Abstract
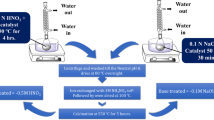
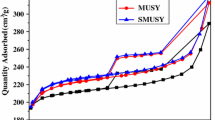
Mordenite was synthesized at 170 ºC under stirring (60 rpm) with different contents of L-glutamic acid monosodium salt (MSG) between 4 and 48 h. The materials were characterized by XRD, EDX, NH3-TPD, TG/DTG, SEM and N2 physisorption. The catalytic activity of zeolites was evaluated in the n-hexane cracking reaction at 550 ºC for 180 min. Mordenite was obtained with high relative crystallinity (83 ˗ 101%). The insertion of the amino acid salt reduced the acidity of the zeolites from 0.788 to 0.612 mmol g-1. Thermal analyses indicated the complete removal of MSG from the zeolite structure after washing, eliminating the calcination process and consequently, reducing costs. The micrographs revealed crystals of < 10 μm. N2 physisorption isotherms showed an increase in microporosity (SMicro from 411 to 445 m2 g-1 and VMicro from 0.165 to 0.178 cm3 g-1) and a higher degree of mesoporosity (VMeso from 0.009 to 0.012 cm3 g-1) for the samples with the lowest contents of MSG, indicating an optimal range for the addition of amino acid salt. The results of the n-hexane cracking reaction over mordenite samples revealed a higher tendency for the production of light olefins. The Mor/6Glut sample demonstrated high activity and a slower deactivation rate than conventional mordenite, due to the synergy of its lower acidity and mesoporosity.






Similar content being viewed by others
References
J. Yao, X. Feng, J. Fan, Y. He, R. Kosol, Y. Zeng, G. Liu, Q. Ma, G. Yang, N. Tsubaki, Effects of mordenite zeolite catalyst synthesis conditions on dimethyl ether carbonylation. Microporous Mesoporous Mater. (2020). https://doi.org/10.1016/j.micromeso.2020.110431
Y. Zhou, J. Zhang, L. Wang, X. Cui, X. Liu, S.S. Wong, H. An, N. Yan, J. Xie, C. Yu, P. Zhang, Y. Du, S. Xi, L. Zheng, X. Cao, Y. Wu, Y. Wang, C. Wang, H. Wen, L. Chen, H. Xing, J. Wang, Self-assembled iron-containing mordenite monolith for carbon dioxide sieving. Science. (2021). https://doi.org/10.1126/science.aax5776
X. Ren, S. Yang, R. Xu, M. Guo, W. Huang, M. Ding, J. Zhong, Mono- and di-valent ion exchange of mordenite membranes for dehydration of acetic acid by pervaporation. J. Membr. Sci. (2022). https://doi.org/10.1016/j.memsci.2021.119998
M. Moshoeshoe, M.S. Nadiye-Tabbiruka, V. Obuseng, A review of the Chemistry, structure, Properties and Applications of Zeolites. Am. J. Mater. Sci. (2017). https://doi.org/10.5923/j.materials.20170705.12
A. Kostyniuk, D. Bajec, B. Likozar, Catalytic hydrocracking reactions of tetralin as aromatic biomass tar model compound to benzene/toluene/xylenes (BTX) over zeolites under ambient pressure conditions. J. Ind. Eng. Chem. (2021). https://doi.org/10.1016/j.jiec.2021.01.010
J.G. Pacheco Filho, M. Schmal, J.L. Monteiro, A study on toluene disproportionation over mordenite. Catal. Today. 5, 4 (1989). https://doi.org/10.1016/0920-5861(89)80014-8
S.V. Konnov, I.I. Ivanova, O. Ponomareva, V.I. Zaikovskii, Hydroisomerization of n-alkanes over Pt-modified micro/mesoporous materials obtained by mordenite recrystallization. Microporous Mesoporous Mater. 164, 222–231 (2012). https://doi.org/10.1016/j.micromeso.2012.08.017
S. Li, H. Wu, R.C.J. van de Poll, R.R.M. Joosten, N. Kosinov, E.J.M. Hensen, Synthesis of Nanocrystalline Mordenite Zeolite with Improved Performance in Benzene Alkylation and n-Paraffins hydroconversion. ChemCatChem. 14, 9 (2022). https://doi.org/10.1002/cctc.202101852
O.V. Shvets, K.M. Konysheva, M.V. Shamzhy, M.V. Opanasenko, P.S. Yaremov, C. Xiao, X. Zou, J. Čejka, Mordenite nanorods and nanosheets prepared in presence of gemini type surfactants. Catal. Today. 324, 115–122 (2019). https://doi.org/10.1016/j.cattod.2018.10.043
M.F. Menoufy, A.E. Nadia, H.S. Ahmed, Catalytic Dewaxing for Lube Oil production. Pet. Sci. Technol. 27, 6 (2009). https://doi.org/10.1080/10916460802104172
K. Segawa, H. Tachibana, Highly selective methylamine synthesis over modified mordenite catalysts. J. Catal. 131, 2 (1991). https://doi.org/10.1016/0021-9517(91)90280-H
F.R. Best, A. Mundstock, P.A. Kißling, H. Richter, K.D.J. Hindricks, A. Huang, P. Behrens, J. Caro, Boosting dimethylamine formation selectivity in a membrane Reactor by in situ water removal. Ind. amp; Eng. Chem. Res. 61, 1 (2021). https://doi.org/10.1021/acs.iecr.1c04149
D. Kwon, M. Numan, J. Kim, M. Yilmaz, S.-E. Park, H. Ihee, C. Jo, Tailoring the CO2 selective adsorption properties of MOR zeolites by post functionalization. J. CO2 Util. 62 (2022). https://doi.org/10.1016/j.jcou.2022.102064
A.A. Alomair, Y. Alqaheem, Optimization of Mordenite membranes using sucrose precursor for pervaporation of water-ethanol mixtures. Membranes. 11, 3 (2021). https://doi.org/10.3390/membranes11030160
Y. Yang, J. Ding, C. Xu, W. Zhu, P. Wu, An insight into crystal morphology-dependent catalytic properties of MOR-type titanosilicate in liquid-phase selective oxidation. J. Catal. 325, 101–110 (2015). https://doi.org/10.1016/j.jcat.2015.03.001
R.C. Lima, C.W. Lopes, J. Villarroel-Rocha, L. Bieseki, K. Sapag, S.B.C. Pergher, Organic-Free Synthesis of Finned Mordenite Zeolite. Nanomaterials. 12, 15 (2022). https://doi.org/10.3390/nano12152623
L. Zhang, S. Xie, W. Xin, X. Li, S. Liu, L. Xu, Crystallization and morphology of mordenite zeolite influenced by various parameters in organic-free synthesis. Mater. Res. Bull. 46, 6 (2011). https://doi.org/10.1016/j.materresbull.2011.02.018
Q. Lang, G. Fu, H. Zhao, J. Wang, X. Yang, V. Valtchev, Biomineralization at the Molecular Level: amino acid-assisted crystallization of Zeotype AlPO4•1.5H2O–H3. Cryst. Growth Des. 21, 12 (2021). https://doi.org/10.1021/acs.cgd.1c01160
R.F. Lobo, S.I. Zones, M.E. Davis, Structure-direction in Zeolite Synthesis. J. Incl. Phenom. Macrocycl. Chem. 21, 1 (1995). https://doi.org/10.1007/BF00709411
M.A. Klunk, S.B. Schröpfer, S. Dasgupta, M. Das, N.R. Caetano, A.N. Impiombato, P.R. Wander, C.A.M. Moraes, Synthesis and characterization of mordenite zeolite from metakaolin and rice husk ash as a source of aluminium and silicon. Chem. Pap. 74, 2481–2489 (2020). https://doi.org/10.1007/s11696-020-01095-4
L. Bai, Z. Xiong, E. Zhan, S. Li, W. Shen, Piperazine as a versatile organic structure-directing agent for zeolite synthesis: effect of SiO2/Al2O3 ratio on phase selectivity. J. Mater. Sci. 54, 7589–7602 (2019). https://doi.org/10.1007/s10853-019-03433-8
E.A. Abdelrahman, R.M. Hegazey, A. Alharbi, Facile synthesis of Mordenite nanoparticles for efficient removal of pb(II) ions from aqueous media. J. Inorg. Organomet. Polym. Mater. 30, 1369–1383 (2019). https://doi.org/10.1007/s10904-019-01238-5
U. Khalil, O. Muraza, Microwave-assisted hydrothermal synthesis of mordenite zeolite: optimization of synthesis parameters. Microporous Mesoporous Mater. 232, 211–217 (2016). https://doi.org/10.1016/j.micromeso.2016.06.016
B.K. Singh, Y. Kim, S.B. Baek, A. Meena, S. Sultan, J.H. Kwak, K.S. Kim, Template free facile synthesis of mesoporous mordenite for bulky molecular catalytic reactions. J. Ind. Eng. Chem. 57, 363–369 (2018). https://doi.org/10.1016/j.jiec.2017.08.044
S. Wang, B. He, R. Tian, C. Sun, R. Dai, X. Li, X. Wu, X. An, X. Xie, Synthesis and catalytic performance of hierarchically structured MOR zeolites by a dual-functional templating approach. J. Colloid Interface Sci. 527, 339–345 (2018). https://doi.org/10.1016/j.jcis.2018.05.053
A. Bolshakov, D.E.R. Hidalgo, A.J.F. van Hoof, N. Kosinov, E.J.M. Hensen, Mordenite Nanorods prepared by an Inexpensive pyrrolidine-based Mesoporogen for Alkane Hydroisomerization. ChemCatChem. 11, 12 (2019). https://doi.org/10.1002/cctc.201900298
A. Kornas, J.E. Olszówka, M. Urbanova, K. Mlekodaj, L. Brabec, J. Rathousky, J. Dedecek, V. Pashkova, Milling Activation for the Solvent-Free Synthesis of the Zeolite Mordenite. EurJIC 2020, 29 (2020). https://doi.org/10.1002/ejic.202000320
A. Kornas, J.E. Olszówka, M. Urbanova, L. Brabec, J. Rathousky, J. Dedecek, V. Pashkova, Ultrasonic pretreatment as a Tool for the Preparation of low-defect Zeolite Mordenite. ACS Omega. 6, 3 (2021). https://doi.org/10.1021/acsomega.0c05655
S.K. Rajabi, Sh. Sohrabnezhad, synthesis and characterization of magnetic core with two shells: Mordenite zeolite and CuO to form Fe3O4@MOR@CuO core-shell: as a visible light driven photocatalyst. Microporous Mesoporous Mater. 242, 136–143 (2016). https://doi.org/10.1016/j.micromeso.2017.01.024
S.K. Rajabi, Sh. Sohrabnezhad, Fabrication and characteristic of Fe3O4@MOR@CuO core-shell for investigation antibacterial properties. J. Fluor. Chem. 206, 36–42 (2018). https://doi.org/10.1016/j.jfluchem.2017.12.010
B.B. Munavalli, M.Y. Kariduraganavar, Development of novel sulfonic acid functionalized zeolites incorporated composite proton exchange membranes for fuel cell application. Electrochim. Acta. 296, 294–307 (2019). https://doi.org/10.1016/j.electacta.2018.11.056
B. Velaga, R.P. Parde, J. Soni, N.R. Peela, Synthesized hierarchical mordenite zeolites for the biomass conversion to levulinic acid and the mechanistic insights into humins formation. Microporous Mesoporous Mater. 287, 18–28 (2019). https://doi.org/10.1016/j.micromeso.2019.05.049
S. Sakthinathan, P. Tamizhdurai, A. Ramesh, T.-W. Chiu, V.L. Mangesh, S. Veerarajan, K. Shanthi, Platinum incorporated mordenite zeolite modified glassy carbon electrode used for selective electrochemical detection of mercury ions. Microporous Mesoporous Mater. 292, 109770 (2020). https://doi.org/10.1016/j.micromeso.2019.109770
Y. Wu, M. Zhou, B. Zhang, B. Wu, J. Li, J. Qiao, X. Guan, F. Li, Amino acid assisted templating synthesis of hierarchical zeolitic imidazolate framework-8 for efficient arsenate removal. Nanoscale. 6, 2 (2014). https://doi.org/10.1039/C3NR04390H
J. Zhang, S. Bai, Z. Chen, Y. Wang, L. Dong, H. Zheng, F. Caib, M. Hong, Core–shell zeolite Y with ant-nest like hollow interior constructed by amino acids and enhanced catalytic activity. J. Mater. Chem. A 5, 39 (2017). https://doi.org/10.1039/C7TA05048H
Q. Zhang, A. Mayoral, O. Terasaki, Q. Zhang, B. Ma, C. Zhao, G. Yang, J. Yu, Amino acid-assisted construction of single-crystalline hierarchical Nanozeolites via oriented-aggregation and intraparticle ripening. J. Am. Chem. Soc. 141, 9 (2019). https://doi.org/10.1021/jacs.8b11734
G. Yang, J. Han, Z. Qiu, X. Chen, Z. Feng, J. Yu, An amino acid-assisted approach to fabricate nanosized hierarchical TS-1 zeolites for efficient oxidative desulfurization. Inorg. Chem. Front. 7, 10 (2020). https://doi.org/10.1039/C9QI01543D
J. Zhao, L. Dong, Y. Wang, J. Zhang, R. Zhu, C. Li, M. Hong, Amino-acid modulated hierarchical In/H-Beta zeolites for selective catalytic reduction of NO with CH4 in the presence of H2O and SO2. Nanoscale. 14, 15 (2022). https://doi.org/10.1039/D2NR00731B
M. Hong, Z.W. Chen, J. Zhang, L. Dong, Y.D. Wang, C. Chen, W. Qian, S.W. Wang, Z.A. Huang, X.N. Yuan, Synthesis and application of hierarchical zeolites prepared using amino-acid mesoporogens. IOP Conf. Ser. : Mater. Sci. Eng. 479, 012113 (2019). https://doi.org/10.1088/1757-899X/479/1/012113
A.M. Naglah, M.A. Al-Omar, A.A. Almehizia, H.M. AlKahtani, A.J. Obaidullah, M.A. Bhat, N.S. Al-Shakliah, Application of Nanosized Zeolite X modified with glutamic acid as a Novel Composite for the efficient removal of Co(II) ions from aqueous media. J. Inorg. Organomet. Polym. Mater. 31, 2105–2115 (2021). https://doi.org/10.1007/s10904-021-01913-6
D.P.S. Silva, A.T. Santos, T.R.S. Ribeiro, J.R.S. Solano, R.K.B.C. Cavalcanti, B.J.B. Silva, P.H.L. Quintela, A.O.S. Silva, Monosodium glutamate-mediated hierarchical porous formation in LTA zeolite to enhance CO2 adsorption performance. J. Sol-Gel Sci. 100, 360–372 (2021). https://doi.org/10.1007/s10971-021-05644-5
A. Behr, T. Seidensticker, Building blocks of life - amino acids. Chemistry of Renewables (Springer, Berlin, 2020), 251–264. https://doi.org/10.1007/978-3-662-61430-3_14
B.O. Hincapie, L.J. Garces, Q. Zhang, A. Sacco, S.L. Suib, Synthesis of mordenite nanocrystals. Microporous Mesoporous Mater. 67, 19–26 (2004). https://doi.org/10.1016/j.micromeso.2003.09.026
T. Xiao, M. Yabushita, T. Nishitoba, R. Osuga, M. Yoshida, M. Matsubara, S. Maki, K. Kanie, T. Yokoi, W. Cao, A. Muramatsu, Amorphous Aluminosilicates, ACS Omega. 6, 5176–5182 (2021). https://doi.org/10.1021/acsomega.0c05059. Organic Structure-Directing Agent-Free Synthesis of Mordenite-Type Zeolites Driven by Al-Rich
L.V.S. Júnior, T.R.S. Ribeiro, B.J.B. Silva, P.H.L. Quintela, S.L. Alencar, J.G.A.P. Filho, A.O.S. Silva, Different approaches to the synthesis of ZSM-22 zeolite with application in n-heptane cracking. Res. Soc. Dev. 11, e6411326070 (2022). https://doi.org/10.33448/rsd-v11i3.26070
O.S. Travkina, M.R. Agliullin, R.Z. Kuvatova, I.N. Pavlova, N. Narender, B.I. Kutepov, New method of synthesis of hierarchical mordenite of high crystallinity and its application in hydroizomerization of benzene-n-heptane mixture. J. Porous Mater. 26, 995–1004 (2019). https://doi.org/10.1007/s10934-018-0694-0
P.K. Bajpal, M.S. Rao, K.V.G.K. Gokhale, Synthesis of Mordenite Type Zeolite. Ind. Eng. Chem. Prod. Res. Dev. 17, 223–227 (1978). https://doi.org/10.1021/i360067a009
G.J. Kim, W.S. Ahn, Direct synthesis and characterization of High-SiO2-Content mordenites. Zeolites. 11, 745–750 (1991). https://doi.org/10.1016/S0144-2449(05)80183-6
M. Choudhury, P.C. Borthakur, T. Bora, Syntheis and characterisation of silicious mordenite. Indian J. Chem. Technol. 5, 1–6 (1998)
F.C. Meunier, D. Verboekend, J.-P. Gilson, J.C. Groen, J. Pérez-Ramírez, Influence of crystal size and probe molecule on diffusion in hierarchical ZSM-5 zeolites prepared by desilication. Microporous Mesoporous Mater. 148, 115–121 (2012). https://doi.org/10.1016/j.micromeso.2011.08.002
F. Lónyi, J. Valyon, On the interpretation of the NH3-TPD patterns of H-ZSM-5 and H-mordenite. Microporous Mesoporous Mater. 47, 293–301 (2001). https://doi.org/10.1016/S1387-1811(01)00389-4
M. Niwa, N. Katada, Measurements of acidic property of zeolites by temperature programmed desorption of ammonia. Catal. Surv. Asia. 1, 215–226 (1997). https://doi.org/10.1023/A:1019033115091
A. Aranzabal, J.A. González-Marcos, M. Romero-Sáez, J.R. González-Velasco, M. Guillemot, P. Magnoux, Stability of protonic zeolites in the catalytic oxidation of chlorinated VOCs (1,2-dichloroethane). Appl. Catal. B 88, 533–541 (2009). https://doi.org/10.1016/j.apcatb.2008.10.007
J. Dhainaut, T.J. Daou, N. Bats, B. Harbuzaru, G. Lapisardi, L. Rouleau, J. Patarin, The influence of L-lysine and PDADMA on the crystal size and porosity of zeolite Y material. Microporous Mesoporous Mater. 170, 346–351 (2013). https://doi.org/10.1016/j.micromeso.2012.12.021
H.M. Aly, M.E. Moustafa, E.A. Abdelrahman, Synthesis of mordenite zeolite in absence of organic template. Adv. Powder Technol. 23, 757–760 (2012). https://doi.org/10.1016/j.apt.2011.10.003
R.S. Nunes, E.T.G. Cavalheiro, Thermal behavior of glutamic acid and its sodium, lithium and ammonium salts. J. Therm. Anal. Calorim. 87, 627–630 (2007). https://doi.org/10.1007/s10973-006-7788-7
G.J. Kim, W.S. Ahn, Direct synthesis and characterization of high-SiO2-content mordenites. Zeolites 11, 745 – 50 (1991). https://doi.org/10.1016/S0144-2449(05)80183-6
L. Zhang, A.N.C. van Laak, P.E. de Jongh, K.P. de Jong, Synthesis of large mordenite crystals with different aspect ratios. Microporous Mesoporous Mater. 126, 115–124 (2009). https://doi.org/10.1016/j.micromeso.2009.05.034
B. Velaga, N.R. Peela, Seed-assisted and OSDA-free synthesis of H-mordenite zeolites for efficient production of 5-hydroxymethylfurfural from glucose. Microporous Mesoporous Mater. 279, 211–219 (2019). https://doi.org/10.1016/j.micromeso.2018.12.028
S. Li, R.C.J. van de Poll, N. Kosinov, E.J.M. Hensen, Facile synthesis of nanosized mordenite and beta zeolites with improved catalytic performance: non-surfactant diquaternary ammonium compounds as structure-directing agents. Inorg. Chem. Front. 9, 3200–3316 (2022). https://doi.org/10.1039/D2QI00696K
T.P. Paula, M.F.V. Marques, M.R.C. Marques, M.S. Oliveira, S.N. Monteiro, Thermal and Catalytic Pyrolysis of Urban Plastic Waste: modified Mordenite and ZSM-5 zeolites. Chemistry. 4, 297–315 (2022). https://doi.org/10.3390/chemistry4020023
J.T. Miller, P.D. Hopkins, B.L. Meyers, G.J. Ray, R.T. Roginski, G.W. Zajac, N.H. Rosenbaum, The effect of nonframework aluminum on acidity in dealuminated mordenite. J. Catal. 138, 115–128 (1992). https://doi.org/10.1016/0021-9517(92)90011-6
G.A. Nasser, T. Kurniawan, T. Tago, I.A. Bakare, T. Taniguchi, Y. Nakasaka, T. Masuda, Cracking of n-hexane over hierarchical MOR zeolites derived from natural minerals. J. Taiwan. Inst. Chem. Eng. 61, 20–25 (2016). https://doi.org/10.1016/j.jtice.2015.11.025
H. Issa, J. Toufaily, T. Hamieh, J.D. Comparot, A. Sachse, L. Pinard, Mordenite etching in pyridine: textural and chemical properties rationalized by toluene disproportionation and n-hexane cracking. J. Catal. 374, 409–421 (2019). https://doi.org/10.1016/j.jcat.2019.05.004
B. Liu, D. Slocombe, M. AlKinany, H. AlMegren, J. Wang, J. Arden, A. Vai, S. Gonzalez-Cortes, T. Xiao, V. Kuznetsov, Edwards. Advances in the study of coke formation over zeolite catalysts in the methanol-to-hydrocarbon process. Appl. Petrochem. Res. 6, 209–215 (2016). https://doi.org/10.1007/s13203-016-0156-z
N. Chaouati, A. Soualah, M. Chater, L. Pinard, Beneficial changes in coke properties with alkaline treatment on aluminum-rich mordenite. J. Catal. 353, 28–36 (2017). https://doi.org/10.1016/j.jcat.2017.06.024
X. Hou, Y. Qiu, X. Zhang, G. Liu, Effects of regeneration of ZSM-5 based catalysts on light olefins production in n-pentane catalytic cracking. Chem. Eng. J. 321, 572–583 (2017). https://doi.org/10.1016/j.cej.2017.03.127
X. Li, W. Li, F. Rezaei, A. Rownaghi, Catalytic cracking of n-hexane for producing light olefins on 3D-printed monoliths of MFI and FAU zeolites. Chem. Eng. J. 333, 545–553 (2018). https://doi.org/10.1016/j.cej.2017.10.001
Acknowledgements
The authors are grateful to Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) and Fundação de Amparo à Ciência e Tecnologia de Pernambuco (FACEPE) for financial support.
Funding
This work was supported by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) and Fundação de Amparo à Ciência e Tecnologia de Pernambuco (FACEPE).
Author information
Authors and Affiliations
Contributions
E.G.C.G. and B.J.B.S.: Writing - original draft. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gouveia, E.G.C., Silva, B.J.B., Motta, R.J.B. et al. L-glutamic monosodium amino acid-assisted approach to mordenite zeolite synthesis with application in the catalytic cracking of n-hexane. J Porous Mater 31, 365–376 (2024). https://doi.org/10.1007/s10934-023-01518-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10934-023-01518-z