Abstract
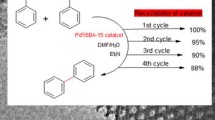
An efficient synthetic method for successful synthesis of basic amine denderon on SBA-15 and related Pd(II) complex as a novel catalyst has been developed. This modified mesoporous material was characterized by various techniques such as TEM, XRD, atomic absorption spectroscopy, N2 adsorption–desorption, and FT-IR. The prepared novel heterogeneous catalyst was evaluated in Heck coupling reaction. The very stable hybrid material catalyst was easily recovered, showed negligible Pd leaching, and retained good activity for at least ten successive runs without any additional activation. The catalytic activities of the prepared catalysts were investigated by employing aerobic oxidation of alcohols and Heck coupling as model reactions. The yields of the products were in the range from 75 to 92 %. The catalyst can be readily recovered and reused at least five consecutive cycles without significant leaching and loss its catalytic activity.
Graphical abstract






Similar content being viewed by others
References
Z. Gao, Y. Feng, F. Cui, Z. Hua, J. Zhou, Y. Zhu, J. Shi, J. Mol. Catal. A Chem. 336, 51 (2011)
V. Polshettiwar, P. Hesemann, J.E.J. Moreau, Tetrahedron 63, 6784 (2007)
K. Qiao, R. Sugimura, Q. Bao, D. Tomida, C. Yokoyama, Catal. Commun. 9, 2470 (2008)
J.Z. Jiang, C.J. Cai, Colloid Interface Sci. 299, 938 (2006)
C. Evangelisti, N. Panziera, P. Pertici, G. Vitulli, P. Salvadori, C. Battocchio, G. Polzonetti, J. Catal. 262, 287 (2009)
C. Luo, Y. Zhang, Y. Wang, J. Mol. Catal. A Chem. 229, 7 (2005)
R. Redona, N.G. GarciaPena, V.M.U. Saldivar, J.J. Garcia, J. Mol. Catal. A Chem. 300, 132 (2009)
F. Zhao, B.M. Bhanage, M. Shirai, M. Arai, J. Mol. Catal. A Chem. 142, 383 (1999)
A. Alimardanov, L.S. Vondervoort, A.H.M. Vries, J.G. Vries, Adv. Synth. Catal. 346, 1812 (2004)
M.T. Reetz, J.G. Vries, Chem. Commun. 14, 1559 (2004)
C. Amatore, A. Jutand, Acc. Chem. Res. 33, 314 (2000)
C. Rocaboy, J.A. Gladysz, Org. Lett. 4, 1993 (2002)
R. Javad Kalbasi, N. Mosaddegh, A. Abbaspourrad, Tetrahedron Lett. 53, 3763 (2012)
D.M. Ford, E.E. Simanek, D.F. Shantz, Nanotechnology 16, S458 (2005)
F. Hoffmann, M. Cornelius, J. Morell, M. Froba, Angew. Chem. Int. Ed. 45, 3216 (2006)
K. Moller, T. Bein, Chem. Mater. 10, 2950 (1998)
A. Taguchi, F. Schüth, Microporous Mesoporous Mater. 77, 1–45 (2004)
U. Ciesla, F. Schüth, Microporous Mesoporous Mater. 27, 131 (1999)
A. Corma, Chem. Rev. 97, 2373 (1997)
M.E. Davis, Nature 417, 813 (2002)
F. Schüth, W. Schmidt, Adv. Mater. 14, 629 (2002)
J.C. Hicks, C.W. Jones, Langmuir 22, 2676 (2006)
H. Li, J. Lü, Z. Zheng, R. Cao, J. Colloid Interface Sci. 353, 149 (2011)
M. Bhagiyalakshmi, S.D. Park, W.S. Cha, H.T. Jang, Appl. Surf. Sci. 256, 6660 (2010)
A. Shahbazi, H. Younesi, A. Badiei, Chem. Eng. J. 168, 505 (2011)
A. Alizadeh, M.M. Khodaei, D. Kordestani, A.H. Fallah, M. Beygzadeh, Microporous Mesoporous Mater. 159, 9 (2012)
A. Alizadeh, M.M. Khodaei, D. Kordestani, A.H. Fallah, M. Beygzadeh, J. Mol. Catal. A Chem. 372, 167 (2013)
D.Y. Zhao, Q.S. Huo, J.L. Feng, B.F. Chmelka, G.D. Stucky, J. Am. Chem. Soc. 120, 6024 (1998)
H. Veisi, Synthesis 42, 2631 (2010)
H. Veisi, Tetrahedron Lett. 51, 2109 (2010)
H. Veisi, Curr. Org. Chem. 15, 2438 (2011)
W.J. Jones, W.J. Orville-Thomas, Trans. Faraday Soc. 55, 203 (1959)
P.J. Larkin, M.P. Makowski, N.B. Colthup, L.A. Flood, Vib. Spectrosc. 17, 53 (1998)
A.D. Becke, J. Chem. Phys. 104, 1040 (1996)
N. Iranpoor, H. Firouzabadi, A. Tarassoli, M. Fereidoonnezhad, Tetrahedron 66, 2415 (2010)
W. Prukala, M. Majchrzak, C. Pietraszuk, B. Marciniec, J. Mol. Catal. A Chem. 254, 58 (2006)
W. Zhang, H. Qi, L. Li, X. Wang, J. Chen, K. Peng, Z. Wang, Green Chem. 11, 1194 (2009)
K. Khler, R.G. Heidenreich, J.G.E. Krauter, J. Pietsch, Chem. Eur. J. 8, 622 (2002)
Z. Wang, P. Xiao, B. Shen, N. He, Colloids Surf. A 276, 116 (2006)
K. Qiao, R. Sugimura, Q. Bao, D. Tomida, C. Yokoyama, Catal. Commun. 9, 2470 (2008)
S. Jana, B. Dutta, R. Bera, S. Koner, Inorg. Chem. 47, 5512 (2008)
Y. Na, S. Park, S.B. Han, H. Han, S. Ko, S. Chang, J. Am. Chem. Soc. 126, 250 (2004)
Acknowledgments
We are appreciates the Payame Noor University (PNU) for their partial support on this project.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Veisi, H., Kordestani, D. & Faraji, A.R. Palladium nanoparticles supported on an organosuperbase denderon-modified mesoporous SBA-15 as a heterogeneous catalyst in Heck coupling reaction. J Porous Mater 21, 141–148 (2014). https://doi.org/10.1007/s10934-013-9758-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10934-013-9758-3