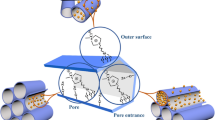
Abstract
Two different ionic liquids (ILs), 1-hexyl-3-methylimidazolium chloride and 1-ethyl-3-methylimidazolium tetrafluoroborate were used to design the mesoporous silica and to prepare a novel catalyst for the synthesis of β-amino alcohols. In this study, a feasible method was utilized for the binding of ionic liquid (IL) with mesoporous assembly, and the strong interaction of each IL with a series of mesoporous silica was confirmed from the various characterization techniques. The structural properties are studied in detail by experimental techniques as well as by DFT calculations. The chosen ILs have different cationic and anionic parts, however, almost similar activity was observed, which is investigated in the present study. This article illustrates how the strong interaction of IL with mesoporous silica culminated in the creation of a robust heterogeneous catalyst. Reusability and structural analysis of spent catalysts are used to analyze the resilience of all prepared catalysts.













Similar content being viewed by others
References
T. Welton, Ionic liquids in catalysis. Coord. Chem. Rev. 248, 2459–2477 (2004). https://doi.org/10.1016/j.ccr.2004.04.015
J. Durand, E. Teuma, M. Gómez, Ionic liquids as a medium for enantioselective catalysis. C. R. Chim. 10(3), 152–177 (2007). https://doi.org/10.1016/j.crci.2006.11.010
M. Freemantle, An introduction to ionic liquids. R. Soc. Chem. (2009)
W. Paul, Molecular weights and electrical conductivity of several fused salts. Bull. Acad. Imper. Sci. (St. Petersburg) 1800(1914).
H.L. Chum, V.R. Koch, L.L. Miller, R.A. Osteryoung, Electrochemical scrutiny of organometallic iron complexes and hexamethylbenzene in a room temperature molten salt. J. Am. Chem. Soc. 97(11), 3264–3265 (1975). https://doi.org/10.1021/ja00844a081
J.S. Wilkes, J.A. Levisky, R.A. Wilson, C.L. Hussey, Dialkylimidazolium chloroaluminate melts: a new class of room-temperature ionic liquids for electrochemistry, spectroscopy and synthesis. Inorg. Chem. 21(3), 1263–1264 (1982). https://doi.org/10.1021/ic00133a078
N. Karodia, S. Guise, C. Newlands, J.A. Andersen, Clean catalysis with ionic solvents–phosphonium tosylates for hydroformylation. Chem. Commun. 21, 2341–2342 (1998). https://doi.org/10.1039/A805376F
P. Wasserscheid, R. van Hal, A. Bösmann, 1-n-Butyl-3-methylimidazolium ([bmim]) octylsulfate–an even ‘greener’ ionic liquid. Green. Chem. 4, 400–404 (2002). https://doi.org/10.1039/B205425F
P. Wasserscheid, M. Sesing, W. Korth, Hydrogensulfate and tetrakis(hydrogensulfato)borate ionic liquids: synthesis and catalytic application in highly Brønsted-acidic systems for Friedel-Crafts alkylation. Green. Chem. 4, 134–138 (2002). https://doi.org/10.1039/B109845B
A.E. Visser, R.P. Swatloski, W.M. Reichert, R. Mayton, S. Sheff, A. Wierzbicki, J.H. Davis Jr., R.D. Rogers, Task-specific ionic liquids for the extraction of metal ions from aqueous solutions. Chem. Commun. 1, 135–136 (2001). https://doi.org/10.1039/B008041L
T.L. Merrigan, E.D. Bates, S.C. Dorman, J.H. Davis Jr., New fluorous ionic liquids function as surfactants in conventional room-temperature ionic liquids. Chem. Commun. 20, 2051–2052 (2000). https://doi.org/10.1039/B005418F
C. Hardacre, J.D. Holbrey, S.J. McMath, A highly efficient synthetic procedure for deuteriating imidazoles and imidazolium salts. Chem. Commun. 4, 367–368 (2001). https://doi.org/10.1039/B009097M
P. Wasserscheid, A. Bösmann, C. Bolm, Synthesis and properties of ionic liquids derived from the ‘chiral pool.’ Chem. Commun. 3, 200–201 (2002). https://doi.org/10.1039/B109493A
H.P. Steinrueck, P. Wasserscheid, Ionic liquids in catalysis. Catal. Lett. 145, 380–397 (2015). https://doi.org/10.1007/s10562-014-1435-x
Y. Chauvin, B. Gilbert, I. Guibard, Catalytic dimerization of alkenes by nickel complexes in organochloroaluminate molten salts. Chem. Commun. 23, 1715–1716 (1990). https://doi.org/10.1039/C39900001715
R.T. Carlin, J.S. Wilkes, Complexation of Cp2MCl2 in a chloroaluminate molten salt: relevance to homogeneous Ziegler-Natta catalysis. J. Mol. Catal. 63, 125–129 (1990). https://doi.org/10.1016/0304-5102(90)85135-5
C.M. Gordon, A.J. McLean, Photoelectron transfer from excited-state ruthenium(ii) tris(bipyridyl) to methylviologen in an ionic liquid. Chem. Commun 15, 1395–1396 (2000). https://doi.org/10.1039/B003754K
R.M. Pagni, C.M. Gordon, in CRC Handbook of Organic Photochemistry and Photobiology, 2nd edn., eds. by W.M. Horspool, F. Lenci (CRC Press, Boca Raton, 2003)
C.L. Hussey, in Chemistry of Nonaqueous Solutions, eds. by G. Mamantov, A.I. Popov (VCH, Weinheim, 1994), pp. 227–276
H. Ohno, ed. Electrochemical Aspects of Ionic Liquids (Wiley, New York, 2005).
J.H. Mazurkiewicz, P.C. Innis, G.G. Wallace, D.R. Macfarlane, M. Forsyth, Conducting polymer electrochemistry in ionic liquids. Synth. Met. 135, 32–32 (2003). https://doi.org/10.1016/S0379-6779(02)00688-4
C.M. Gordon, J.D. Holbrey, A.R. Kennedy, K.R. Seddon, Ionic liquid crystals: hexafluorophosphate salts. J. Mater. Chem. 8, 2627–2636 (1998). https://doi.org/10.1039/A806169F
M. Veber, G. Berruyer, Ionic liquid crystals: synthesis and mesomorphic properties of dimeric 2,4,6-triarylpyrylium tetrafluoroborates. Liq. Cryst. 27(5), 671–676 (2000). https://doi.org/10.1080/026782900202534
P. McNeice, P.C. Marr, A.C. Marr, Basic ionic liquids for catalysis: the road to greater stability. Catal. Sci. Technol. 11, 726–741 (2021). https://doi.org/10.1039/D0CY02274H
Y. Liu, C. Chen, Y.L. Hu, Efficient and convenient catalytic regioselective synthesis of 2-oxazolidinones from CO2 and aziridines over reusable SBA-15 supported hydroxyacetate-functionalized ionic liquid. J. Porous Mater. 29, 131–142 (2022). https://doi.org/10.1007/s10934-021-01153-6
J.R. Li, C. Chen, Y.L. Hu, Novel and efficient knoevenagel condensation over mesoporous SBA-15 supported acetate-functionalized basic ionic liquid catalyst. ChemistrySelect 5, 14578–14582 (2020). https://doi.org/10.1002/slct.202004048
X.B. Liu, Q. Rong, J. Tan, C. Chen, Y.L. Hu, Recent advances in catalytic oxidation of organic sulfides: applications of metal-ionic liquid catalytic systems. Front. Chem. (2021). https://doi.org/10.3389/fchem.2021.798603
Y. Zhang, Q. Jiao, B. Zhen, Q. Wu, H. Li, Transesterification of glycerol trioleate catalyzed by basic ionic liquids immobilized on magnetic nanoparticles: influence of pore diffusion effect. Appl. Catal. A Gen. 453, 327–333 (2013). https://doi.org/10.1016/j.apcata.2012.12.029
W. Xie, L. Hu, X. Yang, Basic ionic liquid supported on mesoporous SBA-15 silica as an efficient heterogeneous catalyst for biodiesel production. Ind. Eng. Chem. Res. 54, 1505–1512 (2015). https://doi.org/10.1021/ie5045007
T. Selvam, A. Machoke, W. Schwieger, Supported ionic liquids on non-porous and porous inorganic materials—a topical review. Appl. Catal. A Gen. 445, 92–101 (2012). https://doi.org/10.1016/j.apcata.2012.08.007
O.C. Vangeli, G.E. Romanos, K.G. Beltsios, D. Fokas, E.P. Kouvelos, K.L. Stefanopoulos, N.K. Kanellopoulos, Grafting of imidazolium based ionic liquid on the pore surface of nanoporous materials-study of physicochemical and thermodynamic properties. J. Phy. Chem. B 114, 6480–6491 (2010). https://doi.org/10.1021/jp912205y
D.W. Kim, D.O. Lim, D.H. Cho, J.C. Koh, D.W. Park, Production of dimethyl carbonate from ethylene carbonate and methanol using immobilized ionic liquids on MCM-41. Catal. Today 164, 556–560 (2011). https://doi.org/10.1016/j.cattod.2010.11.010
S. Udayakumar, Y.S. Son, M.K. Lee, S.W. Park, D.W. Park, The synthesis of chloropropylated MCM-41 through co-condensation technique: the path finding process. Appl. Catal. A Gen. 347, 192–199 (2008). https://doi.org/10.1016/j.apcata.2008.06.009
Y.S. Son, S.W. Park, D.W. Park, K.J. Oh, S.S. Kim, Absorption of carbon dioxide into glycidyl methacrylate solution containing the triethylamine immobilized ionic liquid on MCM-41 catalyst. Korean J. Chem. Eng. 26, 783–790 (2009). https://doi.org/10.1007/s11814-009-0131-9
K.S. Hwang, Y.S. Son, S.W. Park, D.W.K. Park, J. Oh, Reaction kinetics between carbon dioxide and glycidyl methacrylate using trihexylamine immobilized ionic liquid on MCM41 catalyst. J. Ind. Eng. Chem. 15, 854–859 (2009). https://doi.org/10.1016/j.jiec.2009.09.012
M.H. Valkenberg, C. DeCastro, W.F. Hölderich, Immobilisation of chloroaluminate ionic liquids on silica materials. Top. Catal. 14, 139–144 (2000). https://doi.org/10.1023/A:1009023520210
Y. Yang, C. Deng, Y. Yuan, Characterization and hydroformylation performance of mesoporous MCM-41-supported water-soluble Rh complex dissolved in ionic liquids. J. Catal. 232, 108–116 (2005). https://doi.org/10.1016/j.jcat.2005.02.017
S.M. Coman, M. Florea, V.I. Parvulescu, V. David, A. Medvedovici, D. De Vos, P. Grange, Metal-triflate ionic liquid systems immobilized onto mesoporous MS41 materials as new and efficient catalysts for N-acylation. J. Catal. 249, 359–369 (2007). https://doi.org/10.1016/j.jcat.2007.04.022
A.L. Petre, W.F. Hoelderich, M.L. Gorbaty, Dodecylbenzene transformations: dealkylation and disproportionation over immobilized ionic liquid catalysts. Appl. Catal. A Gen. 363, 100–108 (2009). https://doi.org/10.1016/j.apcata.2009.04.047
L.L. Lou, Y. Dong, K. Yu, S. Jiang, Y. Song, S. Cao, S. Liu, Chiral Ru complex immobilized on mesoporous materials by ionic liquids as heterogeneous catalysts for hydrogenation of aromatic ketones. J. Mol. Catal. A Chem. 333, 20–27 (2010). https://doi.org/10.1016/j.molcata.2010.08.018
H. Zhao, N. Yu, Y. Ding, R. Tan, C. Liu, D. Yin, H. Qiu, D. Yin, Task-specific basic ionic liquid immobilized on mesoporous silicas: efficient and reusable catalysts for Knoevenagel condensation in aqueous media. Microporous Mesoporous Mater. 136, 10–17 (2010). https://doi.org/10.1016/j.micromeso.2010.07.010
H. Wang, B. Wang, C.L. Liu, W.S. Dong, Oxidative carbonylation of methanol over copper ion-containing ionic liquids immobilized on SBA-15. Microporous Mesoporous Mater. 134, 51–57 (2010). https://doi.org/10.1016/j.micromeso.2010.05.006
S. Sahoo, P. Kumar, F. Lefebvre, S.B. Halligudi, Oxidative kinetic resolution of alcohols using chiral Mn–salen complex immobilized onto ionic liquid modified silica. Appl. Catal. A Gen. 354, 17–25 (2009). https://doi.org/10.1016/j.apcata.2008.10.039
M. Li, P.J. Pham, C.U. Pittman Jr., T. Li, SBA-15-supported ionic liquid compounds containing silver salts: novel mesoporous π-complexing sorbents for separating polyunsaturated fatty acid methyl esters. Microporous Mesoporous Mater. 117, 436–443 (2009). https://doi.org/10.1016/j.micromeso.2008.07.017
J. Huang, T. Jiang, B. Han, W. Wu, Z. Liu, Z. Xie, J. Zhang, A novel method to immobilize Ru nanoparticles on SBA-15 firmly by ionic liquid and hydrogenation of arene. Catal. Lett. 103, 59–62 (2005). https://doi.org/10.1007/s10562-005-6503-9
X. Ma, Y. Zhou, J. Zhang, A. Zhu, T. Jiang, B. Han, Solvent-free Heck reaction catalyzed by a recyclable Pd catalyst supported on SBA-15 via an ionic liquid. Green Chem. 10, 59–66 (2008). https://doi.org/10.1039/B712627A
G. Liu, M. Hou, T. Wu, T. Jiang, H. Fan, G. Yang, B. Han, Pd(ii) immobilized on mesoporous silica by N-heterocyclic carbeneionic liquids and catalysis for hydrogenation. Phys. Chem. Chem. Phys. 13, 2062–2068 (2011). https://doi.org/10.1039/C0CP01213K
B. Gadenne, P. Hesemann, J.J. Moreau, Supported ionic liquids: ordered mesoporous silicas containing covalently linked ionic species. Chem. Commun. 15, 1768–1769 (2004). https://doi.org/10.1039/B405036C
M. Vafaeezadeh, Z.B. Dizicheh, M.M. Hashemi, Mesoporous silica-functionalized dual Brønsted acidic ionic liquid as an efficient catalyst for thioacetalization of carbonyl compounds in water. Catal. Commun. 41, 96–100 (2013). https://doi.org/10.1016/j.catcom.2013.07.004
S. Sahoo, P. Kumar, F. Lefebvre, S.B. Halligudi, A chiral Mn(III) salen complex immobilized onto ionic liquid modified mesoporous silica for oxidative kinetic resolution of secondary alcohols. Tetrahedron Lett. 49, 4865–4868 (2008). https://doi.org/10.1016/j.tetlet.2008.06.014
P. Han, H. Zhang, X. Qiu, X. Ji, L. Gao, Palladium within ionic liquid functionalized mesoporous silica SBA-15 and its catalytic application in room-temperature Suzuki coupling reaction. J. Mol. Catal. A Chem. 295, 57–67 (2008). https://doi.org/10.1016/j.molcata.2008.08.016
C.J. Carrasco, F. Montilla, L. Bobadilla, S. Ivanova, J.A. Odriozola, A. Galindo, Oxodiperoxomolybdenum complex immobilized onto ionic liquid modified SBA-15 as an effective catalysis for sulfide oxidation to sulfoxides using hydrogen peroxide. Catal. Today 255, 102–108 (2015). https://doi.org/10.1016/j.cattod.2014.10.053
H.B. Wang, N. Yao, L. Wang, Y.L. Hu, Brønsted-Lewis dual acidic ionic liquid immobilized on mesoporous silica materials as an efficient cooperative catalyst for Mannich reactions. New J. Chem. 41, 10528–10531 (2017). https://doi.org/10.1039/C7NJ02541F
N. Yao, M. Lu, X.B. Liu, J. Tan, Y.L. Hu, Copper-doped mesoporous silica supported dual acidic ionic liquid as an efficient and cooperative reusability catalyst for Biginelli reaction. J. Mol. Liq. 262, 328–335 (2018). https://doi.org/10.1016/j.molliq.2018.04.121
Y.L. Hu, H.B. Wang, Z.W. Chen, X.G. Li, Titanium incorporated mesoporous silica immobilized functional ionic liquid as an efficient reusable catalyst for cycloaddition of carbon dioxide to epoxides. ChemistrySelect 3, 5087–5091 (2018). https://doi.org/10.1002/slct.201800984
E.N. Kusumawati, T. Sasaki, Highly active and stable supported Pd catalysts on ionic liquid-functionalized SBA-15 for Suzuki-Miyaura cross-coupling and transfer hydrogenation reactions. Green Energy Environ. 4, 180–189 (2019). https://doi.org/10.1016/j.gee.2019.02.003
S. Rezayati, E. Salehi, R. Hajinasiri, S.A.S. Abad, Acetic acid functionalized ionic liquid systems: an efficient and recyclable catalyst for the regioselective ring opening of epoxides with NaN3. C. R. Chim. 20, 554–558 (2017). https://doi.org/10.1016/j.crci.2016.07.004
A.K. Tripathi, R.K. Singh, Immobilization induced molecular compression of ionic liquid in ordered mesoporous matrix. J. Phys. D Appl. Phys. 51, 075301 (2018)
J. Chen, H. Wu, C. Jin, X. Zhang, Y. Xie, W. Su, Highly regioselective ring-opening of epoxides with thiophenols in ionic liquids without the use of any catalyst. Green Chem. 8, 30–332 (2006). https://doi.org/10.1039/B600620E
E.R. Nezhad, S. Sajjadifar, F. Heidarizadeh, S. Karimian, Task specific ionic liquid as solvent, catalyst and reagent for regioselective ring opening of epoxides in water. Arab. J. Chem. 12, 2098–2103 (2019). https://doi.org/10.1016/j.arabjc.2014.12.037
J.S. Yadav, B.V.S. Reddy, A.K. Basak, A.V. Narsaiah, [Bmim]BF4 ionic liquid: a novel reaction medium for the synthesis of β-amino alcohols. Tetrahedron Lett. 44, 1047–1050 (2003). https://doi.org/10.1016/S0040-4039(02)02735-1
D. Jadav, P. Shukla, R. Bandyopadhyay, P.J. Sarma, R. Deka, R. Kumar, S. Das, N. Tsunoji, M. Bandyopadhyay, Immobilization of a Zn4 complex on functionalized layered HUS-7: synthesis, structural investigation and catalytic activity. N. J. Chem. 46, 9418–9431 (2022). https://doi.org/10.1039/D2NJ00669C
M. Pandey, D. Jadav, A. Manhas, S. Kediya, N. Tsunoji, R. Kumar, S. Das, M. Bandyopadhyay, Synthesis and characterization of mononuclear Zn complex, immobilized on ordered mesoporous silica and their tunable catalytic properties. Mol. Catal. 525, 112365 (2022). https://doi.org/10.1016/j.mcat.2022.112365
R. Prajapati, D. Jadav, M. Pandey, K. Nishimura, S. Inagaki, Y. Kubota, R. Bandyopadhyay, M. Bandyopadhyay, Synthesis of hierarchical silicoaluminophosphate (SAPO) molecular sieves by post-synthetic modification and their catalytic application. Eur. J. Inorg. Chem. e20220018 (2022). https://doi.org/10.1002/ejic.202200185(Published Online)
S.A. Dharaskar, K.L. Wasewar, M.N. Varma, D.Z. Shende, K.K. Tadi, C.K. Yoo, Synthesis, characterization, and application of novel trihexyl tetradecyl phosphonium bis (2,4,4-trimethylpentyl) phosphinate for extractive desulfurization of liquid fuel. Fuel Process Technol. 123, 1–10 (2014). https://doi.org/10.1016/j.fuproc.2014.02.001
X. Bao, F. Zhang, Q. Liu, Sulfonated poly(2,5-benzimidazole) (ABPBI)/ MMT/ionic liquids composite membranes for high temperature PEM applications. Int. J. Hydrog. Energy 40, 16767–16774 (2015). https://doi.org/10.1016/j.ijhydene.2015.07.127
M. Bandyopadhyay, N. Tsunoji, T. Sano, Mesoporous MCM-48 immobilized with aminopropyltriethoxysilane: a potential catalyst for transesterification of triacetin. Catal. Lett. 147, 1040–1050 (2017). https://doi.org/10.1007/s10562-017-1997-5
D. Jadav, P. Shukla, R. Bandyopadhyay, Y. Kubota, S. Das, M. Bandyopadhyay, Tetranuclear Zn complex covalently immobilized on sulfopropylsilylated mesoporous silica: an efficient catalyst for ring opening reaction of epoxide with amine. Mol. Catal. 497, 111220 (2020). https://doi.org/10.1016/j.mcat.2020.111220
R. Dennington, T.A. Keith, J.M. Millam, GaussView 6 (Semichem Inc., Shawnee Mission, 2016)
R. Das, D.K. Pandey, V. Nimma, P. Madhusudhan, P. Bhardwaj, P. Chandravanshi, K.M.M. Shameem, D.K. Singh, R.K. Kushawaha, Strong-field ionization of polyatomic molecules: ultrafast H atom migration and bond formation in the photodissociation of CH3OH. Faraday Discuss. 228, 432–450 (2021). https://doi.org/10.1039/D0FD00129E
A.D. Becke, Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 98, 5648–5652 (1993). https://doi.org/10.1063/1.464913?ver=pdfcov
B. Marler, U. Oberhagemann, S. Vortmann, H. Gies, Influence of the sorbate type on the XRD peak intensities of loaded MCM-41. Microporous Mater. 6, 375–383 (1996). https://doi.org/10.1016/0927-6513(96)00016-8
M. Kuddushi, D.K. Pandey, D.K. Singh, J. Mata, N. Malek, An ionic hydrogel with stimuli-responsive, self-healable and injectable characteristics for the targeted and sustained delivery of doxorubicin in the treatment of breast cancer. Mater. Adv. 3, 632–646 (2022). https://doi.org/10.1039/D1MA00835H
R. Khorram, H. Raissi, A. Morsali, Assessment of solvent effects on the interaction of Carmustine drug with the pristine and COOH-functionalized single-walled carbon nanotubes: a DFT perspective. J. Mol. Liq. 240, 87–97 (2017). https://doi.org/10.1016/j.molliq.2017.05.035
M. Shahabi, H. Raissi, Comprehensive theoretical prediction of the dynamics and stability properties of Tegafur pharmaceutical agent on the Graphene based nanostructures in aqueous environment. Appl. Surf. Sci. 455, 32–36 (2018). https://doi.org/10.1016/j.apsusc.2018.05.168
D.A. McQuarrie, in Statistical Mechanics (AIP Publishing, 1965)
V. Sundararajan, N.K. Farhana, H.M. Ng, S. Ramesh, K. Ramesh, Efficiency enhancement study on addition of 1-hexyl-3-methylimidazolium iodide ionic liquid to the poly(methyl methacrylate-co-methacrylic acid) electrolyte system as applied in dye-sensitized solar cells. J. Phys. Chem. Solids. 129, 252–260 (2019). https://doi.org/10.1016/j.jpcs.2019.01.016
Y. Jeon, J. Sung, C. Seo, H. Lim, H. Cheong, M. Kang, B. Moon, Y. Ouchi, D. Kim, Structures of ionic liquids with different anions studied by infrared vibration spectroscopy. J. Phys. Chem. B. 112, 4735–4740 (2008). https://doi.org/10.1021/jp7120752
D.K. Pandey, P. Sanchora, D. Rana, P. Donfack, A. Materny, D.K. Singh, Impact of water on the hydrogen bonding between halide-based ion-pairs investigated by Raman scattering and density functional theory calculations. J. Raman. Spectrosc. 51, 147–164 (2020). https://doi.org/10.1002/jrs.5755
J. Saien, M.M.S. Badieh, M. Norouzi, S. Salehzadeh, Ionic liquid 1-hexyl-3-methylimidazolium hexafluorophosphate, an efficient solvent for extraction of acetone from aqueous solutions. J. Chem. Thermodyn. 91, 404–413 (2015). https://doi.org/10.1016/j.jct.2015.08.027
P. Sanchora, D.K. Pandey, H.L. Kagdada, A. Materny, D.K. Singh, Impact of alkyl chain length and water on the structure and properties of 1-alkyl-3-methylimidazolium chloride ionic liquids. Phys. Chem. Chem. Phys. 22, 17687–17704 (2020). https://doi.org/10.1039/D0CP01686A
M.J. Frisch, G.W. Trucks, H.B. Schlegel, G.E. Scuseria, M.A. Robb, J.R. Cheeseman, G. Scalmani, V. Barone, G.A. Petersson, H. Nakatsuji, X. Li, M. Caricato, A.V. Marenich, J. Bloino, B.G. Janesko, R. Gomperts, B. Mennucci, H.P. Hratchian, J.V. Ortiz, A.F. Izmaylov, J.L. Sonnenberg, F. Williams, F. Ding, F. Lipparini, F. Egidi, J. Goings, B. Peng, A. Petrone, T. Henderson, D. Ranasinghe, V.G. Zakrzewski, J. Gao, N. Rega, G. Zheng, W. Liang, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, K. Throssell, J.A. Montgomery Jr, J.E. Peralta, F. Ogliaro, M.J. Bearpark, J.J. Heyd, E.N. Brothers, K.N. Kudin, V.N. Staroverov, T.A. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A.P. Rendell, J.C. Burant, S.S. Iyengar, J. Tomasi, M. Cossi, J.M. Millam, M. Klene, C. Adamo, R. Cammi, J.W. Ochterski, R.L. Martin, K. Morokuma, O. Farkas, J.B. Foresman, D.J. Fox, D.J. Gaussian 16 Rev. B.01; Wallingford, CT (2016)
P. Sanchora, D.K. Pandey, D. Rana, A. Materny, D.K. Singh, Impact of size and electronegativity of halide anions on hydrogen bonds and properties of 1-ethyl-3-methylimidazolium-based ionic liquids. J. Phys. Chem. A 123, 4948–4963 (2019). https://doi.org/10.1021/acs.jpca.9b04116
D.K. Pandey, H.L. Kagdada, A. Materny, D.K. Singh, Hybrid structure of ionic liquid and TiO2 nanoclusters for efficient hydrogen evolution reaction. J. Phys. Chem. A 125, 2653–2665 (2021). https://doi.org/10.1021/acs.jpca.0c10912
Z. Wang, D. Wang, Z. Zhao, Y. Chen, J. Lan, A DFT study of the structural units in SBA-15 mesoporous molecular sieve. Comput. Theor. Chem. 963, 403–411 (2011). https://doi.org/10.1016/j.comptc.2010.11.004
K. Noack, P.S. Schulz, N. Paape, J. Kiefer, P. Wasserscheid, A. Leipertz, The role of the C2 position in interionic interactions of imidazolium based ionic liquids: a vibrational and NMR spectroscopic study. Phys. Chem. Chem. Phys. 12, 14153–14161 (2010). https://doi.org/10.1039/C0CP00486C
M. Bandyopadhyay, D. Jadav, N. Tsunoji, T. Sano, M. Sadakane, Immobilizaion of Preyssler type heteropoly acids on siliceous mesporous supports and their catalytic activities in the dehydration of ethanol. React. Kinet. Mech. Catal. 128, 139–147 (2019). https://doi.org/10.1007/s11144-019-01646-1
U. Domańska, A. Marciniak, Solubility of ionic liquid [emim][PF6] in alcohols. J. Phys. Chem. B 108, 2376–2382 (2004). https://doi.org/10.1021/jp030582h
Acknowledgements
DKS acknowledges the Science and Engineering Research Board-Department of Science and Technology (SERB-DST) for Early Career Research (ECR) project “ECR/2016/001289”. DKP acknowledges the Department of Science and Technology (DST, India) for INSPIRE Fellowship (IF170625).
Author information
Authors and Affiliations
Contributions
DJ and DKP: synthesis, catalysis, writing the original manuscript and contributed equally for this work; TP: synthesis of ionic liquid and writing of this part; DKS: reviewed the manuscript; SD: reviewed the manuscript; RB: SEM and TG analysis; NT and RK: XRD, BET analysis; MB: conceptualization, reviewing, editing, supervision, execution.
Corresponding author
Ethics declarations
Conflict of interest
There are no conflict of interests to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jadav, D., Pandey, D.K., Patil, T. et al. Ordered silica matrices supported ionic liquids as highly efficient catalysts for fine chemical synthesis. J Porous Mater 29, 2003–2017 (2022). https://doi.org/10.1007/s10934-022-01312-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10934-022-01312-3