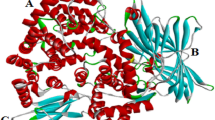
Although TauA shares few common characteristics with other known periplasmic binding protein, TauA is a putative periplasmic binding protein, part of tauABCD gene cluster involved in sulfonate transport in sulphate starvation condition. This protein was expressed in E. coli BL 21 and purified before to assess its binding functionalities. Measurement of K d value (mean 11.3 nM) by binding/dialysis studies revealed high affinity and specificity with taurine and also indicated that TauA possessed a unique binding site for its ligand. Comparisons with other periplasmic binding proteins suggests TauA plays a major role in ABC transport system and could be ideal candidate to serve as taurine catcher in biological fluids.


Similar content being viewed by others
Abbreviations
- Cys:
-
cysteine
- E coli :
-
Eschericia coli
- ATP:
-
adenosine tri phospate
- HEPES:
-
4–(2-hydroxyethyl)-piperazine-1-ethanesulfonic acid
- MOPS:
-
3-(N-morpholino)-propanesulfonic acid
- IPTG:
-
isopropyl-β-thiogalactopyranoside
- IMAC:
-
immobilized metal ion chromatography
- TCA:
-
trichloroacetic acid
- HPLC:
-
high pressure liquid chromatography
- MWCO:
-
molecular weight cutt-off
- OD:
-
optical density
- Ssi:
-
sulfate starvation induced protein
- RT-PCR:
-
reverse transcriptase polymerase chain reaction
- ORF:
-
open reading frame
- LIV:
-
leucine-isoleucine-valine binding protein
- HBP:
-
histidine binding protein
- η:
-
the fractional saturation of protein molecule that are saturated with ligand
References
Ames G. F. L. (1986). Annu. Rev. Biochem. 55: 397–425
Ames G. F. L., Mimura C. S., Shyamala V. (1990). FEMS Microbiol Rev 75: 429–446
Ames G. F. L., Joshi K. A. (1990). J. Bacteriol. 172(8): 4133–4137
Berger E. A., Heppel L. A. (1972). J. Biol. Chem. 247(23): 7684–7694
Blattner F. R., Plunkett G. (3rd), Bloch C. A., Perna N. T., Burland V., Riley M., Collado-Vides J., Glasner J. D., Rode C. K., Mayhew G. F., Gregor J., Davis N. W., Kirkpatrick H. A., Goeden M. A., Rose D. J., Mau B., Shao Y. (1997). Science, 277: 1453–1474
Boos, W., and Lucht, J. M. (1996). In: Neidhardt, F. C., Curtis III, R., Ingraham, J.L., Lin, E. C. C., Low, K. B., Magasanik, B., Reznikoff, W. S., Riley, M., Schaechter, M. and Umbarger, H. E. (eds.) Escherichia coli and Salmonella: cellular and molecular biology, 2nd ed. vol. 1, ASM Press, Washington, DC, pp. 1175–1209
Dahchour A., Quertemont E, De Witte P. (1996). Brain Res. 735: 9–19
Della Corte L., Crichton R. R., Duburs G., Nolan K., Tipton K. F., Tirzitis G., Ward R. J. (2002). Amino Acids. 23: 367–379
Eichhorn E., Van der Ploeg J. R., Kertesz M. A., Leisinger T. (1997). J. Biol. Chem. 272(37): 23031–23036
Eichhorn E., Van Der Ploeg J. R., Leisinger T. (1999). J. Biol. Chem. 274(38): 26639–26646
Eichhorn E., Van Der Ploeg J. R., Leisinger T. (2000). J. Bacteriol 182(10): 2687–2695
Higgins C. F., Gibson M. M. (1986). Methods Enzymol 125: 365–377
Higgins C. F. (1992). Annu. Rev. Cell. Biol. 8: 67–113
Huxtable R. J. (1992). Physiol. Rev. 72(1): 101–163
Kertesz M. A., Leisinger T., Cook A. M. (1993). J. Bacteriol. 175(4): 1187–1190
Kredich, N. M. (1996). In: Neidhardt, F. C., Curtis III, R., Ingraham, J. L., Lin, E. C. C., Low, K. B., Magasanik, B., Reznikoff, W. S., Riley, M., Schacchter, M., and Umbarger, H. E. (eds.), Escherichia coli and Salmonella: cellular and molecular biology, 2nd ed., Amercican Society for Microbiology, Washington, DC, pp. 514–527
Lie T. J., Leadbetter J. R., Leadbetter E. R. (1998). Geomicrobiol. J. 15: 135–149
Militante J. D., Lombardini J. B. (2002). Amino Acids. 23: 381–393
Miller D. M., Olson J. S., Pflugrath J. W., Quicho F. A. (1983). J. Biol. Chem. 258(22): 13665–13672
Olive M. F. (2002). Amino Acids 23: 345–357
Oshima R. G., Willis R. C., Furlong C. E., Sneider J. A. (1974). J. Biol. Chem. 249(19): 6033–6039
Quadroni M., Staudenmann W., Kertesz M., James P. (1996). Eur. J. Biochem. 239: 773–781
Quadroni M., James P., Dainese-Hatt P., Kertesz M. A. (1999). Eur. J. Biochem. 266: 989–996
Quertemont E., Dahchour A., Ward R. J., De Witte P. (1999). Addict. Biol. 4: 47–54
Quirk P. G., Guffanti A. A., Clejan S., Cheng J., Krulwich T. A. (1994). Biochim. Biophys. Acta 1186: 27–34
Rosenberg H., Gerdes R. G., Chegwidden K. (1977). J. Bacteriol. 131: 505–511
Santangelo F. (2002). Amino Acids 23: 359–365
Saurin W., Dassa E. (1994). Protein Sc. 3: 325–344
Scatchard G., (1949). Ann. NY Acad. Sci. 51: 660–672
Sekowska A., Kung H. F., Danchin A. (2000). J. Mol. Microbiol. Biotechnol. 2(2): 145–177
Szmelcman S., Scwartz M., Silhavy T. J., Boos W. (1976). Eur. J. Biochem. 65: 13–19
Tam R., Saier M. J. R. (1993). Microbiol. Rev. 57(2): 320–346
Trainer V. L., Baden D. G. (1999). Aquat. Toxicol. 46: 139–148
Van der Ploeg J. R., Weiss M. A., Saller E., Nashimoto H., Saito N., Kertesz M. A., Leisinger T. (1996). J. Bacteriol. 178(18): 5438–5446
Van der Ploeg J. R., Iwanicka-Nowicka R., Kertesz M. A., Leisinger T., Hryniewicz M. M. (1997). J. Bacteriol. 179(24): 7671–7678
Van der Ploeg J. R., Cummings N. J., Leisinger T., Connerton I. F. (1998). Microbiology 144(9): 2555–2561
Van der Ploeg J. R., Iwanicka-Nowickas R., Hryniewicz M. M., Leisinger T., (1999). J. Biol. Chem. 274(8): 29538–29365
Van der Ploeg J. R., Eichhorn E., Leisinger T. (2001). Arch Microbiol 176: 1–8
Van Zoelen E. J., Kramer R. H., van Moerkerk H. T., Veerkamp J. H. (1998). Trends Pharmacol. Sci. 19(12): 487–90
Vermeij P., Wietek C., Kahnert A., Wüest T., Kertesz M. A. (1999). Mol. Microbiol. 32(5): 913–926
Ward R. J., Marshall E., Ball D., Martinez J., De Witte P. (1998). Neurosci. Res. Commun. 24(1): 41–49
Yogoshi H., Oda H. (2002). Amino Acids 23: 433–439
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Javaux, C., Joris, B. & De Witte, P. Functional Characteristics of TauA Binding Protein from TauABC Escherichia coli System. Protein J 26, 231–238 (2007). https://doi.org/10.1007/s10930-006-9064-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10930-006-9064-x