Abstract
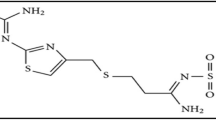
A UV–Vis spectrophotometric method was used to confirm the intermolecular charge-transfer complexes of the drug triamterene (TM) donor with four nitro π-acceptors (2,4,6-trinitrophenol (PA), 4-nitrophenol (4-NP), 4-nitroacetophenone (4-NAP) and m-dinirobenzene (m-DNB)) in methanol, ethanol, acetone, acetonitrile, chloroform, dichloromethane, and dichloroethane at 25 °C. The spectra showed chemical shifts upon the addition of TM to solutions of the nitro acceptors in the solvents. The appearance of new bands or hyper- and hypo-chromic effects confirmed the presence of charge-transfer complexes. The spectra of the complexes of TM and nitro π-acceptors show different absorption bands at different wavelengths for different solvents. The molar ratio method showed a result of 1:1 for the TM–PA system and 1:2 for the TM–4-NP, TM–4NAP, and TM–m-DNB charge-transfer complexes. The physical spectroscopic parameters were calculated, including the formation constant (KCT), molar extinction coefficient (εCT), oscillator strengths (f), ionization potential (Ip), resonance energy (RN), and Gibbs energy (ΔG°). The optical band gap of the TM-nitro charge-transfer complexes was estimated, and there was clearly a relationship between the different kinds of acceptors and their optical band gaps.
















Similar content being viewed by others
References
Khan, I.M., Shakya, S.: Exploring colorimetric real-time sensing behavior of a newly designed CT complex toward nitrobenzene and Co2+: spectrophotometric DFT/TD-DFT and mechanistic insights. ACS Omega 4, 9983–9995 (2019)
Shakya, S., Khan, I.M., Ahmad, M.: Charge transfer complex based real-time colorimetric chemosensor for rapid recognition of dinitrobenzene and discriminative detection of Fe2+ ions in aqueous media and human hemoglobin. J. Photochem. Photobiol. A: Chem. 392, 112402 (2020)
Khan, I.M., Shakya, S., Singh, N.: Preparation, single-crystal investigation and spectrophotometric studies of proton transfer complex of 2,6-diaminopyridine with oxalic acid in various polar solvents. J. Mol. Liq. 250, 150–161 (2018)
Khan, I.M., Islam, M., Shakya, S., Alam, K., Alam, N., Shahid, M.: Synthesis, characterization, antimicrobial and DNA binding properties of an organic charge transfer complex obtained from pyrazole and chloranilic acid. Bioorg. Chem. 99, 103779 (2020)
Khan, I.M., Alam, K., Alam, M.J., Ahmad, M.: Spectrophotometric and photocatalytic studies of H-bonded charge transfer complex of oxalic acid with imidazole: single crystal XRD, experimental and DFT/TD-DFT studies. New J. Chem. 43, 9039–9051 (2019)
Khan, I.M., Alam, K., Alam, M.J.: Exploring charge transfer dynamics and photocatalytic behavior of designed donor–acceptor complex: Characterization, spectrophotometric and theoretical studies (DFT/TD-DFT). J. Mol. Liq. 310, 113213 (2020)
Alam, K., Khan, I.M.: Crystallographic, dynamic and Hirshfeld surface studies of charge transfer complex of imidazole as a donor with 3,5-dinitrobenzoic acid as an acceptor: determination of various physical parameters. Org. Electron. 63, 7–22 (2018)
Singh, N., Khan, I.M., Ahmad, A., Javed, S.: Synthesis, spectrophotometric and thermodynamic studies of charge transfer complex of 5,6-dimethylbenzimidazole with chloranilic acid at various temperatures in acetonitrile and methanol solvents. J. Mol. Liq. 221, 1111–1120 (2016)
Khan, I.M., Ahmad, A.: Synthesis, spectrophotometric, structural and thermal studies of the charge transfer complex of p-phenylenediamine, as an electron donor with π acceptor 3,5-dinitrobenzoic acid. Spectrochim. Acta A 76, 315–321 (2010)
Singh, N., Khan, I.M., Ahmad, A., Javed, S.: Synthesis and dynamics of a novel proton transfer complex containing 3,5-dimethylpyrazole as a donor and 2,4-dinitro-1-naphthol as an acceptor: crystallographic, UV–visible spectrophotometric, molecular docking and Hirshfeld surface analyses. New J. Chem. 41, 6810–6821 (2017)
Vickers, M.D., Morgan, M., Spencer, P.S.J., Read, M.S.: Drugs in Anaesthetic and Intensive Care Practice, 8th edn., p. 415. Butterworth Heinemann, Oxford (1999)
Ibañez, G.A., Escandar, G.M., Mansilla, A.E., de la Peña, A.M.: Determination of triamterene in pharmaceutical formulations and of triamterene and its main metabolite hydroxytriamterene sulfate in urine using solid-phase and aqueous solution luminescence. Anal. Chim. Acta 538, 77–84 (2005)
Horstkötter, C., Kober, S., Spahn-Langguth, H., Mutschler, E., Blaschke, G.: Determination of triamterene and its main metabolite hydroxytriamterene sulfate in human urine by capillary electrophoresis using ultraviolet absorbance and laser-induced fluorescence detection. J. Chromatogr. B 769, 107–117 (2002)
Arvand, M., Mousavi, M.F., Zanjanchi, M.A., Shamsipur, M.: Direct determination of triamterene by potentiometry using a coated wire selective electrode. Pharm. Biomed. Anal. 33, 975–982 (2003)
Wagieh, N.E., Abbas, S.S., Abdelkawy, M., Abdelrahman, M.M.: Spectrophotometric and spectrodensitometric determination of triamterene and xipamide in pure form and in pharmaceutical formulation. Drug Test. Anal. 2, 113–121 (2010)
Hudari, F.F., da Silva, B.F., Pividori, M.I., Zanoni, M.V.B.: Voltammetric sensor based on magnetic particles modified composite electrode for determination of triamterene in biological sample. Solid State Electrochem. 20, 2491–2501 (2015)
Hemavathi, G., Hipparagi, S.M.: Sensitive LC-MS/MS method for the simultaneous determination of bisoprolol and triamterene in human plasma. Asian J. Pharmaceut. Clin. Res. 10(4), 477–484 (2017)
Maślanka, A., Krzek, J., Stolarczyk, M.: simultaneous analysis of hydrochlorothiazide, triamterene, furosemide, and spironolactone by densitometric TLC. J. Planar Chromatogr. Modern TLC 22, 405–410 (2009)
Hudari, F.F., Souza, J.C., Zanoni, M.V.B.: Adsorptive stripping voltammetry for simultaneous determination of hydrochlorothiazide and triamterene in hemodialysis samples using a multi-walled carbon nanotube-modified glassy carbon electrode. Talanta 179, 652–657 (2018)
Chen, X., Teng, W., Miao, L., Fang, J., Liang, Y.: Simultaneous analysis of hydrochlorothiazide, triamterene and reserpine in rat plasma by HPLC and DSPE. Chromatographia 79, 451–456 (2016)
Pandeeswaran, M., Elango, K.P.: Electronic, Raman and FT-IR spectral investigations of the charge transfer interactions between ketoconazole and povidone drugs with iodine. Spectrochim. Acta A 72, 789–795 (2009)
Adam, A.M.A., Refat, M.S., Saad, H.A., Hegab, M.S.: Charge transfer complexation of the anticholinergic drug clidinium bromide and picric acid in different polar solvents: Solvent effect on the spectroscopic and structural morphology properties of the product. J. Mol. Liq. 216, 192–208 (2016)
Elqudaby, H.M., Mohamed, G.G., El-Din, G.M.G.: Analytical studies on the charge transfer complexes of loperamide hydrochloride and trimebutine drugs. Spectroscopic and thermal characterization of CT complexes. Spectrochim. Acta A 129, 84–95 (2014)
Mohamed, M.E., Frag, E.Y.Z., Hathoot, A.A., Shalaby, E.A.: Spectrophotometric determination of fenoprofen calcium drug in pure and pharmaceutical preparations. Spectroscopic characterization of the charge transfer solid complexes. Spectrochim. Acta A 189, 357–365 (2018)
El-Habeeb, A.A., Refat, M.S.: Charge transfer complexes beneficial method for determination of fluoxetine HCl pure drug: spectroscopic and thermal analyses discussions. Russ. J. Gen. Chem. 84, 1810–1818 (2014)
El-Habeeb, A.A., Refat, M.S.: Spectroscopic studies on the utility of charge transfer interactions for determination of phenytoin sodium. Russ. J. Gen. Chem. 85, 939–950 (2015)
El-Habeeb, A.A., Refat, M.S.: Synthesis and spectroscopic characterization of trazodone charge transfer complexes with different types of π-acceptors. Russ. J. Gen. Chem. 85, 951–958 (2015)
Adam, A.M.A., Refat, M.S.: Chemistry of drug interactions: characterization of charge-transfer complexes of Guaifenesin with various acceptors using spectroscopic and thermal methods. Russ. J. Gen. Chem. 84, 1847–1856 (2014)
Briegleb, G.: Elektronen affinitäten organischer Moleküle. Angew. Chem. 76, 326–341 (1964)
Hasani, M., Mafakheri, N.A.: A Spectrophotometric and thermodynamic study of the charge-transfer complexes of iodine with Nortriptyline and Imipramine drugs in chloroform and dichloromethane solutions. Russ. J. Phys. Chem. B 10, 929–934 (2016)
Skoog, D.A.: Principle of International Analysis, 3rd edn. Sannder College Publishing, New York (1985)
Tsubomura, H., Lang, R.P.: Molecular complexes and their spectra. XIII. Complexes of iodine with amides, diethyl sulfide and diethyl disulfide. J. Am. Chem. Soc. 83, 2085–2092 (1961)
Aloisi, G.G., Pignataro, S.: Molecular complexes of substituted thiophens with σ and π acceptors. Charge transfer spectra and ionization potentials of the donors. J. Chem. Soc. Faraday Trans. 69, 534–539 (1973)
Lever, A.B.P.: Inorganic Electronic Spectroscopy, 2nd edn., p. 161. Elsevier, Amsterdam (1985)
Rathore, R., Lindeman, S.V., Kochi, J.K.: Charge-transfer probes for molecular recognition via steric hindrance in donor–acceptor pairs. J. Am. Chem. Soc. 119, 9393–9404 (1997)
Briegleb, G., Czekalla, J.: Intensity of electron transition bands in electron donator–acceptor complexes. Z. Phys. Chem. (Frankfurt) 24, 37–54 (1960)
Martin, A.N., Swarbrick, J., Cammarata, A.: Physical Pharmacy, 3rd edn., p. 344. Philadelphia, PA, Lee and Febiger (1969)
Mott, N.F., Davis, E.A.: Electrochemical Process in Non-crystalline Materials. Calendron Press, Oxford (1979)
Acknowledgement
This research was funded by the Deanship of Scientific Research at Princess Nourah Bint Abdulrahman University, through the Pioneer Researcher Funding Program (Grant No. PR-1440-3).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
El-Habeeb, A.A., Al-Balawi, A., Al-Motiri, A. et al. In Situ Spectrophotometric Investigation of Charge Transfer Complexes Between Triamterene, a Management of Hypertension Drug, and Four Kinds of Nitro Acceptors in Different Organic Solvents. J Solution Chem 50, 277–289 (2021). https://doi.org/10.1007/s10953-021-01060-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10953-021-01060-z