Abstract
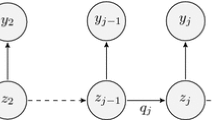
Parameter variation in pharmacometric analysis studies can be characterized as within subject parameter variability (WSV) in pharmacometric models. WSV has previously been successfully modeled using inter-occasion variability (IOV), but also stochastic differential equations (SDEs). In this study, two approaches, dynamic inter-occasion variability (dIOV) and adapted stochastic differential equations, were proposed to investigate WSV in pharmacometric count data analysis. These approaches were applied to published count models for seizure counts and Likert pain scores. Both approaches improved the model fits significantly. In addition, stochastic simulation and estimation were used to explore further the capability of the two approaches to diagnose and improve models where existing WSV is not recognized. The results of simulations confirmed the gain in introducing WSV as dIOV and SDEs when parameters vary randomly over time. Further, the approaches were also informative as diagnostics of model misspecification, when parameters changed systematically over time but this was not recognized in the structural model. The proposed approaches in this study offer strategies to characterize WSV and are not restricted to count data.




Similar content being viewed by others
References
Karlsson MO, Sheiner LB (1993) The importance of modeling interoccasion variability in population pharmacokinetic analyses. J Pharmacokinet Biopharm 21(6):735–750
Cramer JA, French J (2001) Quantitative assessment of seizure severity for clinical trials: a review of approaches to seizure components. Epilepsia 42(1):119–129
Troconiz IF, Plan EL, Miller R, Karlsson MO (2009) Modelling overdispersion and Markovian features in count data. J Pharmacokinet Pharmacodyn 36(5):461–477. doi:10.1007/s10928-009-9131-y
Plan EL, Elshoff JP, Stockis A, Sargentini-Maier ML, Karlsson MO (2012) Likert pain score modeling: a Markov integer model and an autoregressive continuous model. Clin Pharmacol Ther 91(5):820–828. doi:10.1038/clpt.2011.301
Tornoe CW, Jacobsen JL, Madsen H (2004) Grey-box pharmacokinetic/pharmacodynamic modelling of a euglycaemic clamp study. J Math Biol 48(6):591–604. doi:10.1007/s00285-003-0257-z
Tornoe CW, Agerso H, Senderovitz T, Nielsen HA, Madsen H, Karlsson MO, Jonsson EN (2007) Population pharmacokinetic/pharmacodynamic (PK/PD) modelling of the hypothalamic-pituitary-gonadal axis following treatment with GnRH analogues. Br J Clin Pharmacol 63(6):648–664. doi:10.1111/j.1365-2125.2006.02820.x
Tornoe CW, Overgaard RV, Agerso H, Nielsen HA, Madsen H, Jonsson EN (2005) Stochastic differential equations in NONMEM: implementation, application, and comparison with ordinary differential equations. Pharm Res 22(8):1247–1258. doi:10.1007/s11095-005-5269-5
Lönnebo A, Grahnén A, Karlsson MO (2007) An integrated model for the effect of budesonide on ACTH and cortisol in healthy volunteers. Br J Clin Pharmacol 64(2):125–132. doi:10.1111/j.1365-2125.2007.02867.x
Nagaraja NV, Pechstein B, Erb K, Klipping C, Hermann R, Locher M, Derendorf H (2003) Pharmacokinetic/pharmacodynamic modeling of luteinizing hormone (LH) suppression and LH surge delay by cetrorelix after single and multiple doses in healthy premenopausal women. J Clin Pharmacol 43(3):243–251
Lindbom L, Pihlgren P, Jonsson EN (2005) PsN-Toolkit–a collection of computer intensive statistical methods for non-linear mixed effect modeling using NONMEM. Comput Methods Programs Biomed 79(3):241–257. doi:10.1016/j.cmpb.2005.04.005
Keizer RJ, Karlsson MO, Hooker A (2013) Modeling and simulation workbench for NONMEM: tutorial on Pirana, PsN, and Xpose. CPT 2:e50. doi:10.1038/psp.2013.24
Plan EL (2014) Modeling and simulation of count data. CPT 3(8):1–12. doi:10.1038/psp.2014.27
Wahlby U, Thomson AH, Milligan PA, Karlsson MO (2004) Models for time-varying covariates in population pharmacokinetic-pharmacodynamic analysis. Br J Clin Pharmacol 58(4):367–377. doi:10.1111/j.1365-2125.2004.02170.x
Lacroix BD, Lovern MR, Stockis A, Sargentini-Maier ML, Karlsson MO, Friberg LE (2009) A pharmacodynamic Markov mixed-effects model for determining the effect of exposure to certolizumab pegol on the ACR20 score in patients with rheumatoid arthritis. Clin Pharmacol Ther 86(4):387–395. doi:10.1038/clpt.2009.136
Karlsson MO, Holford N (2008) A tutorial on visual predictive checks. Paper presented at the PAGE, Marseille, France
Acknowledgments
The research leading to these results has received support from the Innovative Medicines Initiative Joint Undertaking under Grant Agreement No. 115156, resources of which are composed of financial contributions from the European Union’s Seventh Framework Programme (FP7/2007–2013) and EFPIA companies’ in kind contribution. The DDMoRe project is also supported by financial contribution from Academic and SME partners. The authors thank Dr. Christoffer W. Tornøe for his helpful inputs of SDEs.
Author Contributions
CD., E.P. and M.K. designed the research. C.D. performed the research. C.D, E.P. and M.K. analyzed the results. C.D., E. P. and M.K. wrote the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Chenhui Deng was employed by Pfizer when this paper was submitted.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Deng, C., Plan, E.L. & Karlsson, M.O. Approaches for modeling within subject variability in pharmacometric count data analysis: dynamic inter-occasion variability and stochastic differential equations. J Pharmacokinet Pharmacodyn 43, 305–314 (2016). https://doi.org/10.1007/s10928-016-9473-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10928-016-9473-1