Abstract
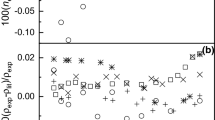
Densities of dilute solutions of water in acetone, with solute mole fractions ranging up to 0.03, have been measured with an error of 8 ×10−6 g⋅cm−3, at 288.15, 298.15, 308.15 and 318.15 K, using a precision vibrating-tube densimeter. The partial molar volumes of the solute water (down to infinite dilution) and solvent acetone, as well as the excess molar volumes of the specified mixtures, have been calculated. The effects of the solute concentration and temperature on the volume packing changes, caused by solvation of water in acetone, have been considered.
Similar content being viewed by others
References
Abrosimov, V.K., Ivanov, E.V.: Water in nonaqueous solutions: State and solvation. In: Water Kutepov, A.M. (ed.) Structure, State and Solvation, Achievements of Last Years, pp. 277–346. Nauka, Moscow (2003). (in Russian)
Sirotkin, V.A., Solomonov, B.N., Faizullin, D.A., Fedotov, V.D.: IR spectroscopic study of the state of water in dioxane and acetonitrile: Relationship with the thermodynamic activity of water at 278–318 K. J. Struct. Chem. 41, 997–1003 (2000)
Graziano, G.: Solvation of a water molecule in cyclohexane and water. Can. J. Chem. 79, 105–109 (2001)
Scharlin, P., Steinby, K., Domańska, U.: Volumetric properties of binary mixtures of N,N-dimethylformamide with water and water-d2 at temperatures from 277.13 K to 318.15 K. J. Chem. Thermodyn. 34, 927–957 (2002)
Ivanov, E.V., Abrosimov, V.K.: Lebedeva, E. Yu.: Isotope effect on fractional dilatability of solute water as an indicator of the H-bonding ability of an aprotic dipolar solvent. J. Struct. Chem. 45, 974–980 (2004)
Burger, K.: Solvation, Ionic, and Complex Formation Reactions in Non-aqueous Solvents: Experimental Methods for Their Investigation. Elsevier, Amsterdam (1983)
Mamantov, G., Popov, A.I. (eds.): Chemistry of Nonaqueous Solutions: Current Progress. VCH, New York (1994)
Belousov, V.P., Panov, M.Y.: Thermodynamics of Aqueous Solutions of Non-electrolytes. Khimiya, Leningrad (1983). (in Russian)
Venables, D.S., Schmuttenmaer, C.: Spectroscopy and dynamics of mixtures of water with acetone, acetonitrile, and methanol. J. Chem. Phys. 113, 11222–11236 (1122) (2000)
Max, J.-J., Chapados, C.: Infrared spectroscopy of acetone − water liquid mixtures. I. Factor analysis. J. Chem. Phys. 119, 5632–5643 (2003)
Perera, A., Sokolić, F.: Modeling nonionic aqueous solutions: The acetone − water mixture. J. Chem. Phys. 121, 11272–11282 (1127) (2004)
Baratoux, Y., Juillard, J.: Density of anhydrous acetone and estimation of water content of acetone from density measurements. Talanta 27, 427–428 (1980)
Dickens, B., Dickens, S.H.: Estimation of concentration and bonding environment of water dissolved in common solvents using near infrared absorptivity. J. Res. Natl. Inst. Stand. Technol. 104, 173–183 (1999)
Marcus, Y.: Ion Solvation, pp. 133–138. Wiley, New York (1985)
Yerlett, T.K., Wormald, C.J.: The enthalpy of acetone. J. Chem. Thermodyn. 18, 371–379 (1986)
Krestov, G.A., Afanas’ev, V.A., Efremova, L.S.: Physical and Chemical Properties of Binary Solvents, p. 155. Khimiya, Leningrad (1988). (in Russian)
Tasić, A.Ž., Grozdanić, D.K., Djordjević, B.D., Šerbanović, S.P., Radojković, N.: Refractive indices and densities of the system acetone + benzene + cyclohexane at 298.15 K. Changes of refractivity and of volume on mixing. J. Chem. Eng. Data 40, 586–588 (1995)
Thomas, K.T., McAllister, R.A.: Densities of liquid acetone − water solutions up to their normal boiling points. AIChE J. 3, 161–164 (1957)
Abrosimov, V.K., Korolev, V.V.: Densimetry. In: Kutepov, A.M. (ed.) Experimental Methods of Solution Chemistry: Densimetry, Viscosimetry, Conductometry and Others Methods, pp. 5–45. Nauka, Moscow (1997). (in Russian)
Ivanov, E.V., Abrosimov, V.K.: D2O−H2O Solvent isotope effects on the apparent molar volumes of tetramethyl-bis-urea (Mebicarum) solutions. J. Solution Chem. 36, 313–325 (2007)
Korolev, V.V.: Precise vibrating-tube densimeter for measurement of liquid and gas densities. Russ. J. Phys. Chem. 63, 1701–1704 (1989)
Kell, G.S.: Effect of isotope composition, temperature, pressure, and dissolved gases on the density of liquid water. J. Phys. Chem. Ref. Data 6, 1109–1131 (1977)
Ivanov, E.V.: Interrelation between the enthalpy and volume effects of solution of liquid nonelectrolytes. Russ. J. Phys. Chem. 78, 1225–1229 (2004)
Klofutar, C., Nemec, T.: Apparent molar volumes and expansibilities of 1-octanol, 1-nonanol, and 1-decanol in dilute cyclohexane solutions. J. Solution Chem. 25, 1151–1162 (1996)
Lepori, L., Gianni, P.: Partial molar volumes of ionic and nonionic organic solvents in water: A simple additivity scheme based on the intrinsic volume approach. J. Solution Chem. 29, 405–447 (2000)
Abrosimov, V.K.: Non-extrapolating method of determination of partial molar volumes and compressibilities at infinite dilution. Russ. J. Phys. Chem. 62, 1913–1916 (1988)
Daniels, F., Mathews, J.H., Williams, J.W., Bender, P., Murphy, G.W., Alberty, R.A.: Experimental Physical Chemistry, 4th edn. McGraw-Hill, New York (1949)
Sakurai, M.: Partial molar volumes for acetonitrile + water. J. Chem. Eng. Data 37, 358–362 (1992)
Sakurai, M.: Partial molar volumes for 1,4-dioxane + water. J. Chem. Eng. Data 37, 492–496 (1992)
Jedlovszky, P., Pálinkás, G.: Monte Carlo simulation of liquid acetone with a polarizable molecular model. Mol. Phys. 84, 217–233 (1995)
Bushuev, Yu.G., Davletbaeva, S.V.: Structural properties of liquid acetone. Russ. Chem. Bull. 48, 25–34 (1999)
Korolev, V.P., Batov, D.V., Krestov, G.A.: Enthalpic characteristics of water, methanol, and ethanol in solutions. Russ. J. Gen. Chem. 61, 1921–1927 (1991)
Bushuev, Y.G., Korolev, V.P.: Structure-concentration changes in binary solutions on the data of computer simulation. In: Kutepov, A.M. (ed.) Concentrated and Saturated Solutions, pp. 255–313. Nauka, Moscow (2002). (in Russian)
Chen, D.H.C., Chu, P.M., Tanaka, S.H., To, E.C.H., Koga, Y.: Excess chemical potentials, partial molar enthalpies and entropies in binary aqueous acetone and tetramethyl urea at 25 °C. Fluid Phase Equil. 175, 35–43 (2000)
de Visser, C., Heuvelsland, W.J.M., Dunn, L.A., Somsen, G.: Some properties of binary aqueous liquid mixtures. Apparent molar volumes and heat capacities at 298.15 K over the whole mole fraction range. J. Chem. Soc. Faraday Trans. 1 74, 1159–1169 (1978)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ivanov, E.V., Abrosimov, V.K. & Lebedeva, E.Y. Volumetric Properties of Dilute Solutions of Water in Acetone between 288.15 and 318.15 K. J Solution Chem 37, 1261–1270 (2008). https://doi.org/10.1007/s10953-008-9301-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10953-008-9301-3