Abstract
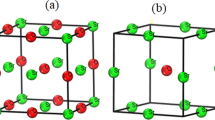
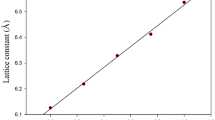
The all-electron approach implemented in the CRYSTAL06 program is used along with a pseudopotential method in the pseudo-atomic orbital basis set to study the crystal structure, elastic constants and bulk moduli, the band structure and density of states for the family of silver nitrides. Calculations are performed within density functional theory with the use of local and gradient functionals to describe exchange and correlation. For the general type of the cubic lattice, all considered compounds can be put in the following order of their relative stability: AgN (rock salt structure), AgN2 (fluorite structure), Ag2N (cuprite structure), and Ag3N (anti-ReO2). It is shown that AgN, AgN2, and Ag2N are metals, whereas Ag3N is a semiconductor with a band gap of 0.25 eV. Chemical bonding in these compounds has ionic and covalent components, apart from the metal one.
Similar content being viewed by others
References
M. B. Kanoun and S. Goumri-Said, Phys. Lett. A, 362, 73–83 (2007).
M. B. Kanoun and S. Goumri-Said, Phys. Rev. B, 72, No. 113103 (2005).
R. Yu and X. F. Zhang, ibid., 72, No. 054103 (2002).
U. Hahn and W. Weber,ibid., 53, 12684–12694 (1996).
E. S. Shanley and J. L. Ennis, Industrial and Engineering Chem. Res., 30, 2503–2506 (1991).
R. De Paiva, R. A. Nogueiza, and J. L. A. Alves, Phys. Rev. B, 75, No. 085105 (2007).
E. Deligoz, K. Colakoglu, and Y. O. Ciftcl, Chin. Phys. Lett., 25, No. 6, 2154–2157 (2008).
R. Dovesi, V. R. Saunders, C. Roetti, et al., CRYSTAL 06 Users’s Manual, Univ. Torino, Torino (2006).
J. Slater, Self-Consistent Field for Molecules and Solids, McGraw-Hill ( 1974).
J. P. Perdew and A. Zunger, Phys. Rev. B, 23, 5048–5079 (1981).
J. P. Perdew and Y. Wang, ibid., 33, 8800–8802 (1986).
J. P. Perdew and Y. Wang, ibid., 45, 13244–13248 (1992).
Internet resource: www.crystal.initio.it/Basic_Set/ptable.html.
C. Gatti, V. R. Saunders, and C. Roetti, J. Chem. Phys., 101, 10686–10696 (1994).
R. W. Jansen and O. F. Sankey, Phys. Rev. B, 35, 6520–6531 (1987).
A. B. Gordienko and A. S. Poplavnoi, Izv. Vyssh. Uchebn. Zaved., Fizika, No. 1, 1–8 (1997).
G. B. Bachelet, D. R. Hamann, and M. Schlüter, Phys. Rev. B, 26, 4199–4228 (1982).
T. Tsuchiya and K. Kawamura, J. Chem. Phys., 114, No. 22, 10086–10093 (2001).
O. Beckstein, J. E. Klepeis, G. L. W. Hart, and O. Pankratov, Phys. Rev. B, 63, No. 134112 (2001).
F. D. Murnaghan, Proc. Nat. Acad. Sci. USA, 50, 244–247 (1944).
Z. Wu, E. Zhao, H. Xiang, et al., Phys. Rev. B, 76, No. 054115 (2007).
W. Voigt, Lehrbuch der Kristallphysik, Teubner, Leipzig (1928).
A. Z. Reuss, Angew. Math. Mech., 49, 49–58 (1929).
R. Hill, Proc. Phys. Soc. (London), A65, 349–354 (1952).
F. Gao, Phys. Rev. B, 73, No. 132104 (2006).
R. Yang, T. Zhang, P. Jiang, and Y. Bai, Appl. Phys. Lett., 92, No. 231906 (2008).
P. Ravidran, L. Fast, P. A. Korzhavyi, and B. Johansson, J. Appl. Phys., 84, No. 9, 4891–4903 (1998).
H. J. Frost and M. F. Ashby, Deformation-Mechanism Maps, Pergamon, Oxford (1982).
V. L. Moruzzi, J. F. Janak, and K. Schwarz, Phys. Rev. B, 37, 790–799 (1988).
A. B. Gordienko, Yu. N. Zhuravlev, and D. G. Fedorov, Fiz. Tverd. Tela, 49, No. 2, 216–220 (2007).
A. B. Gordienko and S. A. Poplavnoi, Phys. Stat. Sol.(b), 204, 407–411 (1998).
J. E. Klepeis, O. Beckstein, O. Pankratov, and G. L. W. Hart, Phys. Rev. B, 64, No. 155110 (2001).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Zhurnal Strukturnoi Khimii, Vol. 51, No. 3, pp. 425-432, May–June, 2010.
Original Russian Text Copyright © 2010 by A. B. Gordienko and Yu. N. Zhuravlev
Rights and permissions
About this article
Cite this article
Gordienko, A.B., Zhuravlev, Y.N. Ab initio calculations of structural, elastic, and electronic properties of silver nitrides. J Struct Chem 51, 401–408 (2010). https://doi.org/10.1007/s10947-010-0061-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10947-010-0061-8