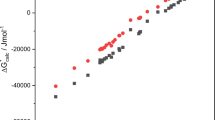
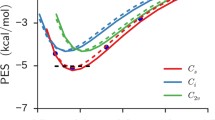
Abstract
Using concepts from perturbation and local molecular field theories of liquids we divide the potential of the SPC/E water model into short and long ranged parts. The short ranged parts define a minimal reference network model that captures very well the structure of the local hydrogen bond network in bulk water while ignoring effects of the remaining long ranged interactions. This deconstruction can provide insight into the different roles that the local hydrogen bond network, dispersion forces, and long ranged dipolar interactions play in determining a variety of properties of SPC/E and related classical models of water. Here we focus on the anomalous behavior of the internal pressure and the temperature dependence of the density of bulk water. We further utilize these short ranged models along with local molecular field theory to quantify the influence of these interactions on the structure of hydrophobic interfaces and the crossover from small to large scale hydration behavior. The implications of our findings for theories of hydrophobicity and possible refinements of classical water models are also discussed.
Similar content being viewed by others
References
Allen, M.P., Tildesley, D.J.: Computer Simulation of Liquids. Oxford, New York (1987)
Ashbaugh, H.S.: Entropy crossover from molecular to macroscopic cavity hydration. Chem. Phys. Lett. 477, 109–111 (2009)
Ashbaugh, H.S., Collett, N.J., Hatch, H.W., Staton, J.A.: Assessing the thermodynamic signatures of hydrophobic hydration for several common water models. J. Chem. Phys. 132, 124504 (2010)
Ben-Naim, A., Stillinger, F.H.: Water and Aqueous Solutions: Structure, Thermodynamics, and Transport Processes. Wiley-Interscience, New York (1972)
Berendsen, H.J.C., Postma, J.P.M., van Gunsteren, W.F., DiNiola, A., Haak, J.R.: Molecular dynamics with coupling to an external bath. J. Chem. Phys. 81, 3684 (1984)
Berendsen, H.J.C., Grigera, J.R., Straatsma, T.P.: The missing term in effective pair potentials. J. Phys. Chem. 91, 6269–6271 (1987)
Berne, B.J., Weeks, J.D., Zhou, R.: Dewetting and hydrophobic interaction in physical and biological systems. Annu. Rev. Phys. Chem. 60, 85–103 (2009)
Buldyrev, S.V., Kumar, P., Debenedetti, P.G., Rossky, P.J., Stanley, H.E.: Water-like solvation thermodynamics in a spherically symmetric solvent model with two characteristic lengths. Proc. Natl. Acad. Sci. USA 104, 20177–20182 (2007)
Chandler, D.: Interfaces and the driving force of hydrophobic assembly. Nature 437, 640–647 (2005)
Chen, Y.G., Weeks, J.D.: Local molecular field theory for effective attractions between like charged objects in systems with strong Coulomb interactions. Proc. Natl. Acad. Sci. USA 103, 7560 (2006)
Chen, Y.G., Kaur, C., Weeks, J.D.: Connecting systems with short and long ranged interactions: local molecular field theory for ionic fluids. J. Phys. Chem. B 108, 19874 (2004)
Denesyuk, N.A., Weeks, J.D.: A new approach for efficient simulation of Coulomb interactions in ionic fluids. J. Chem. Phys. 128, 124109 (2008)
Goharshadi, E.K., Morsali, A., Mansoori, G.A.: A molecular dynamics study on the role of attractive and repulsive forces in internal energy, internal pressure and structure of dense fluids. Chem. Phys. 331, 332–338 (2007)
Guillot, B.: A reappraisal of what we have learnt during three decades of computer simulations of water. J. Mol. Liq. 101, 219–260 (2002)
Hansen, J.P., McDonald, I.R.: Theory of Simple Liquids, 3rd edn. Academic Press, New York (2006)
Haynes, W.M. (ed.): CRC Handbook of Chemistry and Physics, 91st edn. (Internet Version 2011). CRC Press/Taylor and Francis, Boca Raton (2011)
Huang, D.M., Chandler, D.: The hydrophobic effect and the influence of solute-solvent attractions. J. Phys. Chem. B 106, 2047–2053 (2002)
Huang, D.M., Geissler, P.L., Chandler, D.: Scaling of hydrophobic solvation free energies. J. Phys. Chem. B 105, 6704–6709 (2001)
Iordanov, T.D., Schenter, G.K., Garrett, B.C.: Sensitivity analysis of thermodynamic properties of liquid water: a general approach to improve empirical potentials. J. Phys. Chem. A 110, 762–771 (2006)
Jirsák, J., Nezbeda, I.: Molecular mechanisms underlying the thermodynamics properties of water. J. Mol. Liq. 134, 99–106 (2007)
Kuo, I.-F.W., Mundy, C.J., Eggimann, B.L., McGrath, M.J., Siepmann, J.I., Chen, B., Vieceli, J., Tobias, D.J.: Structure and dynamics of the aqueous liquid-vapor interface: a comprehensive particle-based simulation study. J. Phys. Chem. B 110, 3738–3746 (2006)
Lee, S.H., Rossky, P.J.: A comparison of the structure and dynamics of liquid water at hydrophobic and hydrophilic surfaces—a molecular dynamics simulation study. J. Chem. Phys. 100, 3334–3345 (1994)
Lee, C.Y., McCammon, J.A., Rossky, P.J.: The structure of liquid water at an extended hydrophobic surface. J. Chem. Phys. 80, 4448–4455 (1984)
Lum, K., Chandler, D., Weeks, J.D.: Hydrophobicity at small and large length scales. J. Phys. Chem. B 103, 4570–4577 (1999)
Luzar, A., Chandler, D.: Effect of environment on hydrogen bond dynamics in liquid water. Phys. Rev. Lett. 76, 928–931 (1996)
Marcotte, E., Stillinger, F.H., Torquato, S.: Optimized monotonic convex pair potentials stabilize low-coordinated crystals. Soft Matter 7, 2332–2335 (2011)
Remsing, R.C., Rodgers, J.M., Weeks, J.D.: Unpublished
Rodgers, J.M., Weeks, J.D.: Interplay of local hydrogen-bonding and long-ranged dipolar forces in simulations of confined water. Proc. Natl. Acad. Sci. USA 105, 19136 (2008)
Rodgers, J.M., Weeks, J.D.: Local molecular field theory for the treatment of electrostatics. J. Phys., Condens. Matter 20, 494206 (2008)
Rodgers, J.M., Weeks, J.D.: Accurate thermodynamics for short-ranged truncations of Coulomb interactions in site-site molecular models. J. Chem. Phys. 131, 244108 (2009)
Rodgers, J.M., Hu, Z., Weeks, J.D.: On the efficient and accurate short-ranged simulations of uniform polar molecular liquids. Mol. Phys. 109, 1195–1211 (2011)
Schmidt, J., VandeVondele, J., Kuo, I.-F.W., Sebastiani, D., Siepmann, J.I., Hutter, J., Mundy, C.J.: Isobaric-isothermal molecular dynamics simulations utilizing density functional theory: an assessment of the structure and density of water at near-ambient conditions. J. Phys. Chem. B 113, 11959–11964 (2009)
Shah, J.K., Asthagiri, D., Pratt, L.R., Paulaitis, M.E.: Balancing local order and long-ranged interactions in the molecular theory of liquid water. J. Chem. Phys. 127, 144508 (2007)
Smith, W., Yong, C., Rodger, P.: DL_POLY: application to molecular simulation. Mol. Simul. 28, 385–471 (2002)
Stillinger, F.H.: Structure in aqueous solutions of nonpolar solutes from the standpoint of scaled-particle theory. J. Solution Chem. 2, 141–158 (1973)
Varilly, P., Patel, A.J., Chandler, D.: An improved coarse-grained model of solvation and the hydrophobic effect. J. Chem. Phys. 134, 074109 (2011)
Vega, C., Abascal, J.L.F., Conde, M.M., Aragones, J.L.: What ice can teach us about water interactions: a critical comparison of the performance of different water models. Faraday Discuss. 141, 251–276 (2009)
Wang, J., Román-Pérez, G., Soler, J.M., Artacho, E., Fernández-Serra, M.-V.: Density, structure, and dynamics of water: the effect of van der Waals interactions. J. Chem. Phys. 134, 024516 (2011)
Weeks, J.D.: Connecting local structure to interface formation: a molecular scale van der Waals theory of nonuniform liquids. Annu. Rev. Phys. Chem. 53, 533–562 (2002)
Weeks, J.D., Chandler, D., Andersen, H.C.: Role of repulsive forces in determining the equilibrium structure of simple liquids. J. Chem. Phys. 54, 5237–5247 (1971)
Widom, B.: Intermolecular forces and the nature of the liquid state. Science 157, 375–382 (1967)
Xu, L., Buldyrev, S.V., Angell, C.A., Stanley, H.E.: Thermodynamics and dynamics of the two-scale spherically symmetric Jagla ramp model of anomalous liquids. Phys. Rev. E 74, 031108 (2006)
Yan, Z., Buldyrev, S.V., Giovambattista, N., Stanley, H.E.: Structural order for one-scale and two-scale potentials. Phys. Rev. Lett. 95, 130604 (2005)
Yeh, I.C., Berkowitz, M.L.: Ewald summation for systems with slab geometry. J. Chem. Phys. 111, 3155–3162 (1999)
Zhu, S.B., Wong, C.F.: Sensitivity analysis of water thermodynamics. J. Chem. Phys. 98, 8892–8899 (1993)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Remsing, R.C., Rodgers, J.M. & Weeks, J.D. Deconstructing Classical Water Models at Interfaces and in Bulk. J Stat Phys 145, 313–334 (2011). https://doi.org/10.1007/s10955-011-0299-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10955-011-0299-3