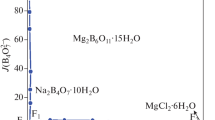Abstract
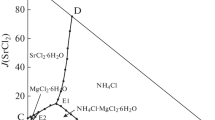
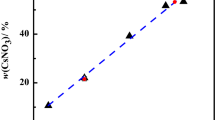
The solid–liquid phase equilibrium of the quaternary system (CaCl2 + CaSO4 + CaB6O10 + H2O) at 288.15 K and atmospheric pressure were investigated with the method of isothermal dissolution equilibrium. The dry-salt, water-phase diagrams and physicochemical properties including refractive index, density and pH versus composition diagrams were plotted on the basis of the experimental data. In the dry-salt diagram of the quaternary system, there is one invariant point, three solubility curves and three crystallization regions corresponding to single salts antarcticite (CaCl2·6H2O, Ant), gypsum (CaSO4·2H2O, Gy) and gowerite (CaB6O10·5H2O, Gow). No double salt or solid solution was formed. The area of crystallization region of gypsum is the largest, and that of antarcticite is the smallest. In addition, the physicochemical properties showed a regular change with the Jänecke index value of J(CaCl2).





Similar content being viewed by others
References
Ishii, T., Kokaku, H., Nagai, A., Nishita, T., Kakimoto, M.: Calcium borate flame retardation system for epoxy molding compounds. Polym. Eng. Sci. 46, 799–806 (2006)
Han, X., Day, D.E.: Reaction of sodium calcium borate glasses to form hydroxyapatite. J. Mater. Sci. Mater. Med. 18, 1837–1847 (2007)
Li, J.S., Li, W.T., Li, H.Z., Peng, X.M.: Investigation of the chemical characteristics and its geological significance of the tertiary oil-field brine in the western Qaidam basin. Hydrogeol. Eng. Geol. 40, 28–36 (2013)
Li, Q.H., Gu, T.X., Yu, S.S., Yao, Y., Li, B., Li, L.: Study on the precipitation pathway of Nanyishan oilfield brine at subzero temperatures. Acta Phys. Chim. Sin. 27, 1803–1808 (2011)
Fu, J.L., Yu, S.S., Li, S.J., Ren, H.Y.: Availability of tertiary oilfield water resources in western Qaidam basin. J. Salt Lake Res. 13, 17–21 (2005)
Deng, T.L., Zhou, H., Chen, X.: Phase Diagrams of Salt–Water Systems and applications. Chemical Industry Press, Beijing (2013)
Databases of Salt–water Systems on Ourway Co., Ltd. http://www.ourwaystd.com, (2016)
Lei, L.Y., Li, L., Cao, L.N., Guo, Y.F., Zhang, S.S., Wang, S.Q., Deng, T.L.: Solid–liquid phase equilibria of the aqueous ternary system (CaCl2 + CaB6O10 + H2O) at 308.15, 323.15 K and 101.325 kPa. J. Chem. Eng. Jpn. 50, 231 (2017)
Wang, S.Q., Guo, Y.F., Liu, W.J., Deng, T.L.: Phase equilibria in the aqueous ternary system (LiBO2 + CaB2O4 + H2O) at 288.15 and 298.15 K. J. Solution Chem. 44, 1545–1554 (2015)
Deng, T.L., Li, D.: Solid–liquid metastable equilibria in the quaternary system (NaCl + LiCl + CaCl2 + H2O) at 288.15 K. J. Chem. Eng. Data 53, 2488–2492 (2008)
Wang, M.X., Lei, L.Y., Guo, Y.F., Meng, L.Z., Wang, S.Q., Deng, T.L.: Phase equilibria of the reciprocal quaternary system (Na+, Ca2+//Cl–, borate–H2O) at 288.15 K and 101.325 kPa. J. Chem. Eng. Data 63, 2931–2939 (2018)
Institute of Qinghai Salt-Lake of Chinese Academy of Sciences: Analytical Methods of Brines and Salts, 2nd edn. Chinese Science Press, Beijing (1984)
Acknowledgements
Financial support from the Major Scientific and Technological Project of the Tibet Autonomous Region (XZ201801-GB-01), and the Yangtze Scholars and Innovative Research Team of the Chinese University (IRT_17R81).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, M., Shi, M., Zhou, Y. et al. Solid–Liquid Phase Equilibrium of the Quaternary System (CaCl2 + CaSO4 + CaB6O10 + H2O) at 288.15 K and Atmospheric Pressure. J Solution Chem 49, 1328–1338 (2020). https://doi.org/10.1007/s10953-020-01037-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10953-020-01037-4