Abstract
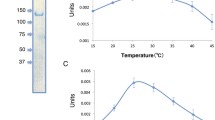
The kinetic affinity for CO2 of phosphoenolpyruvate PEP5 carboxykinase from Anaerobiospirillum succiniciproducens, an obligate anaerobe which PEP carboxykinase catalyzes the carboxylation of PEP in one of the final steps of succinate production from glucose, is compared with that of the PEP carboxykinase from Saccharomyces cerevisiae, which catalyzes the decarboxylation of oxaloacetate in one of the first steps in the biosynthesis of glucose. For the A. succiniciproducens enzyme, at physiological concentrations of Mn2+ and Mg2+, the affinity for CO2 increases as the ATP/ADP ratio is increased in the assay medium, while the opposite effect is seen for the S. cerevisiae enzyme. The results show that a high ATP/ADP ratio favors CO2 fixation by the PEP carboxykinase from A. succiniciproducens but not for the S. cerevisiae enzyme. These findings are in agreement with the proposed physiological roles of S. cerevisiae and A. succiniciproducens PEP carboxykinases, and expand recent observations performed with the enzyme isolated from Panicum maximum (Chen et al. (2002) Plant Physiology 128: 160–164).

Similar content being viewed by others
Abbreviations
- PEP:
-
phosphoenolpyruvate
- OAA:
-
oxaloacetate
- MOPS:
-
3-(N-morpholino) propanesulfonic acid
References
Cannata J. J. B., Stoppani A. O. M. (1963) J. Biol. Chem. 238:1919–1927
Cazzulo J. J. (1992) FASEB J. 6:3153–3161
Chang H. C., Lane M. D. (1966) J. Biol. Chem. 241:2413–2420
Chen Z - H., Walker R. P., Acheson R. M., Leeggod R. C. (2002). Plant. Physiology, 128:160–164
Davis C. P., Cleven D., Brown J., Balish E. (1976) Int. J. Syst. Bacteriol. 26:498–504
Encinas M. V., Rojas M. C., Goldie H., Cardemil H. (1993) Biochim. Biophys. Acta 1162:195–202
Gancedo C., Schwerzmann K. (1976) Arch. Microbiol. 109:221–225
Hunt M., Köhler P. (1995) Biochim. Biophys. Acta 1249:15–22
Jabalquinto, A. M., Laivenieks M., Zeikus J. G., and Cardemil E. (1999). J. Prot. Chem. 18: 659–664
Krautwurst H., Bazaes S., González F. D., Jabalquinto A. M., Frey P., Cardemil E. (1998) Biochemistry 37:6295–6302
Laivenieks M., Vieille C., Zeikus J. G. (1997) Appl. Environ. Microbiol. 63:2273–2280
Leegood, R. C., and Walker, R. P. (2003). Arch. Biochem. Biophys. 414: 204–210.
Llanos L., Briones R., Yévenes, A., González-Nilo F. D., Frey P., and Cardemil, E. (2001). FEBS Lett. 493: 1–5
Martel, A. E., and Smith, R. M. (1998). NIST critically selected stability constants of metal complexes. NSIT standard references database 45 (1998) version 5.0
Podkovyrov S., Zeikus J. G. (1993. J. Gen. Microbiol. 139:223–228
Reiskind, J. B., and Bowes, G. (1991). Proc. Natl. Academ. Sci. USA 88: 2883–2887.
Utter M. F., and Kolenbrander H. M. (1972). In: Boyer, P. (ed.), The Enzymes, 3rd ed., vol 6, Academic Press, New York, pp. 117–168
Walker, R. P., Acheson, R. M., Técsi, L. I., and Leegood, R. C. (1997). Aust. J. Physiol. 24: 450–468.
Acknowledgments
This work was supported by research grant FONDECYT 1030760.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bazaes, S., Toncio, M., Laivenieks, M. et al. Comparative Kinetic Effects of Mn (II), Mg (II) and the ATP/ADP Ratio on Phosphoenolpyruvate Carboxykinases from Anaerobiospirillum succiniciproducens and Saccharomyces cerevisiae . Protein J 26, 265–269 (2007). https://doi.org/10.1007/s10930-006-9068-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10930-006-9068-6