Abstract
The sudden emergence of the novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) causing the coronavirus disease of 2019 (COVID-19) has brought the world to a standstill. Thousands of people across the globe are biting the dust with every passing day and yet more are being tested positive for the SARS-CoV-2 infection. In order to dispense this current crisis, numerous treatment options have been tried and tested and many more are still under scrutiny. The development of vaccines may help in the prevention of the global pandemic, however, there is still a need for the development of alternate approaches to combat the disease. In this review we highlight the new discoveries and furtherance in the antibody based therapeutic options and the potent drugs, with special emphasis on the development of the monoclonal and polyclonal antibodies and the repurposed drugs, which may prove to be of significant importance for the treatment of COVID-19, in the days to come. It is an attempt to evaluate the currently presented challenges so as to provide a scope for the ongoing research and assistance in the development of the effective therapeutic options against SARS-CoV-2.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
The SARS-CoV-2 (severe acute respiratory syndrome coronavirus 2) or the 2019-nCoV, responsible for the COVID-19, is a member of the ‘Coronaviridae’ family of viruses which are known to mainly cause respiratory tract illnesses ranging from common cold to SARS [1]. The first occurrence of the disease was observed during early December in Wuhan, China and it was reported to the WHO (World Health Organisation) on 31st December 2019. Thereafter, it was soon declared as a global pandemic on March 11, 2020, by the WHO. As on 18th August, 2020, there are more than 21 million confirmed active cases of COVID-19 and more than 7 lakh deaths globally [2]. The 2019-nCoV are enveloped viruses containing single-stranded positive-sense RNA. This RNA is used as a template that encodes for both non-structural (NSP) and structural proteins. There are 16 NSPs encoded by the viral genome, which play a specific role in the viral replication and transcription, 4 major structural proteins and 5–8 accessory proteins [3]. The 4 structural proteins of the virus include the membrane (M), spike (S), envelope (E) and nucleocapsid (N) proteins, that are essentially required for the viral attachment, infection and assembly. The Spike protein [S], which is a structural protein, is one of the major drug targets and is known to be composed of two subunits, S1 and S2. The S1 unit contains the RBD (receptor binding domain) which mediates the attachment of the virus to the ACE2 (angiotensin converting enzyme 2) receptor of the host (humans), whereas the S2 unit is responsible for the fusion of the host and viral cell membranes [4].
It was also confirmed that the genomic sequence of the SARS-CoV-2 was ~ 79.6% and 96% identical to the SARS-CoV and bat coronavirus, respectively [5]. In spite of being similar to the SARS-CoV (Severe acute respiratory syndrome coronavirus) and MERS-CoV (Middle East respiratory syndrome coronavirus) viruses, which had led to a similar outbreak in the years 2002 and 2012, respectively, the SARS-CoV-2 is considered to be more dangerous, since its rate of human to human transmission is much more as compared to the aforesaid viruses. It was also observed that the binding affinity of the SARS-CoV-2 to the ACE2 receptor is much higher as compared to the SARS-CoV virus, making it even riskier [6].
The very fact that the discovery and large-scale production of a SARS-CoV-2 targeted vaccine would take a very long time to get into market, makes it important to analyse and research about the existing medicinal drugs which can be utilised for the treatment. We may also develop new therapeutic options that may take a shorter time to be produced and are effective in treating the COVID-19 patients [7]. This review hence consists of a comprehensive study of some of the re-purposed drugs and antibody based therapeutic options, that have been developed or are in pipeline, for controlling the COVID-19 pandemic outcomes in the future. It provides insights into the recent discoveries and advancements related to them.
2 Antibodies for the Expeditious Rescue
Today the major research focuses on the development of antibody (Ab) molecules against the SARS-CoV-2. The administration of passive Abs either help to neutralize the antigen (Ag), which is expected to be present in smaller quantities during the initial phase of infection or mediate the modification of the inflammatory and cellular immune response, which is also easy to manage before the actual symptoms start to appear. The efficiency and effectiveness of the therapy is largely dependent on the composition and amount of Abs conferred [8].
As the technology has advanced, it has become possible over the last few decades, to purify and manufacture immunoglobulins (IGs) and hyperimmune globulins (H-IGs). The H-IGs preparations are often reported to be more effective as compared to the IVIgs (intravenous immunoglobins), since these preparations contain known titers of Ab against a specific Ag, derived from the plasma of sero positive donors with sufficient titers of NAbs (Neutralizing antibodies). The H-IGs also present an advantage over the CP (convalescent plasma), because it prevents the transfer of any harmful coagulation factors present in the serum and its composition and preparation is more consistent as compared to CP [9]. Recently, a Japanese drug making company called Takeda Pharmaceutical Co., was reported to experimentally develop anti-SARS-CoV-2 polyclonal hyperimmune globulins (H-IGs)) that was derived from the blood plasma of the recovered COVID-19 individuals [10].
The onset of the coronavirus infection begins when the RBD in the spike protein of the virus binds to the host cell surface receptor, ACE2, in case of both SARS-CoV and SARS-CoV2. Hence an effective anti-SARS-CoV2 mAb (monoclonal antibody) or an anti-ACE2 mAb might prove to be an effective method of fighting against CoV-19. NAbs like 80R, CR3014, F26G18, F26G19, etc. binding to the spike receptor binding protein in SARS-CoV were discovered earlier [11]. It was observed that the NAbs that targeted the ACE2 binding site of SARS-CoV, did not bind to the SARS-CoV2, implying that there was a difference between the RBDs of the two strains, that have an important role to play. Thus, the development of novel NAbs specific for SARS-CoV-2 RBD is the need of the hour [12]. An interesting hypothesis which gathered attention was that the rabbit sera, derived from rabbits that are immune to SARS and contained Abs raised against its S1 subunit, was observed to reduce the entry of both SARS-CoV and SARS-CoV-2 efficiently. Thus, Abs raised in response to SARS-CoV were observed to offer some level of protection against SARS-CoV-2, however the level of cross reactivity and efficiency of these Abs to neutralize the novel virus has not yet been determined. Two available IVIgs preparations: Gamunex®-C (Grifols Therapeutics, Inc., NC, USA) and Flebogamma® DIF (Instituto Grifols S.A., Barcelona, Spain), initially developed using plasma from donor patients of USA, were also observed to contain Abs that cross reacted positively with the SARS-CoV-2, at a concentration of 100 μg/ml and 1 mg/ml, respectively. These can thus be used as a potent therapeutic option; however further validation of these IVIgs is still under process [13, 14]. In a recent study a group of scientists characterized 206 RBD specific mAbs from single cell B cells of eight COVID-19 infected patients by using the FACS (fluorescence activated cell sorting) technique. The effectiveness of binding and neutralization of these Abs were tested using surface plasmon resonance assay and neutralization assay on both the pseudotyped and original SARS-CoV-2 virus. The crystal structure analysis of the RBD bound Ab revealed 2 potential human monoclonal NAbs named P2C-1F11 and P2B-2F6 [15].
Another study based on ELISA-cross reactivity of Ab containing supernatants, of SARS-S hybridomas (spike protein antibodies obtained using hybridoma technology) obtained from immunized mice encoding chimeric immunoglobulins (Human- Heavy and Light chains; H2L2 and constant region of rat origin), was also conducted recently. The hybridomas were made by the fusion of the spleen cells of the immunized mouse and the SP2/0 myeloma cells. 4 of these Abs showed cross reactivity with the S1 subunit and 1 out of these even showed cross neutralizing activity towards both SARS-CoV and SARS-CoV-2. This chimeric Ab was named as 47D11 H2L2, which was fully humanized by cloning the VH and VL human chain regions into a human IgG1 isotype backbone. This finally led to the creation of the first recombinantly expressed human 47D11 mAb, against the SARS-CoV-2. The Ab was also successful in preventing the VeroE6 cells from the infection of SARS-CoV and SARS-CoV-2 pseudo typed viruses [16]. Attempts have also been made to develop a recombinant Ab by the fusion of the extracellular domain of the ACE2 to the constant region of the human IgG1 immunoglobulin. The recombinant Ab showed high affinity towards the RBD and was suggested and tested to neutralize both SARS-CoV and SARS-CoV-2 pseudotyped viruses, in mice. However, for high scale development for therapeutic purposes, this rAb was limited by the short half-life and was also not tested in-vivo on the live viruses [17].
In the U.S., many teams are independently running various studies to discover mAbs, for the emerging threat of COVID-19. The research groups are funded by the Pentagon’s defense advanced research projects agency (DARPA) and even by the Bill and Melinda Gates foundation. AbCellera Biologics, Canada, is currently working on an engineered version of spike protein as a bait to collect Abs against SARS-CoV-2. They collected 500 Abs against the spike protein from a single patient of Seattle, out of which 24 were the most potent ones, since they retained their shape on mass production and were bound to the S protein for the longest time. Another US based company, Regeneron, is also trying to develop a pair of Ab formulation or an Ab cocktail preparation (human and mouse derived Abs) which targets or binds to different sites of the spike protein itself, ensuring a better neutralizing ability, taking into account the mutations that may occur in the SARS-CoV-2. Vir Biotechnology, has also come up with an Ab that was obtained from a SARS-CoV recovered patients of 2003, which cross neutralizes the SARS-CoV-2 by binding to the highly conserved region of its RBD. They are now trying to make further modifications to the Ab, to increase its effective half-life. The modifications being made are of two kinds: one which extends the viable life of the antibody by preventing it from degradation while the other improves the supposed vaccinal impact, which signals T cells to help decimate the infected cells.
In his recent research, Brouwer et al. also isolated mAbs from the convalescent plasma of 3 COVID-19 patients. In order to capture the S protein specific Abs, present in the sera of the patients, stabilization techniques were used to generate a soluble and stabilized S protein in its prefusion conformation. These S proteins were further fluorescently labelled and used to analyse the S-protein specific B cells using flow cytometry technique. These cells were then sorted out from the entire pool of B cells, for the isolation of S protein specific mAbs. The Abs were then expressed in the HEK293T cells and screened for the binding to the S protein using ELISA technique. On the other hand, Surface Plasmon Resonance based competition assays and electron microscopy methods were used to demonstrate that the S protein of SARS-CoV-2 contained various antigenic sites (RBD or non-RBD epitopes). Further, 84 mAbs that showed affinity towards the S protein were selected and these were further narrowed down to 19 Abs that efficiently inhibited the pseudo SARS-CoV-2 virus by binding to various different antigenic sites on the spike protein. 14 out of these 19 mAbs were found to bind to the receptor binding domain and 2 were expected to be highly potent NAbs which showed a 47 and 60 pM (picomolar) neutralizing activity [18].
Many other groups like the collaborative team of AstraZeneca and Vanderbilt University, are creating a huge library of random Abs, using phages that are targeting regions other than the RBD of the spike protein of the novel virus. Ever since the search for the Ab against the SARS-CoV-2 began, many attempts have been made to develop these Abs using the phage display technology. In this technique a gene that encodes for the protein (antibody) of interest, is integrated into a phage (virus that infects bacteria) genome, such that it allows the expression of this gene on the surface or capsid of the phage, thus, displaying it on the exterior. This phage can then keep producing similar new phages that are displaying desired peptides, once they infect a bacteria (commonly E.Coli) and uses its replication machinery for this purpose. A few of these developments include: a single-domain Ab from llama which neutralizes the S antigen of SARS-CoV and pseudotyped SARS-CoV-2 as a bivalent human IgG Fc-fusion (fragment crystallizable) protein and a synthetic human Fab (fragment antigen binding) library screened against the RBD domain of the SARS-CoV-2 [19, 20]. Computer techniques like molecular modelling softwares are also being applied, by scientist like, Jacob Glanville, Distributed Bio, to develop a huge library of variants of Abs of SARS, which could be further sorted to find Abs that binds to the SARS-CoV-2 and neutralize a broad range of other coronaviruses in future [21].
GigaGen Inc., has also initiated development of recombinant anti-coronavirus (COVID-19) hyperimmune gamma globulin (rCIG) polyclonal antibody (pAbs) after agreement from the FDA (Food and Drug Administration). The company uses single cell technology to recreate the library of COVID-19 Abs. rCIG helps in reproduction of the complete Ab reserves (B cells reserve) from recovered patients and this will be used to intravenously inject the COVID-19 patients. It can also be used as a prophylaxis in case of high-risk individuals. The recombinant pAbs are expected to have a higher potency as compared to plasma derived antibodies [22]. Another pAb called COVID 19 Spike Protein Coronavirus Polyclonal Antibody or SARS-CoV-2 Spike Antibody, is also under investigation. It is an IgG isotype derived from rabbit (the host system) [23]. Attempts are also being made to design pAbs which target different regions including the S1 RBD, Nucleocapsid protein, S2 subunit, etc. [24].
2.1 Therapeutic Aspects of Convalescent Plasma in COVID-19
The convalescent plasma (CP) therapy is a very old approach which involves the transfusion of blood serum of a person, who has just recovered from a viral infection and hence contains the Abs raised against the infecting antigen, into another infected person. This helps the infected person in fighting the viral antigen, due to the presence of Abs that either bind to it or activate the immune response (cellular cytotoxicity or phagocytosis) against it [25]. Several cases and studies based on treating various viral infections by the early administration of the CP have been reported earlier [Supplementary Material Table 1].
These findings helped hypothesize that the use CP therapy may also prove to be beneficial as an immediate solution for the patients of CoV-19. A study was conducted in the Third People’s Hospital in Shenzhen, China, between 20th January and 25th March, 2020, where 5 patients that were critically ill with the CoV-19 infection, were treated with CP containing NAbs [26, 27]. Similar studies were also carried out in different parts of China and South Korea, for different durations between January-March, 2020, on critically ill CoV-19 patients [28,29,30]. All 27 patients, however, showed no adverse effects, indicating that the CP therapy was well tolerated among patients. The clinical status of all the patients improved and an increase in the Ab titre and decrease in the viral load within 12–45 days of transfusion were observed. These patients were however, supported by antiviral drugs or hydroxychloroquine, which made it difficult to conclude the exact consequences of the therapy alone and the results were not completely confirmative, since these were tested on a very small number of patients, that included no control. All the studies, though in favour of the therapy, suggested that an optimal dose, timing and procedure, is still required for further investigation via controlled clinical trials on a larger scale [31, 32].
CP therapy was proposed by US FDA for treating severely ill CoV-19 patients on 24th March 2020, whereas the requirements to be fulfilled by the donor were established by the FDA on 11th April, 2020 [33]. The ICMR (Indian Council of Medical Research) has also approved to conduct the clinical trials for the CP therapy at the SCTIMST (Sree Chitra Tirunal Institute for Medical Sciences and Technology), India [34]. CP therapy is also currently in clinical trials under the COVID-19 Convalescent Plasma Project, a program in the U.S. In France, Germany, Italy, Netherlands and Belgium as well, the blood transfusion centers are aiming at collection of plasma from donor patients [35].
Even though CP therapy has shown positive outcomes in numerous cases, it also has some risks associated with it. These risks broadly fall into two categories: serum sickness and Ab dependent enhancement of infection (ADE). The serum disease is associated with the risk related to the transfer of components like other infectious agents from the blood serum of the donor, to the patient being treated whereas the ADE occurs when the Abs contrarily mediate the viral entry and eventually infection in host cells [36]. There is no evidence of occurrence of ADE in COVID-19 patients, until date. However, the ADE was observed to occur in the infection of SARS-CoV. Also, studies in macaque models (primates) suggested an enhanced pro-inflammatory response in alveolar macrophage, which lead to acute lung injuries, when treated with anti-SARS-CoV Spike IgG [37]. CP also has other issues associated with its administration, which include the variable NAbs titres, infectious agents getting transferred during transfusion, difficulty in collecting the plasma from suitable donors, etc.[38]. To avoid such risks to occur while treating the SARS-CoV-2 infection, various conditions have to be met before actually implementing the administration of the convalescent serum to the CoV-19 patients.
2.2 Controlling the Cytokine Storm Syndrome
The cytokine storm has been observed to occur during infectious diseases including the ones caused by influenza viruses and coronaviruses. It is an exuberant immune response which leads to an elevated level of cytokines/chemokines, causing acute lung injuries, ARDS (acute respiratory distress syndrome), multiple organ failure and even death, in many infected patients. A report from China showed that there is an elevated concentration of all the anti and pro-inflammatory response factors, that is, ILs (Interleukins: IL-2ra, IL-6, IL-10, IL-18), IF γ (Interferon), monocyte chemotactic protein 3, granulocyte colony stimulating factor, interferon gamma induced protein 10, macrophage inflammatory protein 1 alpha, tumor necrosis factors (TNF), etc., in the critically ill COVID-19 patients [39, 40]. These studies helped understand that the cytokines may be utilized as biomarkers to predict the progression and severity of the infection. This leads to the implication that cytokine storm should also be considered of significant importance while looking for therapeutic opportunities, for COVID-19 [41].
A good therapeutic option is Tocilizumab, which is a recombinant and humanized mAb that specifically binds and blocks the IL-6 receptors [42, 43]. It has the ability to bind to both the mIL6R (transmembrane IL-6 receptor) and sIL-6R (soluble IL-6 receptors) receptors of IL-6. It is sold under the name Actemra, which was earlier approved by FDA for use in treatment of diseases like rheumatoid arthritis, etc. [44]. The clinical examinations with this therapeutic mAb have shown to reduce fever and C-proteins (complement proteins) within the patients of COVID-19 worldwide [45,46,47,48]. Recently, Roche’s Genentech unit received US FDA approval after clearing the phase 3 trial of Actemra, in severely ill COVID-19 patients [49]. Several other Ab preparations like the completely humanized IgG1 mAb, called Emapalumab, which earlier got an FDA approval for the treatment of haemophagocytic disorder and Infliximab and Etanercept, known for their use in treating autoimmune diseases, could also be suggestive therapeutic options. Emapalumab is known to bind to both free and receptor bound IF γ whereas Infliximab and Etanercept target the TNF α, reducing the hyper immune response. Although more studies, research and clinical trials need to be done on the safety and efficacy of these drugs [50]. Recently, IL-1 targeting mAb called Canakinumab, a humanized IgG1 mAb called Gimsilumab targeting the granulocyte–macrophage colony-stimulating factor (GM-CSF) and Rituximab-CD20, an anti-CD-20 mAb, have also entered into clinical trials for the treatment of patients with COVID-19 [50,51,52]. Lenzilumab, a GM-CSF targeting drug has also received an approval by the FDA for the phase three trials, considering the significant role GM-CSF may play in the hyperinflammatory response in the COVID-19 patients [53].
The immunomodulatory agents may also be used in combination with the antiviral drugs, to reduce the cytokine storm effects in the COVID-19 patients. These agents include: corticosteroids, interferon gamma, IVIg, IL-1 and IL-6 antagonists (like Anakinra, etc.), TNF blockers, chloroquine, hydroxychloroquine, ulinastatin, etc. [54,55,56]. All these agents may directly or indirectly help in suppressing the effects of the cytokine storm. Some other drugs that are currently under Phase 2 clinical trials include Sarilumab, an IL-6 receptor inhibitor; Baricitinib and Ruxolitinib, JAK-STAT signalling inhibitors; Fingolimod, which inactivates IL-6 and also STAT3 pathway and statins which show anti-inflammatory effects by blocking NF-κB and NLRP3, are being explored [57, 58]. Minocycline, another drug with potential re-purposing capabilities, has also been recently proposed for the management of the cytokine storm effects in the COVID-19 patients due to its potent immunomodulatory effects [59].
A recent study suggested the use of ursodeoxycholic acid or UDCA, in the treatment of COVID-19, may help in preventing inflammation and apoptosis, thereby, projecting new opportunities which still require to be evaluated. This study was in support of China promoting the use of Bear Bile, which is rich in the UDCA [60]. Hypothesis of usage of drugs like Pirfenidone and Thalidomide, that had initially been developed for the treatment of Idiopathic Pulmonary Fibrosis and morning sickness, respectively, have also gained much attention. Both of the drugs have been observed to decrease the effects of a number of released cytokines. Pirfenidone is also known to reduce the expression of the ACE2 host receptor and shows antioxidant and antiapoptotic effects. On the other hand, Thalidomide shows anti-inflammatory effects, thus, making both these drugs potent fighters in the battle against COVID-19 [61, 62]. Another therapeutic option, which has also been observed to downregulate the ACE2 and TMPRSS2 (activates spike protein in SARS-CoV-2) expression levels in the human liver derived cells, is Nrf2-activator PB125® [63].
3 Other Pharmacotherapies to Counter COVID-19
Apart from the various attempts in developing the monoclonal and polyclonal antibodies targeting either the human receptor, viral proteins or the immune response elements, the other major focus is towards the medications/drugs that have been repurposed, in order to treat the COVID-19 patients (Fig. 1). Some of these majorly used medications, such as Remdesivir, Lopinavir-Ritonavir, Hydroxychloroquine (HCQ), Favipiravir, etc. are extensively under clinical trials. Each of these drugs have their own mechanism of action, advantages and side effects of administration.
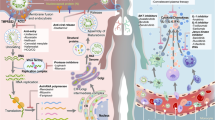
Inhibitory functions and roles of different antibodies and re-purposed drugs for battling COVID-19. The COVID-19 (coronavirus disease of 2019) infection begins with the attachment of the spike protein of the SARS-CoV-2 (severe acute respiratory syndrome coronavirus 2) to the ACE2 (angiotensin converting enzyme 2) human receptor. Various monoclonal (mAb) and polyclonal antibodies are being designed and convalescent plasma collected from the donor patients are being used to inhibit the attachment of the novel virus. This is followed by the virus and host membrane fusion and entry of the virus which is inhibited by drugs like Hydroxychloroquine (HCQ) and Chloroquine (CQ). Once the uncoating occurs and the viral RNA is released, it starts replicating with the help of the enzyme RNA dependent RNA polymerase (RdRp). Drugs like Remdesivir, Favipiravir and Ribavirin, interfere with the activity of this enzyme. In order to further generate viral proteins, the processing (capping) of the viral RNA is required for maintaining its stability. This processing is inhibited by drugs like Ribavirin. On the other hand, combination of Lopinavir and Ritonavir is used for inhibiting the proteases like 3CLpro (chymotrypsin like cysteine protease) which are mainly responsible for the proteolytic cleavage and generation of non-structural proteins involved in viral replication. Once the viral assembly is complete and the new virions are ready, the exocytosis of these virions occur. This is inhibited by drugs like CQ and HCQ. The infection by the novel virus and release of the new virions which infect the neighbouring cells of the body lead to an exaggerated cytokine release which can be managed using drugs like corticosteroids, various mAbs targeting different cytokines and cytokine receptors, etc.
3.1 Remdesivir
Remdesivir is a well-known antiviral drug. It is metabolized into its active adenine analogue that interferes with the activity of RNA dependent RNA polymerase (RdRp), further promoting the viral exoribonuclease activity of proofreading and thereby inhibit the synthesis of viral RNA. The drug is responsible for lowering the RNA levels of the virus by acting at an early stage of viral infection [64]. It had previously shown its efficacy while treating the Ebola virus infection in Rhesus monkeys and was also approved for use in treating infections caused by MERS and SARS-CoV. The potential of Remdesivir for the treatment of SARS-CoV-2 was proven in the clinical trials that were held in various parts of the world. The drug effectively treated 14 American cruise passengers in Japan who were infected by the novel virus and even patients in the USA who administered this drug after showing pneumonia symptoms [65, 66]. The importance of this drug was also stated in the guidelines issued by American Society of Heath System Pharmacists (ASHP) and International Pharmaceutical Federation [67]. In March 2020, the third phase clinical trial of the drug began in humans, where each patient was given 200 mg of dose on the first day followed by 100 mg from the second day in continuation [68, 69]. It showed great responses in the treatment of COVID-19 patients but was accompanied by certain side effects such as nausea, skin disorders, etc. [70].
Post the approval by FDA on 1st of May 2020, the drug was also announced to be used as an emergency treatment option for COVID-19 treatment by the Drug Controller General of India on 1st of June, 2020. Companies such as Cipla, Jubilant Life Sciences, Dr. Reddy’s Laboratory, Ferozsons Laboratory and Zydus Cadila were approached to produce the drug in India. Hetero and Cipla, being the only approved manufactures priced the drug for Rs.5400 and less than Rs. 5000, respectively [71, 72].
3.2 Lopinavir-Ritonavir
Lopinavir-ritonavir act as inhibitors of proteases such as 3C like and papain-like proteases, which are important targets of the Ortho Coronaviridae family. In India, this drug has been previously used to prevent the transmission of HIV from infected mother to child. Amongst several other anti-HIV drugs which possess abilities of efficient polyprotein processing, it has been observed that Lopinavir showed the highest inhibitory activity against SARS-CoV-2. The recommended drug dosage includes 200 mg/50 mg. In patients having trouble taking oral dosage, 400 mg/100 mg concentration of 5 ml suspension, until 12 or 14 days is prescribed [73]. Lopinavir in combination with low doses of ritonavir is efficient in inhibiting the antiretroviral protease. This combination is sold under the brand Kaletra. Ritonavir acts as an obstruction in the metabolism of Lopinavir, thereby increasing the half-life of the drug and hence its antiviral activity [65]. Ritonavir assisted combination therapy was promoted by both Infectious Disease Society of America and American Association for study of Liver Diseases, as a potential treatment for COVID-19 [74]. In the guidelines issued by Government of India, Ministry of Health and Welfare, as dated on 17th March 2020, the use of Lopinavir-ritonavir combination was recommended for the treatment of COVID-19 [75].
However, the clinical trials for the drug, conducted on 199 patients in Wuhan, that were further divided into two groups, one group getting the lopinavir -ritonavir combination while other treated with standard cure showed no difference in the combination therapy as compared to the standard care. Though the clinical improvements were seen in both the groups. The adverse effects were less in the patients treated with the combination of both drugs, but no reduction in the levels of viral load was observed when compared with the standard care [76]. Even according to the British researchers, the combination of lopinavir and ritonavir showed no efficacy during the UK recovery trials of COVID 19 patients. The chief investigator of the trials conducted by the Oxford, stated that the drug had no clinical benefits in treating the COVID-19 patients [77] and hence more studies are required to be conducted in this regard.
3.3 Favipiravir
This antiviral drug was used to treat the influenza virus infection in 2014 after being approved in Japan. The drug hinders the viral replication, by binding to the conserved domain of polymerase which further puts a stop to the addition of nucleotides into the growing viral RNA, during replication [78]. The drug gets converted into an active phosphoribosyl form which is recognized by the viral RdRp (RNA dependent RNA polymerase) as a substrate thus, inhibiting its polymerase activity [79].
On 17th March, 2020, the Director of China National Centre for Biotechnology announced that Favipiravir was successfully tested during a clinical trial held on 80 patients infected by SARS-CoV-2 [80]. Studies were also conducted in other parts of China and Japan on a larger section of patients. It was observed in all these studies, that the patients administered with Favipiravir, showed to be negative for the infection caused by the virus, in a shorter period as compared to some other anti-viral drugs. Researchers from Stanford are also planning to conduct clinical trials for further checking the effectiveness of Favipiravir on 120 COVID-19 patients [81]. Recently the Glenmark Pharmaceuticals was given the approval for manufacturing the drug in India, on 19th June 2020. The recommended dosage of the drug is 1800 mg twice on the first day followed by 800 mg twice for the next 14 days. The drug is currently being sold at a price of Rs 103 per tablet for mild to moderate cases of COVID-19 [82].
3.4 Hydroxychloroquine in Combination with Azithromycin
Towards the end of World War II, the Germans found the substitute of quinacrine (compound derived from Cinchona bark) called Chloroquine (CQ), which was explored to be effective not only as an antimalarial drug but also against arthritis, lupus erythematosus rashes, prophylaxis and other diseases with symptoms of fever. Later, an even safer derivative of CQ, with an OH group added to its composition was developed, called HCQ. Though both these drugs have commonly been used for many years now, but its usual symptoms also include dermatological changes, pruritus, nausea, cardiovascular issues, headaches, abdominal pain, etc. [83]. The mechanism of action of both the drugs in treating COVID-19, is not very well studied. However, it is suggested that the intracellular phagolysosome is alkalised (rise in endosomal pH) by these drugs, thus preventing the fusion and uncoating of the viral membrane and hence the spread of the virus [84]. As per the observation made after the clinical trials of CQ/HCQ, it was concluded that these drugs inhibit the entry of both SARS-CoV and SARS-CoV-2, by impeding the terminal glycosylation of ACE2 host receptors [85]. Studies show that HCQ had the better potential as a drug against COVID-19 as compared to CQ [86]. In view of a paper published in Lancet describing the concerns and increase in the mortality rate due to use of HCQ for the treatment of COVID-19, the WHO officially halted the ongoing clinical trials of the drug on 25th May, 2020. A week later, after further clarification and better understanding these trials were resumed [87, 88]. However, soon studies revealed that the benefits of the drug were not able to outnumber the risks that came along with the usage of this drug in the treatment of COVID-19. Thus on 15th of June, 2020, FDA in consultation with BARDA, announced the cancellation of its usage as an emergency authorized drug [89].
Azithromycin belonging to the macrolide class of antibiotics, is another drug used in combination with HCQ, for the treatment of COVID-19 patients. This drug is commonly used for the treatment of MAC (Mycobacterium avium complex), bronchitis, or pneumonia infections. A group of researchers from the University of New Mexico demonstrated that Azithromycin behaves like a weak base having acidotropic lyophilic properties and regulates the pH of the Golgi transport network as well as endosomes. Its effects on various organelles were similar to that caused by HCQ [90]. In a controlled study on 86 infected patients, treated with a combination of HCQ and Azithromycin, negative results of the viral infection by 97.5%, were observed on day 5 after administration, implying it to be an effective therapeutic option against CoV-2 infection [91]. A comparative study also revealed that the combination of these two drugs helped cured 100% of the patients (under study) after 5–6 days while the recovery was observed in only 57.1% patients when treated with HCQ alone [92].
3.5 Ribavirin
Ribavirin is a drug that is not only known to interfere with the DNA and RNA polymerisation process in viruses, but also with the capping process of the RNA. It also inhibits the inosine monophosphate dehydrogenase, which is an essential enzyme for the production of guanosine [93]. The usage of this drug and its benefits were also analysed in earlier outbreaks of SARS-CoV and MERS-CoV, where it was used in combination with several other drugs like corticosteroids, lopinavir/ritonavir, oseltamivir, prednisolone, interferons, etc. However, no previously conducted studies gave any clear answers regarding its advantages [83, 94,95,96]. These previously conducted studies of the use of ribavirin in treating the viral infections caused by viruses similar to SARS-CoV-2, encouraged the use of this drug in treating patients of COVID-19 as well. Many researchers are still trying to evaluate the role of this drug in fighting the novel coronavirus, around the world [97,98,99,100,101]. However, no studies have yet proven its efficiency and even the optimum dosage prescriptions are yet to be finalised for the combinational or non-combinational use of this drug.
4 Discussion
The seamless spread of the novel coronavirus and the augmented rise in the number of infected patients, indicate that quarantine alone cannot help in controlling the devastating risks and threats that the SARS-CoV-2 infection is imposing on the world. As the spread of the virus and consequently the mortality rate of the infected individuals is increasing at a rapid pace, even the research and health/clinical care community are facing several challenges, globally, to find potential therapies or drugs to fight the novel virus. Studies related to the genome and structure of the novel coronavirus, has immensely boosted the process of designing rapid test kits, vaccines, antibodies and drugs to combat the viral infection. Many structural proteins of the SARS-CoV-2, host receptor: ACE2 and cytokines/chemokines have been projected to be significant drug targets. Although, it is important to emphasize that there exist many non-structural and accessory proteins of the virus, which are yet to be characterized and well-understood. These might also help in developing suitable therapeutic targets once their roles are well defined.
Several antiviral and immunomodulatory drugs, vaccines, antibodies, protease inhibitors and IFN and stem cell-based therapies are also under extensive study while some are under clinical trials. Very recently, India’s first COVID-19 “inactivated” vaccine, “COVAXIN”, developed by Bharat Biotech, had entered the first phase of the human clinical trials, in collaboration with ICMR and NIV, Pune [102]. Another Indian, plasmid DNA based vaccine, called ZyCoV-D was developed by Zydus Cadila, Ahmedabad. On 15th July, 2020, this vaccine also entered the Phase I/II human clinical trials [103]. Another adenoviral vector-based vaccine, called ChAdOx1 nCoV-19 or AZD1222, was helpful in generating a strong innate, humoral (development of neutralizing Abs within 28 days of vaccination) and cellular (development of T-cell to attack the virus within 14 days of vaccination) immune response to the RBD of the S protein of SARS-CoV-2. It was reported to be developed at the Oxford University in collaboration with Astra Zeneca. The vaccine had successfully completed the Phase I/II trials and has recently entered into the Phase III trials [104, 105]. Some mRNA-based vaccines have also gathered much attention in the recent times. These include the mRNA-1273, developed by Moderna in consortium with NIAID (National Institute of Allergy and Infectious Diseases), U.S. The vaccine utilizes the ability of the mRNA to encode for a full-length S protein, which then commences an immune response allowing generation of Abs against SARS-CoV-2. This vaccine candidate entered into the Phase 3 clinical trials on 27th July, 2020 [106, 107]. Pfizer in association with BioNTech have also developed a similar mRNA vaccine called BNT162b2, with a single nucleoside modification in the mRNA sequence of the S protein. The Phase 2/3 trials have also begun for this vaccine [108].
Even though the vaccine development is occurring at an unprecedented rate, it will take a long time to manufacture and vaccinate a large number of people worldwide. In order to bridge this gap, the focus shifted towards the development of antibody-based therapy and repurposing drugs in order to meet the need of the hour (Table 1). Multiple other drugs apart from the repurposed drugs discussed earlier are being evaluated for their potential use in combating COVID-19. These include Ivermectin, Nitazoxanide, Lianhuaqingwen capsules, Dipyridamole, etc.[109].
Though such extensive research work is being carried out all over the world, no specific treatment/therapy/drug for COVID-19 is yet available. There are several challenges to overcome, which not only includes the development of effective therapeutic options and diagnosis kits, but also, select, evaluate and mass produce them. There is also a need to conduct further studies to explore and perform in depth analysis about the pathogenesis of the novel virus, which could possibly lead us to identify new drug targets. Thus, the greatest challenge today undoubtedly requires the entire research community to work with cooperation towards the common goal and the day is not far off, when a therapeutic agent will prove to be effective against COVID-19.
Abbreviations
- IL:
-
Interleukin
- GM-CSF:
-
Granulocyte–macrophage colony-stimulating factor
- IFN:
-
Interferon
- TNF:
-
Tumor necrosis factor
- ARDS:
-
Acute respiratory distress syndrome
- APC:
-
Antigen presenting cells
- MHC:
-
Major Histocompatibility Complex
- TCR:
-
T-cell receptor
- CTLs:
-
Cytotoxic T lymphocyte
References
Coronavirus |NIH: National Institute of Allergy and Infectious Diseases. https://www.niaid.nih.gov/diseases-conditions/coronaviruses. Accessed 07 Aug 2020
WHO Coronavirus Disease (COVID-19) Dashboard | WHO Coronavirus Disease (COVID-19) Dashboard. https://covid19.who.int/. Accessed 7 Aug 2020
Chen Y, Liu Q, Guo D (2020) Emerging coronaviruses: genome structure, replication, and pathogenesis. J Med Virol 92(4):418–423. https://doi.org/10.1002/jmv.25681
Woo PC, Lau SK, Wong BH, Tsoi HW, Fung AM, Kao RY, Chan KH, Peiris JS, Yuen KY (2005) Differential sensitivities of severe acute respiratory syndrome (SARS) coronavirus spike polypeptide enzyme-linked immunosorbent assay (ELISA) and SARS coronavirus nucleocapsid protein ELISA for serodiagnosis of SARS coronavirus pneumonia. J Clin Microbiol 43(7):3054–3058. https://doi.org/10.1128/JCM.43.7.3054-3058.2005
Andersen KG, Rambaut A, Lipkin WI, Holmes EC, Garry RF (2020) The proximal origin of SARS-CoV-2. Nat Med 26(4):450–452. https://doi.org/10.1038/s41591-020-0820-9
Wrapp D, Wang N, Corbett KS, Goldsmith JA, Hsieh CL, Abiona O, Graham BS, McLellan JS (2020) Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science (New York, N.Y.) 367(6483):1260–1263. https://doi.org/10.1126/science.abb2507
Sharun K, Tiwari R, Iqbal Yatoo M, Patel SK, Natesan S, Dhama J et al (2020) Antibody-based immunotherapeutics and use of convalescent plasma to counter COVID-19: advances and prospects. Expert Opin Biol Ther. https://doi.org/10.1080/14712598.2020.1796963
Casadevall A, Pirofski LA (2020) The convalescent sera option for containing COVID-19. J Clin Investig 130(4):1545–1548. https://doi.org/10.1172/JCI138003
Valk SJ, Piechotta V, Chai KL, Doree C, Monsef I, Wood EM, Lamikanra A, Kimber C, McQuilten Z, So-Osman C, Estcourt LJ, Skoetz N (2020) Convalescent plasma or hyperimmune immunoglobulin for people with COVID-19: a rapid review. Cochrane Database Syst Rev 5(5):CD013600. https://doi.org/10.1002/14651858.CD013600
Takeda’s attempts to develop a COVID-19 treatment using blood plasma. https://www.clinicaltrialsarena.com/analysis/covid-19-treatment-takeda/. Accessed 7 Aug 2020
Shanmugaraj B, Siriwattananon K, Wangkanont K, Phoolcharoen W (2020) Perspectives on monoclonal antibody therapy as potential therapeutic intervention for Coronavirus disease-19 (COVID-19). Asian Pac J Allergy Immunol 38(1):10–18. https://doi.org/10.12932/AP-200220-0773
Tian X, Li C, Huang A, Xia S, Lu S, Shi Z, Lu L, Jiang S, Yang Z, Wu Y, Ying T (2020) Potent binding of 2019 novel coronavirus spike protein by a SARS coronavirus-specific human monoclonal antibody. Emerg Microbes Infect 9(1):382–385. https://doi.org/10.1080/22221751.2020.1729069
Hoffmann M, Kleine-Weber H, Schroeder S, Krüger N, Herrler T, Erichsen S, Schiergens TS, Herrler G, Wu NH, Nitsche A, Müller MA, Drosten C, Pöhlmann S (2020) SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell 181(2):271–280.e8. https://doi.org/10.1016/j.cell.2020.02.052
Díez JM, Romero C, Gajardo R (2020) Currently available intravenous immunoglobulin contains antibodies reacting against severe acute respiratory syndrome coronavirus 2 antigens. Immunotherapy 12(8):571–576. https://doi.org/10.2217/imt-2020-0095
Ju B, Zhang Q, Ge J et al (2020) Human neutralizing antibodies elicited by SARS-CoV-2 infection. Nature. https://doi.org/10.1038/s41586-020-2380-z
Wang C, Li W, Drabek D et al (2020) A human monoclonal antibody blocking SARS-CoV-2 infection. Nat Commun 11:2251. https://doi.org/10.1038/s41467-020-16256-y
Lei C, Qian K, Li T et al (2020) Neutralization of SARS-CoV-2 spike pseudotyped virus by recombinant ACE2-Ig. Nat Commun 11:2070. https://doi.org/10.1038/s41467-020-16048-4
Brouwer PJM, Caniels TG, van der Straten K, Snitselaar JL, Aldon Y, Bangaru S, van Gils MJ (2020) Potent neutralizing antibodies from COVID-19 patients define multiple targets of vulnerability. Science (New York, N.Y.) 369(6504):643–650. https://doi.org/10.1126/science.abc5902
Wu Y, Li C, Xia S, Tian X, Kong Y, Wang Z, Gu C, Zhang R, Tu C, Xie Y, Yang Z, Lu L, Jiang S, Ying T (2020) Identification of Human single-domain antibodies against SARS-CoV-2. Cell Host Microbe. https://doi.org/10.1016/j.chom.2020.04.023
Zeng X, Li L, Lin J, Li X, Liu B, Kong Y, Zeng S, Du J, Xiao H, Zhang T, Zhang S, Liu J (2020) Isolation of a human monoclonal antibody specific for the receptor binding domain of SARS-CoV-2 using a competitive phage biopanning strategy. Antib Ther. https://doi.org/10.1093/abt/tbaa008
Cohen J (2020) The race is on for antibodies that stop the new coronavirus. Science. https://doi.org/10.1126/science.abc6444
GigaGen Initiates Development of Recombinant Polyclonal Antibody Therapy for COVID-19|Antibodies|News Channels. https://pipelinereview.com/index.php/2020033174161/Antibodies/GigaGen-Initiates-Development-of-Recombinant-Polyclonal-Antibody-Therapy-for-COVID-19.html. Accessed 7 Aug 2020
Anti-COVID-19 antibody|Rabbit COVID 19 Spike Protein Coronavirus Polyclonal Antibody
COVID-19 Antibodies. https://www.raybiotech.com/covid-19-antibodies-sars-cov-2/. Accessed 7 Aug 2020
Abraham J (2020) Passive antibody therapy in COVID-19. Nat Rev Immunol. https://doi.org/10.1038/s41577-020-0365-7
Study shows good results with plasma treatment for COVID-19 - The Hindu. https://www.thehindu.com/sci-tech/health/study-shows-good-results-with-plasma-treatment-for-covid-19/article31194437.ece. Accessed 7 Aug 2020
Venkat Kumar G, Jeyanthi V, Ramakrishnan S (2020) A short review on antibody therapy for COVID-19. New Microbes New Infect. https://doi.org/10.1016/j.nmni.2020.100682
Duan K, Liu B, Li C, Zhang H, Yu T, Qu J, Zhou M, Chen L, Meng S, Hu Y, Peng C, Yuan M, Huang J, Wang Z, Yu J, Gao X, Wang D, Yu X, Li L, Zhang J et al (2020) Effectiveness of convalescent plasma therapy in severe COVID-19 patients. Proc Natl Acad Sci USA 117(17):9490–9496. https://doi.org/10.1073/pnas.2004168117
Zhang B, Liu S, Tan T, Huang W, Dong Y, Chen L, Chen Q, Zhang L, Zhong Q, Zhang X, Zou Y, Zhang S (2020) Treatment with convalescent plasma for critically ill patients with severe acute respiratory syndrome coronavirus 2 infection. Chest 158(1):e9–e13. https://doi.org/10.1016/j.chest.2020.03.039
Ahn JY, Sohn Y, Lee SH, Cho Y, Hyun JH, Baek YJ, Jeong SJ, Kim JH, Ku NS, Yeom JS, Roh J, Ahn MY, Chin BS, Kim YS, Lee H, Yong D, Kim HO, Kim S, Choi JY (2020) Use of convalescent plasma therapy in two COVID-19 patients with acute respiratory distress syndrome in Korea. J Korean Med Sci 35(14):e149. https://doi.org/10.3346/jkms.2020.35.e149
Rajendran K, Krishnasamy N, Rangarajan J, Rathinam J, Natarajan M, Ramachandran A (2020) Convalescent plasma transfusion for the treatment of COVID-19: systematic review. J Med Virol. https://doi.org/10.1002/jmv.25961
Shen C, Wang Z, Zhao F, Yang Y, Li J, Yuan J et al (2020) Treatment of 5 critically Ill patients with COVID-19 with convalescent plasma. JAMA 323(16):1582–1589. https://doi.org/10.1001/jama.2020.4783
Barone P, DeSimone RA (2020) Convalescent plasma to treat coronavirus disease 2019 (COVID-19): considerations for clinical trial design. Transfusion 60(6):1123–1127. https://doi.org/10.1111/trf.15843
Teixeira da Silva JA (2020) Convalescent plasma: a possible treatment of COVID-19 in India. Med J 76(2):236–237. https://doi.org/10.1016/j.mjafi.2020.04.006
Kazatchkine MD, Goldman M, Vincent JL (2020) Antibody-based therapies for COVID-19: can Europe move faster? PLoS Med 17(5):e1003127. https://doi.org/10.1371/journal.pmed.1003127
Wan Y, Shang J, Sun S, Tai W, Chen J, Geng Q, He L, Chen Y, Wu J, Shi Z, Zhou Y, Du L, Li F (2020) Molecular mechanism for antibody-dependent enhancement of coronavirus entry. J Virol 94(5):e02015–e2019. https://doi.org/10.1128/JVI.02015-19
Morabito CJ, Gangadharan B (2020) Active therapy with passive immunotherapy may be effective in the fight against COVID-19. Clin Transl Sci. https://doi.org/10.1111/cts.12816
Marovich M, Mascola JR, Cohen MS (2020) Monoclonal antibodies for prevention and treatment of COVID-19. JAMA. https://doi.org/10.1001/jama.2020.10245
Yang Y, Shen C, Li J, Yuan J, Yang M et al (2020) Exuberant elevation of IP-10, MCP-3 and IL-1ra during SARS-CoV-2 infection is associated with disease severity and fatal outcome. medRxiv. https://doi.org/10.1101/2020.03.02.20029975
Vaninov N (2020) In the eye of the COVID-19 cytokine storm. Nat Rev Immunol 20(5):277. https://doi.org/10.1038/s41577-020-0305-6
Ye Q, Wang B, Mao J (2020) The pathogenesis and treatment of the `Cytokine Storm' in COVID-19. J Infect 80(6):607–613. https://doi.org/10.1016/j.jinf.2020.03.037
Wang SX, Wang Y, Lu YB, Li JY, Song YJ, Nyamgerelt M, Wang XX (2020) Diagnosis and treatment of novel coronavirus pneumonia based on the theory of traditional Chinese medicine. J Integr Med 18(4):275–283. https://doi.org/10.1016/j.joim.2020.04.001
Zhong J, Tang J, Ye C, Dong L (2020) The immunology of COVID-19: is immune modulation an option for treatment? Lancet Rheumatol. https://doi.org/10.1016/S2665-9913(20)30120-X
Kawada J, Kitagawa Y, Iwata N, Ito Y (2013) Clinical characteristics of influenza virus infection in juvenile idiopathic arthritis patients treated with tocilizumab. Mod Rheumatol 23(5):972–976. https://doi.org/10.1007/s10165-012-0780-0
Fu B, Xu X, Wei H (2020) Why tocilizumab could be an effective treatment for severe COVID-19? J Transl Med 18(1):164. https://doi.org/10.1186/s12967-020-02339-3
Toniati P, Piva S, Cattalini M, Garrafa E, Regola F, Castelli F, Franceschini F, Airò P, Bazzani C, Beindorf EA, Berlendis M, Bezzi M, Bossini N, Castellano M, Cattaneo S, Cavazzana I, Contessi GB, Crippa M, Delbarba A, De Peri E et al (2020) Tocilizumab for the treatment of severe COVID-19 pneumonia with hyperinflammatory syndrome and acute respiratory failure: a single center study of 100 patients in Brescia. Italy Autoimmunity Rev 19(7):102568. https://doi.org/10.1016/j.autrev.2020.102568
Colaneri M, Bogliolo L, Valsecchi P, Sacchi P, Zuccaro V, Brandolino F, Montecucco C, Mojoli F, Giusti EM, Bruno R, The Covid Irccs San Matteo Pavia Task Force (2020) Tocilizumab for treatment of severe COVID-19 patients: preliminary results from SMAtteo COvid19 REgistry (SMACORE). Microorganisms 8(5):695. https://doi.org/10.3390/microorganisms8050695
Zhang S, Li L, Shen A, Chen Y, Qi Z (2020) Rational use of Tocilizumab in the treatment of novel coronavirus pneumonia. Clin Drug Investig 40(6):511–518. https://doi.org/10.1007/s40261-020-00917-3
Clinical Trials Arena (n.d.) Roche to start Phase III trial of Actemra in Covid-19 patients. https://www.clinicaltrialsarena.com/news/roche-actemra-covid-19-trial/
Scala S, Pacelli R (2020) Fighting the host reaction to SARS-COv-2 in critically ill patients: the possible contribution of off-label drugs. Front Immunol 11:1201. https://doi.org/10.3389/fimmu.2020.01201
Magro G (2020) COVID-19: review on latest available drugs and therapies against SARS-CoV-2. Coagulation and inflammation cross-talking. Virus Res 286:198070. https://doi.org/10.1016/j.virusres.2020.198070
Rodrigues-Diez RR, Tejera-Muñoz A, Marquez-Exposito L, Rayego-Mateos S, Santos Sanchez L, Marchant V et al (2020) Statins: could an old friend help in the fight against COVID-19? Br J Pharmacol. https://doi.org/10.1111/bph.15166
Mehta P, Porter JC, Manson JJ, Isaacs JD, Openshaw PJM, McInnes IB et al (2020) Therapeutic blockade of granulocyte macrophage colony-stimulating factor in COVID-19-associated hyperinflammation: challenges and opportunities. Lancet Respir Med. https://doi.org/10.1016/s2213-2600(20)30267-8
Zhao M (2020) Cytokine storm and immunomodulatory therapy in COVID-19: role of chloroquine and anti-IL-6 monoclonal antibodies. Int J Antimicrob Agents 55(6):105982. https://doi.org/10.1016/j.ijantimicag.2020.105982
Schrezenmeier E, Dörner T (2020) Mechanisms of action of hydroxychloroquine and chloroquine: implications for rheumatology. Nat Rev Rheumatol 16(3):155–166. https://doi.org/10.1038/s41584-020-0372-x
Tanaka T, Narazaki M, Kishimoto T (2016) Immunotherapeutic implications of IL-6 blockade for cytokine storm. Immunotherapy 8(8):959–970. https://doi.org/10.2217/imt-2016-0020
Zhang W, Zhao Y, Zhang F, Wang Q, Li T, Liu Z, Wang J, Qin Y, Zhang X, Yan X, Zeng X, Zhang S (2020) The use of anti-inflammatory drugs in the treatment of people with severe coronavirus disease 2019 (COVID-19): the perspectives of clinical immunologists from China. Clin Immunol (Orlando, Fla.) 214:108393. https://doi.org/10.1016/j.clim.2020.108393
Bhaskar S, Sinha A, Banach M, Mittoo S, Weissert R, Kass JS et al (2020) Cytokine storm in COVID-19—immunopathological mechanisms, clinical considerations, and therapeutic approaches: the REPROGRAM Consortium Position Paper. Front Immunol. https://doi.org/10.3389/fimmu.2020.01648
Singh H, Kakkar AK, Chauhan P (2020) Repurposing minocycline for COVID-19 management: mechanisms, opportunities, and challenges. Expert Rev Anti-Infect Ther. https://doi.org/10.1080/14787210.2020.1782190
Subramanian S, Iles T, Ikramuddin S, Steer CJ (2020) Merit of an ursodeoxycholic acid clinical trial in COVID-19 patients. Vaccines 8(2):E320. https://doi.org/10.3390/vaccines8020320
Seifirad S (2020) Pirfenidone: a novel hypothetical treatment for COVID-19. Med Hypotheses 144:110005. https://doi.org/10.1016/j.mehy.2020.110005
Khalil A, Kamar A, Nemer G (2020) Thalidomide-revisited: are COVID-19 patients going to be the latest victims of yet another theoretical drug-repurposing? Front Immunol 11:1248. https://doi.org/10.3389/fimmu.2020.01248
McCord JM, Hybertson BM, Cota-Gomez A, Geraci KP, Gao B (2020) Nrf2 activator pb125® as a potential therapeutic agent against covid-19. Antioxidants 9(6):1–15. https://doi.org/10.3390/antiox9060518
McKee DL, Sternberg A, Stange U, Laufer S, Naujokat C (2020) Candidate drugs against SARS-CoV-2 and COVID-19. Pharmacol Res 157:104859. https://doi.org/10.1016/j.phrs.2020.104859
Venkatasubbaiah M, Dwarakanadha Reddy P, Satyanarayana SV (2020) Literature-based review of the drugs used for the treatment of COVID-19. Curr Med Res Pract 10(3):100–109. https://doi.org/10.1016/j.cmrp.2020.05.013
Experimental Drug Helps Some Americans Ride Out Coronavirus, NIH Doctor Says - WSJ (n.d.) https://www.wsj.com/articles/experimental-drug-helps-some-americans-ride-out-coronavirus-nih-doctor-says-11584094955
COVID-19: CLINICAL INFORMATION AND TREATMENT GUIDELINES INTERNATIONAL PHARMACEUTICAL FEDERATION. PDF
Jean SS, Lee PI, Hsueh PR (2020) Treatment options for COVID-19: The reality and challenges. J Microbiol Immunol Infect. https://doi.org/10.1016/j.jmii.2020.03.034
Holshue ML, DeBolt C, Lindquist S, Lofy KH, Wiesman J, Bruce H, Spitters C, Ericson K, Wilkerson S, Tural A, Diaz G, Cohn A, Fox LA, Patel A, Gerber SI, Kim L, Tong S, Lu X, Lindstrom S et al (2020) First case of 2019 novel coronavirus in the United States. N Engl J Med. https://doi.org/10.1056/NEJMoa2001191
VigiAccess: Uppsala Monitoring Centre. https://www.vigiaccess.org/. Accessed 7 Aug 2020
Coronavirus pandemic: India approves emergency use of remdesivir to treat COVID-19 patients - health - Hindustan Times. https://www.hindustantimes.com/health/coronavirus-pandemic-india-approves-emergency-use-of-remdesivir-to-treat-covid-19-patients/story-lvL4HNnOOvTR2Yo3UsyEBI.html. Accessed 7 Aug 2020
These are the coronavirus quarantine facilities in India | Business Insider India (n.d.) https://www.businessinsider.in/india/news/these-are-the-coronavirus-quarantine-facilities-in-delhi-mumbai-hyderabad-and-other-parts-of-india/articleshow/74650950.cms
Bhatnagar T, Murhekar MV, Soneja M, Gupta N, Giri S, Wig N, Gangakhedkar R (2020) Lopinavir/ritonavir combination therapy amongst symptomatic coronavirus disease 2019 patients in India: protocol for restricted public health emergency use. Indian J Med Res 151(2 & 3):184–189. https://doi.org/10.4103/ijmr.IJMR_502_20
Search Result-DrugBank. https://www.drugbank.ca/unearth/q?utf8=%E2%9C%93&query=lopinavir%2Fritonivir&searcher=drugs. Accessed 7 Aug 2020
Triage: Early recognition of patients with COVID-19 (n.d.) https://mohfw.gov.in/media/disease-alerts
Cao B, Wang Y, Wen D, Liu W, Wang J, Fan G, Ruan L, Song B, Cai Y, Wei M, Li X, Xia J, Chen N, Xiang J, Yu T, Bai T, Xie X, Zhang L, Li C, Yuan Y et al (2020) A trial of lopinavir-ritonavir in adults Hospitalized with Severe Covid-19. N Engl J Med 382(19):1787–1799. https://doi.org/10.1056/NEJMoa2001282
Oxford scientists say HIV drugs lopinavir and ritonavir ‘not effective’ for hospitalised Covid-19 patients | Health Tips and News. https://www.timesnownews.com/health/article/oxford-scientists-say-hiv-drugs-lopinavir-and-ritonavir-not-effective-for-hospitalised-covid-19-patients/614580. Accessed 18 Aug 2020
Furuta Y, Gowen BB, Takahashi K, Shiraki K, Smee DF, Barnard DL (2013) Favipiravir (T-705), a novel viral RNA polymerase inhibitor. Antivir Res 100(2):446–454. https://doi.org/10.1016/j.antiviral.2013.09.015
Delang L, Abdelnabi R, Neyts J (2018) Favipiravir as a potential countermeasure against neglected and emerging RNA viruses. Antivir Res 153:85–94. https://doi.org/10.1016/j.antiviral.2018.03.003
Favipiravir shows good clinical efficacy in treating COVID-19: official - Xinhua | English.news.cn. https://www.xinhuanet.com/english/2020-03/17/c_138888102.htm. Accessed 7 Aug 2020
Stanford Medicine trial to test favipiravir for treating COVID-19 outpatients |News Center |Stanford Medicine. https://med.stanford.edu/news/all-news/2020/06/stanford-medicine-trial-tests-favipiravir-for-covid-19.html. Accessed 7 Aug 2020
Fast Check: Here's All You Need to Know About 'Favipiravir', New Covid-19 Medicine. https://thelogicalindian.com/fact-check/glenmark-pharmaceuticals-drug-controller-general-of-india-favipiravir-anti-viral-covid-19-21843. Accessed 7 Aug 2020
Peiris JS, Chu CM, Cheng VC, Chan KS, Hung IF, Poon LL, Law KI, Tang BS, Hon TY, Chan CS, Chan KH, Ng JS, Zheng BJ, Ng WL, Lai RW, Guan Y, Yuen KY, HKU/UCH SARS Study Group (2003) Clinical progression and viral load in a community outbreak of coronavirus-associated SARS pneumonia: a prospective study. Lancet (London, England) 361(9371):1767–1772. https://doi.org/10.1016/s0140-6736(03)13412-5
Vincent MJ, Bergeron E, Benjannet S, Erickson BR, Rollin PE, Ksiazek TG et al (2005) Chloroquine is a potent inhibitor of SARS coronavirus infection and spread. Virol J. https://doi.org/10.1186/1743-422X-2-69
Yao X, Ye F, Zhang M, Cui C, Huang B, Niu P, Liu X, Zhao L, Dong E, Song C, Zhan S, Lu R, Li H, Tan W, Liu D (2020) In vitro antiviral activity and projection of optimized dosing design of hydroxychloroquine for the treatment of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). Clin Infect Dis. https://doi.org/10.1093/cid/ciaa237
WHO halts hydroxychloroquine trial for coronavirus amid safety fears World news| The Guardian https://www.theguardian.com/world/2020/may/25/who-world-health-organization-hydroxychloroquine-trial-trump-coronavirus-safety-fears Accessed 7 Aug 2020
WHO restarts HCQ trial after Lancet concern over paper that trashed it| coronavirus outbreak News The Indian Express https://indianexpress.com/article/coronavirus/who-hydroxychloroquine-trail-restart-lancet-6441507/ Accessed 7 Aug 2020
Coronavirus (COVID-19) Update: FDA Revokes Emergency Use Authorization for Chloroquine and Hydroxychloroquine | FDA. https://www.fda.gov/news-events/press-announcements/coronavirus-covid-19-update-fda-revokes-emergency-use-authorization-chloroquine-and. Accessed 7 Aug 2020
Poschet JF, Perkett EA, Timmins GS, Deretic V (2020) Azithromycin and ciprofloxacin have a chloroquine-like effect on respiratory epithelial cells. bioRxiv. https://doi.org/10.1101/2020.03.29.008631
Kastritis E, Kitas GD, Vassilopoulos D, Giannopoulos G, Dimopoulos MA, Sfikakis PP (2020) Systemic autoimmune diseases, anti-rheumatic therapies, COVID-19 infection risk and patient outcomes. Rheumatol Int 40(9):1353–1360. https://doi.org/10.1007/s00296-020-04629-x
Gautret P, Lagier JC, Parola P, Hoang VT, Meddeb L, Mailhe M, Doudier B, Courjon J, Giordanengo V, Vieira VE, Tissot Dupont H, Honoré S, Colson P, Chabrière E, La Scola B, Rolain JM, Brouqui P, Raoult D (2020) Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial. Int J Antimicrob Agents. https://doi.org/10.1016/j.ijantimicag.2020.105949
Fanin A, Calegari J, Beverina A, Tiraboschi S (2020) Hydroxychloroquine and azithromycin as a treatment of COVID-19. Intern Emerg Med. https://doi.org/10.1007/s11739-020-02388-y
Graci JD, Cameron CE (2006) Mechanisms of action of ribavirin against distinct viruses. Rev Med Virol 16(1):37–48. https://doi.org/10.1002/rmv.483
Muller MP, Richardson SE, McGeer A, Dresser L, Raboud J, Mazzulli T, Loeb M, Louie M, Canadian SARS Research Network (2006) Early diagnosis of SARS: lessons from the Toronto SARS outbreak. Eur J Clin Microbiol Infect Dis 25(4):230–237. https://doi.org/10.1007/s10096-006-0127-x
Falzarano D, de Wit E, Martellaro C et al (2013) Inhibition of novel β coronavirus replication by a combination of interferon-α2b and ribavirin. Sci Rep 3:1686. https://doi.org/10.1038/srep01686
Kim UJ, Won EJ, Kee SJ, Jung SI, Jang HC (2016) Combination therapy with lopinavir/ritonavir, ribavirin and interferon-α for Middle East respiratory syndrome. Antivir Ther 21(5):455–459. https://doi.org/10.3851/IMP3002
Khalili JS, Zhu H, Mak N, Yan Y, Zhu Y (2020) Novel coronavirus treatment with ribavirin: groundwork for an evaluation concerning COVID-19. J Med Virol 92(7):740–746. https://doi.org/10.1002/jmv.25798
Hung IFN, Lung KC, Tso EYK, Liu R, Chung TWH, Chu MY et al (2020) Triple combination of interferon beta-1b, lopinavir–ritonavir, and ribavirin in the treatment of patients admitted to hospital with COVID-19: an open-label, randomised, phase 2 trial. Lancet 395(10238):1695–1704. https://doi.org/10.1016/S0140-6736(20)31042-4
Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, Zhang L, Fan G, Xu J, Gu X, Cheng Z, Yu T, Xia J, Wei Y, Wu W, Xie X, Yin W, Li H, Liu M, Xiao Y et al (2020) Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet (London, England) 395(10223):497–506. https://doi.org/10.1016/S0140-6736(20)30183-5
Zeng YM, Xu XL, He XQ, Tang SQ, Li Y, Huang YQ et al (2020) Comparative effectiveness and safety of ribavirin plus interferon-alpha, lopinavir/ritonavir plus interferon-alpha, and ribavirin plus lopinavir/ritonavir plus interferon-alpha in patients with mild to moderate novel coronavirus disease 2019: study protocol. Chin Med J. https://doi.org/10.1097/CM9.0000000000000790
TCM Differentiation and Treatment Protocol of COVID-19- Full Text View- ClinicalTrails.gov. https://www.clinicaltrials.gov/ct2/show/NCT04306497. Accessed 7 Aug 2020
Covaxin status: Human clinical trials of indigenous Covid-19 vaccine begin in Chennai hospital | Chennai News - Times of India. https://timesofindia.indiatimes.com/city/chennai/covaxin-human-clinical-trials-for-indigenous-covid-19-vaccine-begins-in-chennai-hospital/articleshow/77123194.cms. Accessed 7 Aug 2020
https://www.expresspharma.in/covid19-updates/zydus-starts-human-trials-of-zycov-d-its-covid-19-vaccine/. Accessed 7 Aug 2020
Coronavirus vaccine: Oxford-Astrazeneca COVID-19 vaccine is winning praises, but here's why we should refrain from building false hopes | The Times of India. https://timesofindia.indiatimes.com/life-style/health-fitness/health-news/coronavirus-vaccine-oxford-astrazeneca-covid-19-vaccine-is-winning-praises-but-heres-why-we-should-refrain-from-building-false-hopes/photostory/77123445.cms. Accessed 7 Aug 2020
Bar-Zeev N, Moss WJ (2020) Encouraging results from phase 1/2 COVID-19 vaccine trials. Lancet. https://doi.org/10.1016/S0140-6736(20)31611-1
Oxford Coronavirus (COVID19) Vaccine latest update: Oxford-AstraZeneca shot shows progress: What does this mean in fight to find Covid-19 Vaccine. https://indianexpress.com/article/explained/oxford-astrazeneca-shot-shows-progress-what-does-this-mean-in-fight-to-find-covid-19-vaccine-6515316/ Accessed 7 Aug 2020
https://www.modernatx.com/modernas-work-potential-vaccine-against-covid-19. Accessed 7 Aug 2020
Pfizer and BioNTech Choose Lead mRNA Vaccine Candidate Against COVID-19 and Commence Pivotal Phase 2/3 Global Study | Pfizer. https://www.pfizer.com/news/press-release/press-release-detail/pfizer-and-biontech-choose-lead-mrna-vaccine-candidate-0. Accessed 7 Aug 2020
COVID-19 Treatment Dashboard. https://www.quiverquant.com/sources/covidTreatment. Accessed 7 Aug 2020
Bray M, Rayner C, Noël F, Jans D, Wagstaff K (2020) Ivermectin and COVID-19: a report in Antiviral Research, widespread interest, an FDA warning, two letters to the editor and the authors' responses. Antivir Res 178:104805. https://doi.org/10.1016/j.antiviral.2020.104805
https://www.news-medical.net/news/20200507/Predicting-the-dose-for-repurposing-nitazoxanide-in-COVID-19.aspx. Accessed 7 Aug 2020
COVID-19 treatment update: Here is a list of all drugs used in India; their pros and cons https://www.msn.com/en-in/news/other/covid-19-treatment-update-here-is-a-list-of-all-drugs-used-in-india-their-pros-and-cons/ar-BB16HTF2. Accessed 7 Aug 2020
Coronavirus Disease 2019 (COVID-19) Treatment Guidelines. https://www.covid19treatmentguidelines.nih.gov/. Accessed 7 Aug 2020
Chen, J., Gao, K., Wang, R., Nguyen, D. D., & Wei, G.-W. (2020). Review of COVID-19 Antibody Therapies. ArXiv. https://www.ncbi.nlm.nih.gov/pubmed/32601601https://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=PMC7316097
Novartis announces plan to initiate clinical trial of canakinumab for patients with COVID-19 pneumonia (2020) https://www.novartis.com/news/novartis-announces-plan-initiate-clinical-trial-canakinumab-patients-covid-19-pneumonia
Acknowledgements
Authors are grateful to Bennett University, Greater Noida, for providing the providential opportunity and desired assistance.
Funding
No funding was received to prepare this review.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest:
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Tandon, S., Aggarwal, A., Jain, S. et al. Perspective on the Role of Antibodies and Potential Therapeutic Drugs to Combat COVID-19. Protein J 39, 631–643 (2020). https://doi.org/10.1007/s10930-020-09921-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10930-020-09921-0