-0794-3